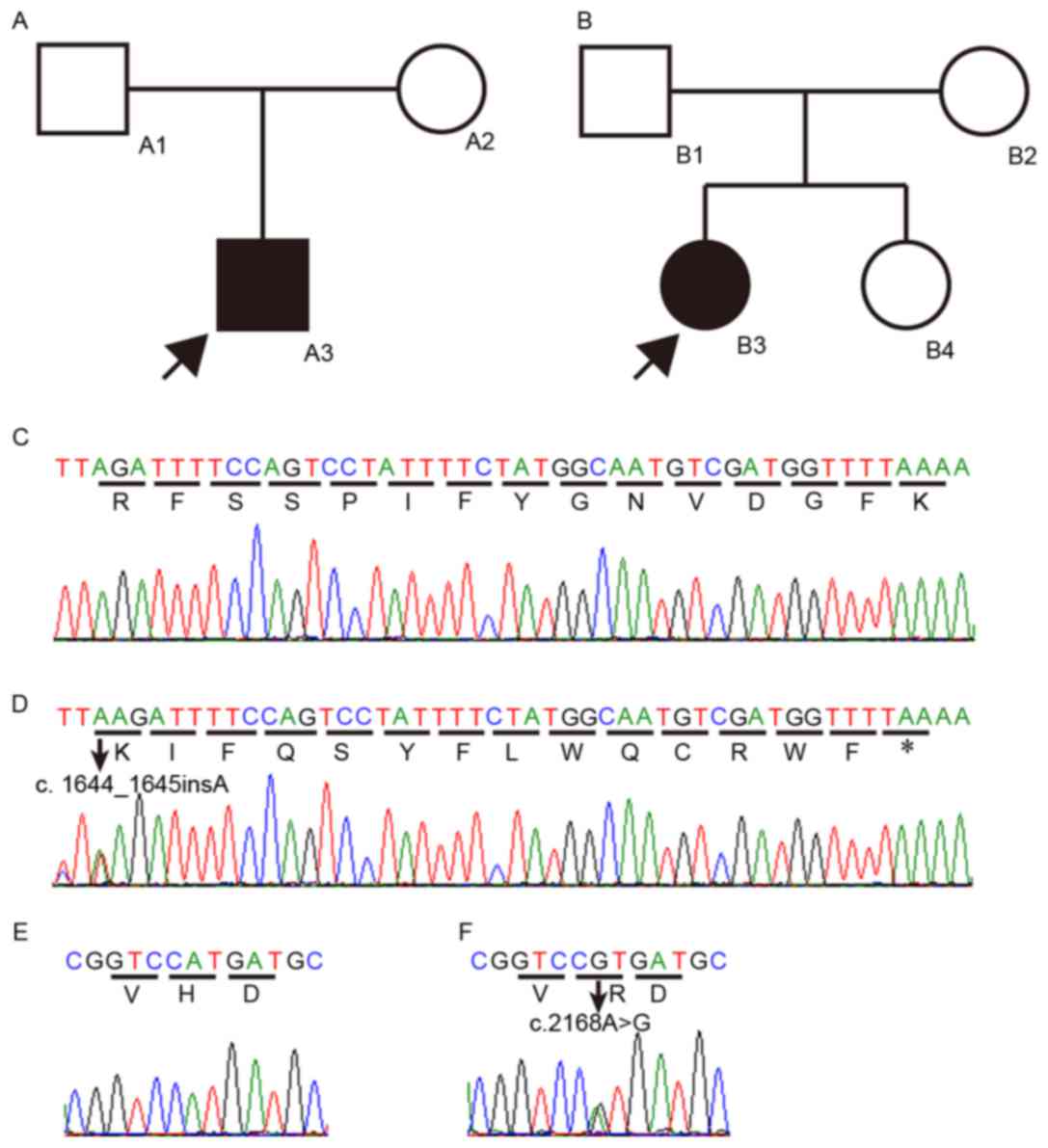
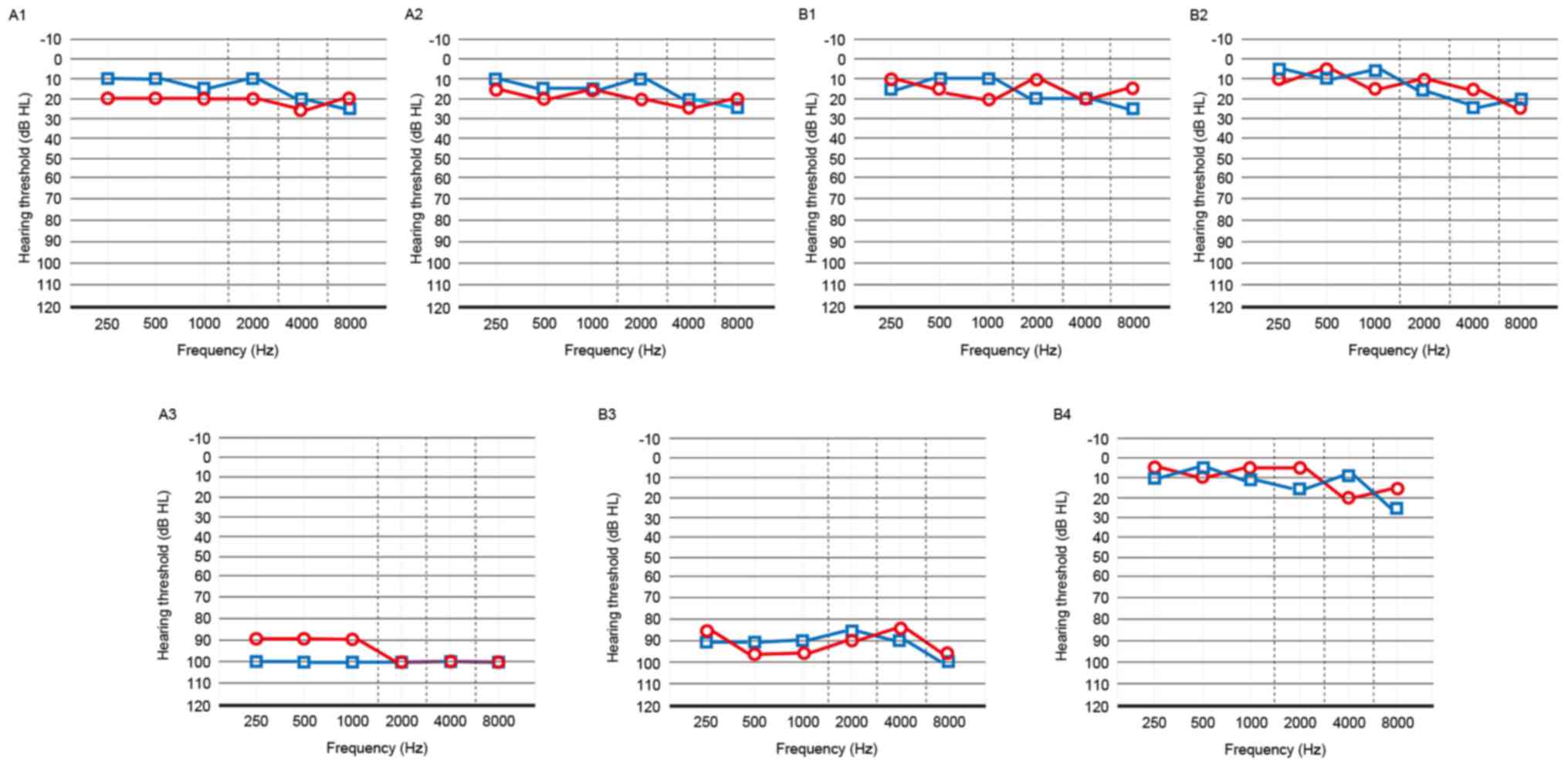
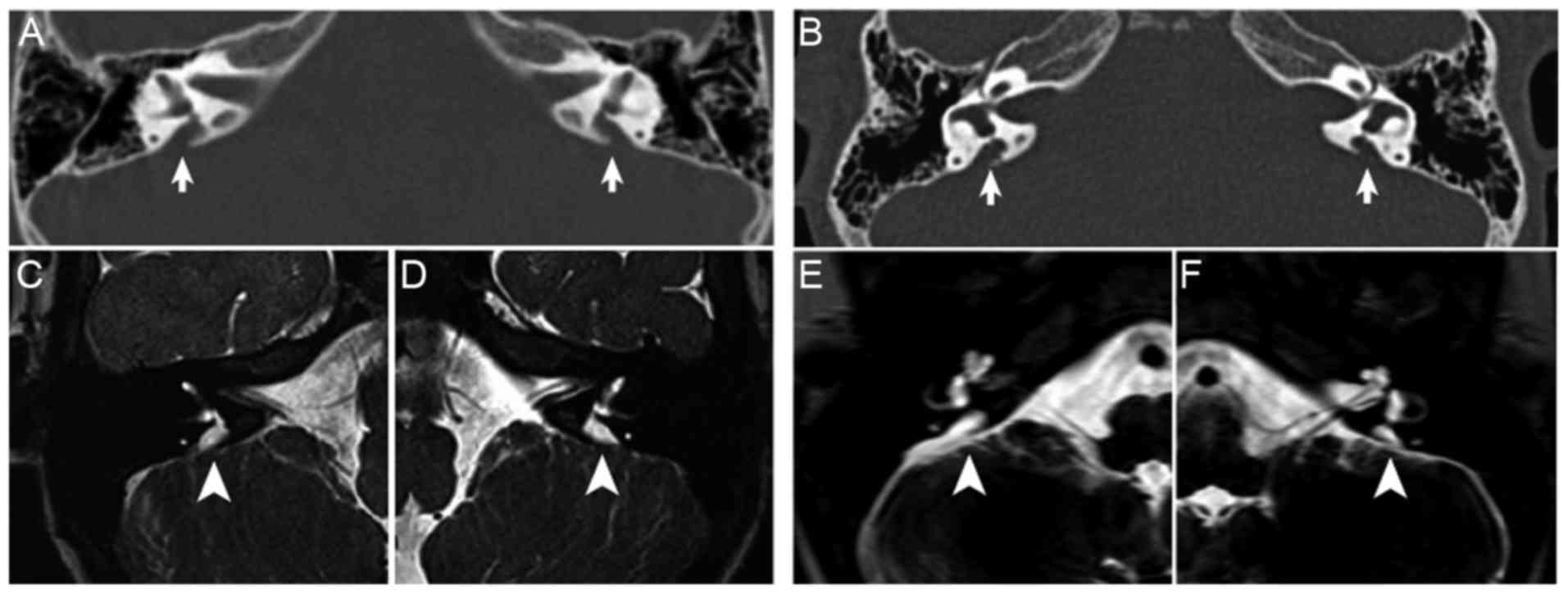
|
1
|
Usami S, Abe S, Weston MD, Shinkawa H, Van
Camp G and Kimberling WJ: Non-syndromic hearing loss associated
with enlarged vestibular aqueduct is caused by PDS mutations. Hum
Genet. 104:188–192. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Pique LM, Brennan ML, Davidson CJ,
Schaefer F, Greinwald J Jr and Schrijver I: Mutation analysis of
the SLC26A4, FOXI1 and KCNJ10 genes in individuals with congenital
hearing loss. Peer J. 2:e3842014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Reardon W, OMahoney CF, Trembath R, Jan H
and Phelps PD: Enlarged vestibular aqueduct: A radiological marker
of pendred syndrome, and mutation of the PDS gene. QJM. 93:99–104.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Campbell C, Cucci RA, Prasad S, Green GE,
Edeal JB, Galer CE, Karniski LP, Sheffield VC and Smith RJ: Pendred
syndrome, DFNB4, and PDS/SLC26A4 identification of eight novel
mutations and possible genotype-phenotype correlations. Hum Mutat.
