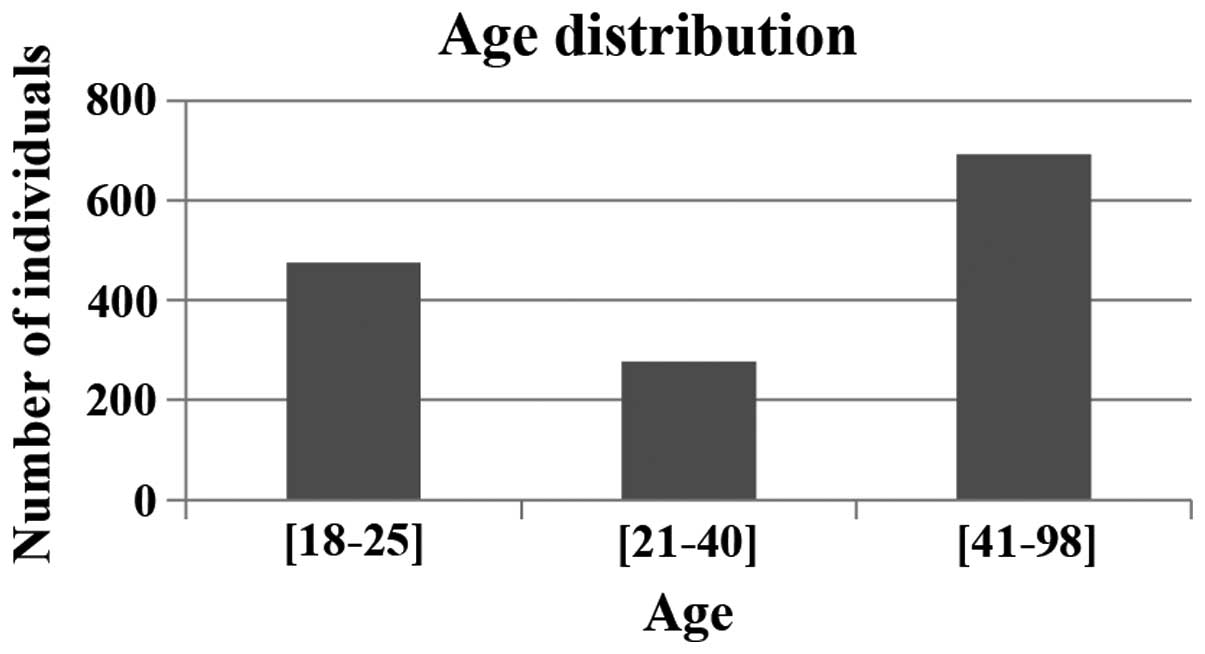
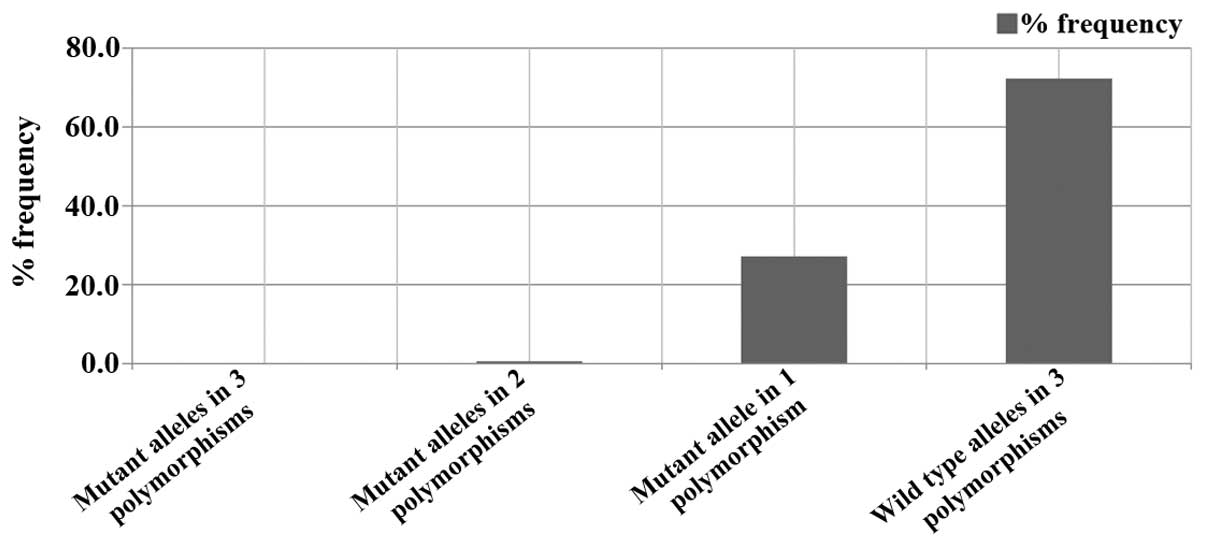
Spandidos Publications style
Katsarou MS, Latsi R, Papasavva M, Demertzis N, Kalogridis T, Tsatsakis AM, Spandidos DA and Drakoulis N: Population-based analysis of the frequency of HFE gene polymorphisms: Correlation with the susceptibility to develop hereditary hemochromatosis. Mol Med Rep 14: 630-636, 2016.
APA
Katsarou, M., Latsi, R., Papasavva, M., Demertzis, N., Kalogridis, T., Tsatsakis, A.M. ... Drakoulis, N. (2016). Population-based analysis of the frequency of HFE gene polymorphisms: Correlation with the susceptibility to develop hereditary hemochromatosis. Molecular Medicine Reports, 14, 630-636. https://doi.org/10.3892/mmr.2016.5317
MLA
Katsarou, M., Latsi, R., Papasavva, M., Demertzis, N., Kalogridis, T., Tsatsakis, A. M., Spandidos, D. A., Drakoulis, N."Population-based analysis of the frequency of HFE gene polymorphisms: Correlation with the susceptibility to develop hereditary hemochromatosis". Molecular Medicine Reports 14.1 (2016): 630-636.
Chicago
Katsarou, M., Latsi, R., Papasavva, M., Demertzis, N., Kalogridis, T., Tsatsakis, A. M., Spandidos, D. A., Drakoulis, N."Population-based analysis of the frequency of HFE gene polymorphisms: Correlation with the susceptibility to develop hereditary hemochromatosis". Molecular Medicine Reports 14, no. 1 (2016): 630-636. https://doi.org/10.3892/mmr.2016.5317