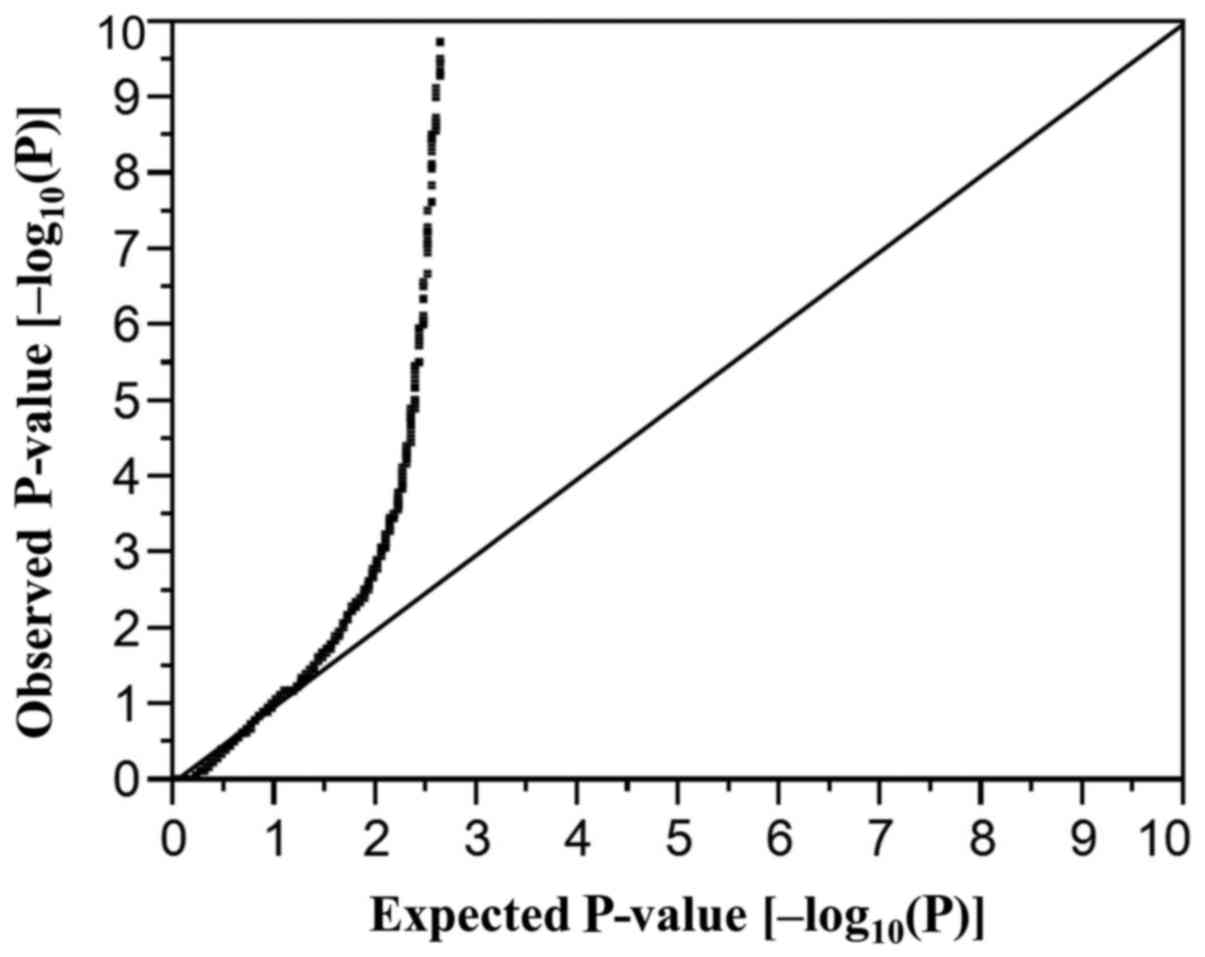
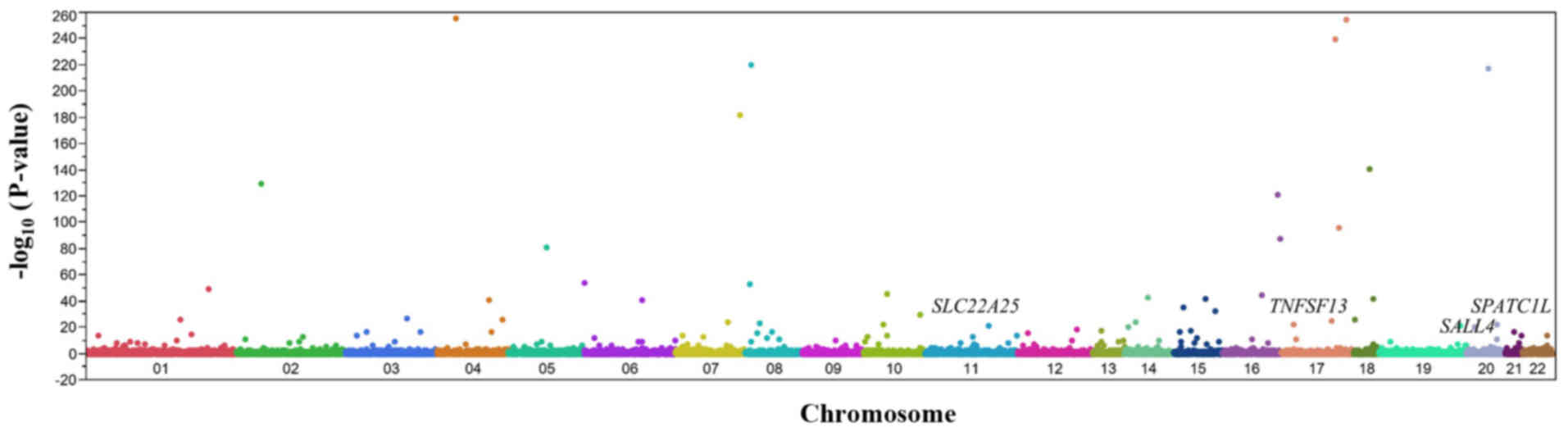
|
1
|
Chugh SS, Havmoeller R, Narayanan K, Singh
D, Rienstra M, Benjamin EJ, Gillum RF, Kim YH, McAnulty JH Jr,
Zheng ZJ, et al: Worldwide epidemiology of atrial fibrillation: A
global burden of disease 2010 study. Circulation. 129:837–847.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Go AS, Hylek EM, Phillips KA, Chang Y,
Henault LE, Selby JV and Singer DE: Prevalence of diagnosed atrial
fibrillation in adults: National implications for rhythm management
and stroke prevention: The An ticoagulation and risk factors in
atrial fibrillation (ATRIA) study. JAMA. 285:2370–2375. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Lip GY, Tse HF and Lane DA: Atrial
fibrillation. Lancet. 379:648–661. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Schnabel RB, Sullivan LM, Levy D, Pencina
MJ, Massaro JM, D'Agostion RB Sr, Newton-Cheh C, Yamamoto JF,
Magnani JW, Tadros TM, et al: Development of a risk score for
atrial fibrillation (Framingham Heart Study): A community-based