17:403–411. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Pryor SP, Madeo AC, Reynolds JC, Sarlis
NJ, Arnos KS, Nance WE, Yang Y, Zalewski CK, Brewer CC, Butman JA
and Griffith AJ: SLC26A4/PDS genotype-phenotype correlation in
hearing loss with enlargement of the vestibular aqueduct (EVA):
Evidence that Pendred syndrome and non-syndromic EVA are distinct
clinical and genetic entities. J Med Genet. 42:159–165. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Everett LA, Glaser B, Beck JC, Idol JR,
Buchs A, Heyman M, Adawi F, Hazani E, Nassir E, Baxevanis AD, et
al: Pendred syndrome is caused by mutations in a putative sulphate
transporter gene (PDS). Nat Genet. 17:411–422. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kopp P, Pesce L and Solis-S JC: Pendred
syndrome and iodide transport in the thyroid. Trends Endocrinol
Metab. 19:260–268. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Chai Y, Huang Z, Tao Z, Li X, Li L, Li Y,
Wu H and Yang T: Molecular etiology of hearing impairment
associated with nonsyndromic enlarged vestibular aqueduct in East
China. Am J Med Genet A. 161A:1–2233. 2013.PubMed/NCBI
|
|
9
|
Van Nierop JW, Huinck WJ, Pennings RJ,
Admiraal RJ, Mylanus EA and Kunst HP: Patients with pendred
syndrome: Is cochlear implantation beneficial? Clin Otolaryngol.
41:386–394. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Cremers CW, Admiraal RJ, Huygen PL, Bolder
C, Everett LA, Joosten FB, Green ED, van Camp G and Otten BJ:
Progressive hearing loss, hypoplasia of the cochlea and widened
vestibular aqueducts are very common features in Pendred's
syndrome. Int J Pediatr Otorhinolaryngol. 45:113–123. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Coyle B, Reardon W, Herbrick JA, Tsui LC,
Gausden E, Lee J, Coffey R, Grueters A, Grossman4 A, Phelps PD, et
al: Molecular analysis of the PDS gene in Pendred syndrome. Hum Mol
Genet. 7:1105–1112. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Reyes S, Wang G, Ouyang X, Han B, Du LL,
Yuan HJ, Yan D, Dai P and Liu XZ: Mutation analysis of SLC26A4 in
mainland Chinese patients with enlarged vestibular aqueduct.
