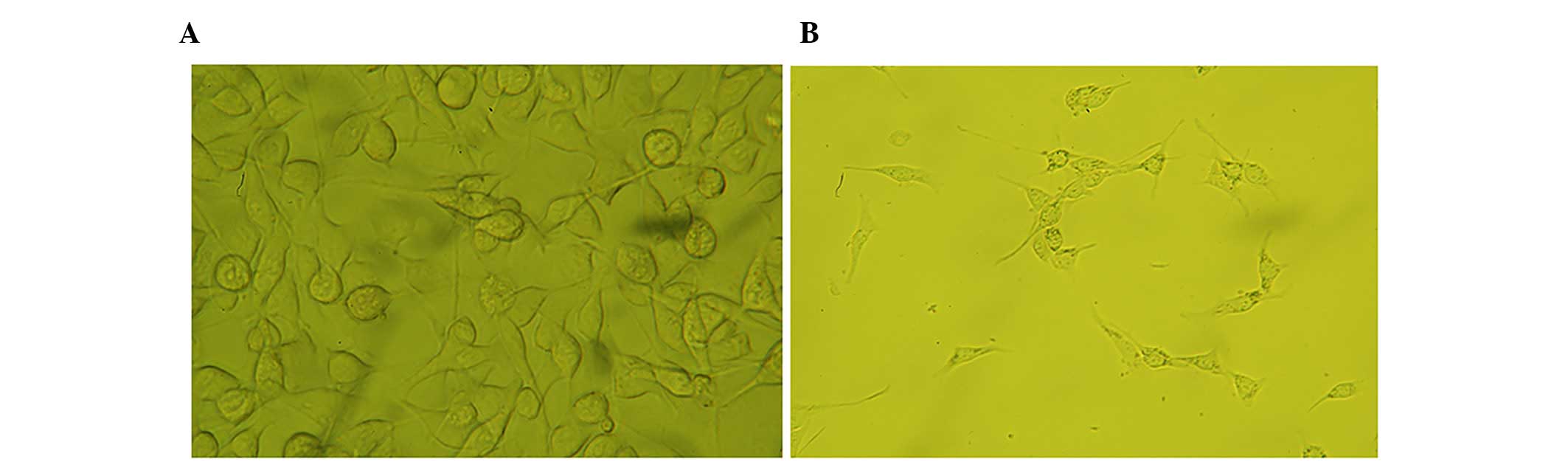
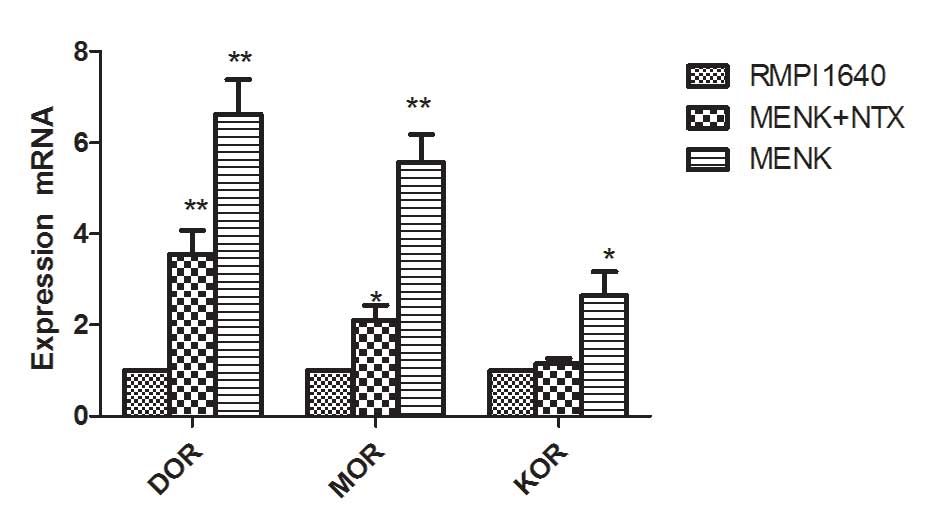
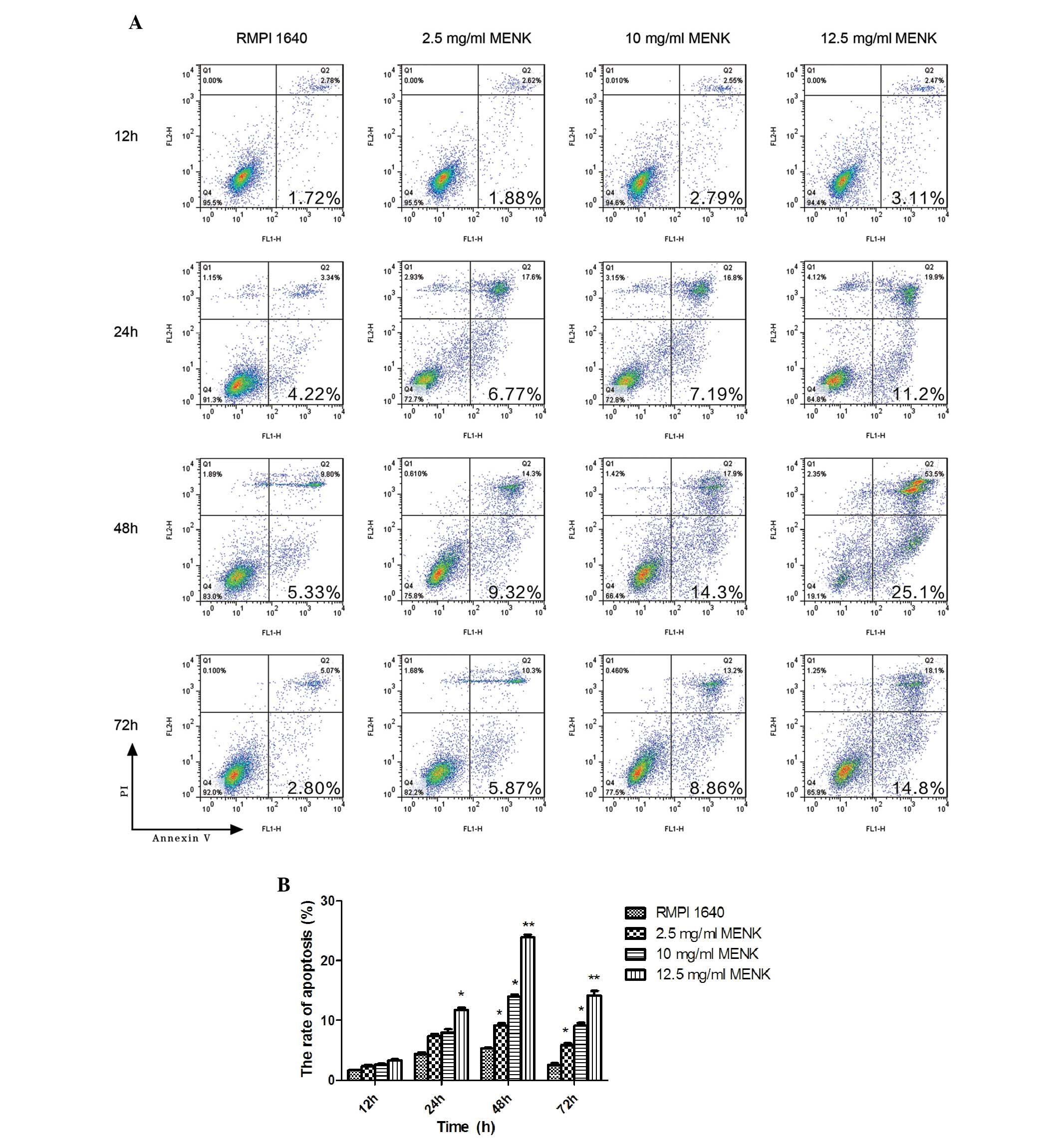
|
1
|
Miller AJ and Mihm MC Jr: Melanoma. N Engl
J Med. 355:51–65. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Linos E, Swetter SM, Cockburn MG, Colditz
GA and Clarke CA: Increasing burden of melanoma in the United
States. J Invest Dermatol. 129:1666–1674. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Howlader N, Noone AM, Krapcho M, Neyman N,
Aminou R, Waldron W, Altekruse SF, Kosary CL, Ruhl J, Tatalovich Z,
et al: SEER Cancer Statistics Review, 1975–2009 (Vintage 2009
Populations). http://seer.cancer.gov/csr/1975_2009_pops09/accessed
on October, 31 2013.
|
|
4
|
Bedikian AY, Millward M, Pehamberger H,
Conry R, Gore M, Trefzer U, Pavlick AC, DeConti R, Hersh EM, Hersey
P, et al: Bcl-2 antisense (oblimersen sodium) plus dacarbazine in
patients with advanced melanoma: The Oblimersen Melanoma Study
Group. J Clin Oncol. 24:4738–4345. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Engelman JA and Jänne PA: Mechanisms of
acquired resistance to epidermal growth factor receptor tyrosine
kinase inhibitors in non-small cell lung cancer. Clin Cancer Res.