cohort study. Lancet. 373:739–745. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lubitz SA, Ozcan C, Magnani JW, Kääb S,
Benjamin EJ and Ellinor PT: Genetics of atrial fibrillation:
Implications for future research directions and personalized
medicine. Circ Arrhythm Electrophysiol. 3:291–299. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Gudbjartsson DF, Arnar DO, Helgadottir A,
Gretarsdottir S, Holm H, Sigurdsson A, Jonasdottir A, Baker A,
Thorleifsson G, Kristjansson K, et al: Variants conferring risk of
atrial fibrillation on chromosome 4q25. Nature. 448:353–357. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Gudbjartsson DF, Holm H, Gretarsdottir S,
Thorleifsson G, Walters GB, Thorgeirsson G, Gulcher J, Mathiesen
EB, Njølstad I, Nyrnes A, et al: A sequence variant in ZFHX3 on
16q22 associates with atrial fibrillation and ischemic stroke. Nat
Genet. 41:876–878. 2009. View
Article : Google Scholar : PubMed/NCBI
|
|
8
|
Benjamin EJ, Rice KM, Arking DE, Pfeufer
A, van Noord C, Smith AV, Schnabel RB, Bis JC, Boerwinkle E, Sinner
MF, et al: Variants in ZFHX3 are associated with atrial
fibrillation in individuals of European ancestry. Nat Genet.