14:2895–2899. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Pao W, Miller VA, Politi KA, Riely GJ,
Somwar R, Zakowski MF, Kris MG and Varmus H: Acquired resistance of
lung adenocarcinomas to gefitinib or erlotinib is associated with a
second mutation in the EGFR kinase domain. PLoS Med. 2:e732005.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Hunt KJ, Hung SK and Ernst E: Botanical
extracts as anti-aging preparations for the skin: A systematic
review. Drugs Aging. 27:973–985. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Antignac E, Nohynek GJ, Re T, Clouzeau J
and Toutain H: Safety of botanical ingredients in personal care
products/cosmetics. Food Chem Toxicol. 49:324–341. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Reuter J, Merfort I and Schempp CM:
Botanicals in dermatology: An evidence-based review. Am J Clin
Dermatol. 11:247–267. 2010.PubMed/NCBI
|
|
10
|
Baumann L, Woolery-Lloyd H and Friedman A:
‘Natural’ ingredients in cosmetic dermatology. J Drugs Dermatol.
8:(Suppl 6). s5–s9. 2009.PubMed/NCBI
|
|
11
|
Donahue RN, McLaughlin PJ and Zagon IS:
Cell proliferation of human ovarian cancer is regulated by the
opioid growth factor-opioid growth factor receptor axis. Am J
Physiol Regul Integr Comp Physiol. 296:R1716–R1725. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Vujic V, Stanojević S and Dimitrijević M:
Methionine-enkephalin stimulates hydrogen peroxide and nitric oxide
production in rat peritoneal macrophages: Interaction of mu, delta
and kappa opioid receptors. Neuroimmunomodulation. 11:392–403.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Avella DM, Kimchi ET, Donahue RN, Tagaram
HR, McLaughlin PJ, Zagon IS and Staveley-O'Carroll KF: The opioid
growth factor-opioid growth factor receptor axis regulates cell
proliferation of human hepatocellular cancer. Am J Physiol Regul
Integr Comp Physiol. 298:R459–R466. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zagon IS, Verderame MF and McLaughlin PJ:
The biology of the opioid growth factor receptor (OGFr). Brain Res
Brain Res Rev. 38:351–76. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zagon IS and McLaughlin PJ: Targeting
opioidergic pathways as a novel biological treatment for advanced
pancreatic cancer. Expert Rev Gastroenterol Hepatol. 6:133–135.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Gredicak M, Supek F, Kralj M, Majer Z,
Hollosi M, Smuc T, Mlinarić-Majerski K and Horvat S: Computational
structure-activity study directs synthesis of novel antitumor
enkephalin analogs. Amino Acids. 38:1185–1191. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zagon IS, Roesener CD, Verderame MF,
Ohlsson-Wilhelm BM, Levin RJ and McLaughlin PJ: Opioid growth
factor regulates the cell cycle of human neoplasias. Int J Oncol.
17:1053–1061. 2000.PubMed/NCBI
|
|
18
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) Method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
DeBenedette MA, Calderhead DM,
Ketteringham H, Gamble AH, Horvatinovich JM, Tcherepanova IY,
Nicolette CA and Healey DG: Priming of a novel subset of CD28+
rapidly expanding high-avidity effector memory CTL by post
maturation electroporation-CD40L dendritic cells is IL-12
dependent. J Immunol. 181:5296–5305. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Johnson AJ, Mendez-Fernandez Y, Moyer AM,
Sloma CR, Pirko I, Block MS, Rodriguez M and Pease LR:
Antigen-specific CD8+ T cells mediate a peptide-induced fatal
syndrome. J Immunol. 174:6854–6862. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Carr DJ and Carpenter GW: Morphine-induced
suppression of cytotoxic T lymphocyte activity in alloimmunized
mice is not mediated through a naltrindole-sensitive delta opioid
receptor. Neuroimmunomodulation. 2:44–53. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Carr DJ, Carpenter GW, Garza HH Jr, Baker
ML and Gebhardt BM: Cellular mechanisms involved in
morphine-mediated suppression of CTL activity. Adv Exp Med Biol.
373:131–139. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zagon IS, Verderame MF, Hankins J and
McLaughlin PJ: Overexpression of the opioid growth factor receptor
poten-tiates growth inhibition in human pancreatic cancer cells.