41:879–881. 2009. View
Article : Google Scholar : PubMed/NCBI
|
|
9
|
Ellinor PT, Lunetta KL, Glazer NL, Pfeufer
A, Alonso A, Chung MK, Sinner MF, de Bakker PI, Mueller M, Lubitz
SA, et al: Common variants in KCNN3 are associated with lone atrial
fibrillation. Nat Genet. 42:240–244. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ellinor PT, Lunetta KL, Albert CM, Glazer
NL, Ritchie MD, Smith AV, Arking DE, Müller-Nurasyid M, Krijthe BP,
Lubitz SA, et al: Meta-analysis identifies six new susceptibility
loci for atrial fibrillation. Nat Genet. 44:670–675. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Manolio TA, Collins FS, Cox NJ, Goldstein
DB, Hindorff LA, Hunter DJ, McCarthy MI, Ramos EM, Cardon LR,
Chakravarti A, et al: Finding the missing heritability of complex
diseases. Nature. 461:747–753. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Grove ML, Yu B, Cochran BJ, Haritunians T,
Bis JC, Taylor KD, Hansen M, Borecki IB, Cupples LA, Fornage M, et
al: Best practices and joint calling of the HumanExome BeadChip:
The CHARGE consortium. PLoS One. 8:e680952013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Anderson CA, Pettersson FH, Clarke GM,
Cardon LR, Morris AP and Zondervan KT: Data quality control in
genetic case-control association studies. Nat Protoc. 5:1564–1573.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Price AL, Patterson NJ, Plenge RM,
Weinblatt ME, Shadick NA and Reich D: Principal components analysis
corrects for stratification in genome-wide association studies. Nat
Genet. 38:904–909. 2006. View
Article : Google Scholar : PubMed/NCBI
|
|
15
|
O'Neal WT, Efird JT, Yeboah J, Nazarian S,
Alonso A, Heckbert SR and Soliman EZ: Brachial flow-mediated
dilation and incident atrial fibrillation: The multi-ethnic study
of atherosclerosis. Arterioscler Thromb Vasc Biol. 34:2717–2720.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kim YM, Guzik TJ, Zhang YH, Zhang MH,
Kattach H, Ratnatunga C, Pillai R, Channon KM and Casadei B: A
myocardial Nox2 containing NAD (P)H oxidase contributes to
oxidative stress in human atrial fibrillation. Circ Res.
97:629–636. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Haïssaguerre M, Jaïs P, Shah DC, Takahashi
A, Hocini M, Quiniou G, Garrigue S, Le Mouroux A, Le Métayer P and
Clémenty J: Spontaneous initiation of atrial fibrillation by
ectopic beats origination in the pulmonary veins. N Engl J Med.
339:659–666. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Herringa J, Van Der Kuip DA, Hofman A,
Kors JA, van Rooij FJ, Lip GY and Witteman JC: Subclinical
atherosclerosis and risk of atrial fibrillation: The rotterdam
study. Arch Intern Med. 167:382–387. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Casaclang-Verzosa G, Gresh BJ and Tsang
TS: Structural and functional remodeling of the left atrium:
Clinical and therapeutic implications for atrial fibrillation. J Am
Coll Cardiol. 51:1–11. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Dillon SR, Gross JA, Ansell SM and Novak
AJ: An APRIL to remember: Novel TNF ligands as therapeutic targets.