Int J Oncol. 30:775–83. 2007.PubMed/NCBI
|
|
24
|
Zagon IS, Donahue RN and McLaughlin PJ:
Opioid growth factor-opioid growth factor receptor axis is a
physiological determinant of cell proliferation in diverse human
cancers. Am J Physiol Regul Integr Comp Physiol. 297:1154–1161.
2009. View Article : Google Scholar
|
|
25
|
Zagon IS, Kreiner S, Heslop JJ, Conway AB,
Morgan CR and McLaughlin PJ: Prevention and delay in progression of
human pancreatic cancer by stable overexpression of the opioid
growth factor receptor. Int J Oncol. 33:317–323. 2008.PubMed/NCBI
|
|
26
|
Goldenberg D, Zagon IS, Fedok F, Crist HS
and McLaughlin PJ: Expression of opioid growth factor (OGF)-OGF
receptor (OGFr)axis in human nonmedullary thyroid cancer. Thyroid.
18:1165–1170. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
McLaughlin PJ, Kreiner S, Morgan CR and
Zagon IS: Prevention and delay in progression of human squamous
cell carcinoma of the head and neck in nude mice by stable
overexpression of the opioid growth factor receptor. Int J Oncol.
33:751–757. 2008.PubMed/NCBI
|
|
28
|
Smith JP, Bingaman SI, Mauger DT, Harvey
HH, Demers LM and Zagon IS: Opioid growth factor improves clinical
benefit and survival in patients with advanced pancreatic cancer.
Open Access J Clin Trials. 2010:37–48. 2010.PubMed/NCBI
|
|
29
|
Wu Y, McLaughlin PJ and Zagon IS: Ontogeny
of the opioid growth factor, [Met5]-enkephalin, preproenkephalin
gene expression, and the zeta opioid receptor in the developing and
adult aorta of rat. Dev Dyn. 211:327–337. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zagon IS, Ruth TB and McLaughlin PJ:
Nucheocytoplasmic distibution of opioid growth factor and its
receptor and its receptor in tongue epithelium. Anat Rec A Discov
Mol Cell Evol Biol. 282:24–37. 2005.PubMed/NCBI
|
|
31
|
McLaughlin PJ, Levin RJ and Zagon IS:
Regulation of human head and neck squamous cell carcinoma growth in
tissue culture by opioid growth factor. Int J Oncol. 14:991–998.
1999.PubMed/NCBI
|
|
32
|
Zagon IS, Porterfield NK and McLaughlin
PJ: Opioid growth factor-opioid growth factor receptor axis
inhibits proliferation of triple negative breast cancer. Exp Biol
Med (Maywood). 238:589–599. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Tsanova A, Jordanova A, Dzimbova T,
Pajpanova T, Golovinsky E and Lalchev Z: Interaction of
methionine-enkephalins with raft-forming lipids: Monolayers and BAM
experiments. Amino Acids. 46:1159–1168. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Liu T, Brouha B and Grossman D: Rapid
induction of mitochondrial events and caspase-independent apoptosis
in Survivin-targeted melanonma cells. Oncogene. 23:39–48. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Yang J, Wahdan-Alaswad R and Danielpour D:
Critical role of Smad2 in tumor suppression and transforming growth
factor-beta-induced apoptosis of prostate epithelial cells. Cancer
Res. 69:2185–2190. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Song K, Krebs TL and Danielpour D: Novel
permissive role of epidermal growth factor in transforming growth
factor beta (TGF-beta) signaling and growth suppression. Mediation
by stabilization of TGF-beta receptor type II. J Biol Chem.
281:7765–7774. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Shehata HH, Ghalia AH Abou, Elsayed EK,
Ziko OO and Mohamed SS: Detection of Survivin protein in aqueous
humor and serum of retinoblastoma patients and its clinical
significance. Clin Biochem. 43:362–366. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Tang L, Ling X, Liu W, Das GM and Li F:
Transcriptional inhibition of p21WAF1/CIP1 gene (CDKN1) expression
by survivin is at least partially p53-dependent: Evidence for
survivin acting as a transcription factor or co-factor. Biochem
Biophys Res Commun. 421:249–254. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Song K, Cornelius SC and Danielpour D:
Development and characterization of DP-153, a nontumorigenic
prostatic cell line that undergoes malignant transformation by
expression of dominant-negative transforming growth factor beta
receptor type II. Cancer Res. 63:4358–4367. 2003.PubMed/NCBI
|
|
40
|
Zagon IS and McLaughlin PJ: Opioid growth
factor and the treatment of human pancreatic cancer: A review.
World J Gastroenterol. 20:2218–2223. 2014. View Article : Google Scholar : PubMed/NCBI
|