Nat Rev Drug Discov. 5:235–246. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Thangarajh M, Masterman T, Rot U, Duvefelt
K, Brynedal B, Karrenbauer VD and Hillert J: Increased levels of
APRIL (a proliferation-inducing ligand) mRNA in multiple sclerosis.
J Neuroimmunol. 167:210–214. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Jonsson MV, Szodoray P, Jellestad S,
Jonsson R and Skarstein K: Association between circulating levels
of the novel TNF family members APRIL and BAFF and lymphoid
organization in primary Sjögren's syndrome. J Clin Immunol.
25:189–201. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Koyama T, Tsukamoto H, Miyagi Y, Himeji D,
Otsuka J, Miyagawa H, Harada M and Horiuchi T: Raised serum APRIL
levels in patients with systemic lupus erythematosus. Ann Rheum
Dis. 64:1065–1067. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Cheema GS, Roschke V, Hilbert DM and Stohl
W: Elevated serum B lymphocyte stimulator levels in patients with
systemic immune-based rheumatic diseases. Arthritis Rheum.
44:1313–1319. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Tan SM, Xu D, Roschke V, Perry JW, Arkfeld
DG, Ehresmann GR, Migone TS, Hilbert DM and Stohl W: Local
production of B lymphocyte stimulator protein and APRIL in
arthritic joints of patients with inflammatory arthritis. Arthritis
Rheum. 48:982–992. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kiryluk K, Li Y, Scolari F, Sanna-Cherchi
S, Choi M, Verbitsky M, Fasel D, Lata S, Prakash S, Shapiro S, et
al: Discovery of new risk loci for IgA nephropathy implicates genes
involved in immunity against intestinal pathogens. Nat Genet.
46:1187–1196. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Libby P: Inflammation in atherosclerosis.
Nature. 420:868–874. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Sandberg WJ, Otterdal K, Gullestad L,
Halvorsen B, Ragnarsson A, Frøland SS, Damås JK, Oie E, Aukrust P,
Hansson GK and Yndestad A: The tumour necrosis factor superfamily
ligand APRIL (TNFSF13) is released upon platelet activation and
expressed in atherosclerosis. Thromb Haemost. 102:704–710.
2009.PubMed/NCBI
|
|
29
|
Lecat S, Matthes HW, Pepperkok R, Simpson
JC and Galzi JL: A fluorescent live imaging screening assay based
on translocation criteria identifies novel cytoplasmic proteins
implicated in G protein-coupled receptor signaling pathways. Mol
Cell Proteomics. 14:1385–1399. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Fry RC, Svensson JP, Valiathan C, Wang E,
Hogan BJ, Bhattacharya S, Bugni JM, Whittaker CA and Samson LD:
Genomic predictors of interindividual differences in response to
DNA damaging agents. Genes Dev. 22:2621–2626. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Jacobsson JA, Haitina T, Lindblom J and
Fredriksson R: Identification of six putative human transporters
with structural similarity to the drug transporter SLC22 family.
Genomics. 90:595–609. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kohlhase J, Heinrich M, Schubert L,
Liebers M, Kispert A, Laccone F, Turnpenny P, Winter RM and Reardon
W: Okihiro syndrome is caused by SALL4 mutations. Hum Mol Genet.
11:2979–2987. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Sakaki-Yumoto M, Kobayashi C, Sato A,
Fujimura S, Matsumoto Y, Takasato M, Kodama T, Aburatani H,
Asashima M, Yoshida N and Nishinakamura R: The murine homolog of
SALL4, a causative gene in Okihiro syndrome, is essential for
embryonic stem cell proliferation, and cooperates with Sall1 in
anorectal, heart, brain and kidney development. Development.
133:3005–3013. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Koshiba-Takeuchi K, Takeuchi JK, Arruda
EP, Kathiriya IS, Mo R, Hui CC, Srivastava D and Bruneau BG:
Cooperative and antagonistic interactions between Sall4 and Tbx5
pattern the mouse limb and heart. Nat Genet. 38:175–183. 2006.
View Article : Google Scholar : PubMed/NCBI
|