Otolaryngol Head Neck Surg. 141:502–508. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Peng Q, Huang S, Liang Y, Ma K, Li S, Yang
L, Li W, Ma Q, Liu Q, Zhong B and Lu X: Concurrent genetic and
standard screening for hearing impairment in 9317 southern chinese
newborns. Genet Test Mol Biomarkers. 20:603–608. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Tsukamoto K, Suzuki H, Harada D, Namba A,
Abe S and Usami S: Distribution and frequencies of PDS (SLC26A4)
mutations in Pendred syndrome and nonsyndromic hearing loss
associated with enlarged vestibular aqueduct: A unique spectrum of
mutations in Japanese. Eur J Hum Genet. 11:916–922. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Shin JW, Lee SC, Lee HK and Park HJ:
Genetic screening of GJB2 and SLC26A4 in korean cochlear
implantees: Experience of soree ear clinic. Clin Exp
Otorhinolaryngol. 5 Suppl 1:S10–S13. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wang QJ, Zhao YL, Rao SQ, Guo YF, Yuan H,
Zong L, Guan J, Xu BC, Wang DY, Han MK, et al: A distinct spectrum
of SLC26A4 mutations in patients with enlarged vestibular aqueduct
in China. Clin Genet. 72:245–254. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wu CC, Yeh TH, Chen PJ and Hsu CJ:
Prevalent SLC26A4 mutations in patients with enlarged vestibular
aqueduct and/or Mondini dysplasia: A unique spectrum of mutations
in Taiwan, including a frequent founder mutation. Laryngoscope.
115:1060–1064. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yoon JS, Park HJ, Yoo SY, Namkung W, Jo
MJ, Koo SK, Park HY, Lee WS, Kim KH and Lee MG: Heterogeneity in
the processing defect of SLC26A4 mutants. J Med Genet. 45:411–419.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Pang X, Chai Y, Chen P, He L, Wang X, Wu H
and Yang T: Mono-allelic mutations of SLC26A4 is over-presented in
deaf patients with non-syndromic enlarged vestibular aqueduct. Int
J Pediatr Otorhinolaryngol. 79:1351–1353. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Sagong B, Baek JI, Lee KY and Kim UK: A
novel frameshift mutation of SLC26A4 in a korean family with
nonsyndromic hearing loss and enlarged vestibular aqueduct. Clin
Exp Otorhinolaryngol. 10:50–55. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Yao J, Qian X, Bao J, Wei Q, Lu Y, Zheng
H, Cao X and Xing G: Probing the effect of two heterozygous
mutations in codon 723 of SLC26A4 on deafness phenotype based on
molecular dynamics simulations. Sci Rep. 5:108312015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Aravind L and Koonin EV: The STAS domain-a
link between anion transporters and antisigma-factor antagonists.
Curr Biol. 10:R53–R55. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Sharma AK, Ye L, Baer CE, Shanmugasundaram
K, Alber T, Alper SL and Rigby AC: Solution structure of the
guanine nucleotide-binding STAS domain of SLC26-related SulP
protein Rv1739c from Mycobacterium tuberculosis. J Biol Chem.
286:8534–8544. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ko SB, Zeng W, Dorwart MR, Luo X, Kim KH,
Millen L, Goto H, Naruse S, Soyombo A, Thomas PJ and Muallem S:
Gating of CFTR by the STAS domain of SLC26 transporters. Nat Cell
Biol. 6:343–350. 2004. View
Article : Google Scholar : PubMed/NCBI
|
|
25
|
Taylor JP, Metcalfe RA, Watson PF, Weetman
AP and Trembath RC: Mutations of the PDS gene, encoding pendrin,
are associated with protein mislocalization and loss of iodide
efflux: Implications for thyroid dysfunction in Pendred syndrome. J
Clin Endocrinol Metab. 87:1778–1784. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Pera A, Dossena S, Rodighiero S, Gandía M,
Bottà G, Meyer G, Moreno F, Nofziger C, Hernández-Chico C and
Paulmichl M: Functional assessment of allelic variants in the
SLC26A4 gene involved in Pendred syndrome and nonsyndromic EVA.
Proc Natl Acad Sci USA. 105:pp. 18608–18613. 2008; View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Scott DA, Wang R, Kreman TM, Andrews M,
McDonald JM, Bishop JR, Smith RJ, Karniski LP and Sheffield VC:
Functional differences of the PDS gene product are associated with
phenotypic variation in patients with Pendred syndrome and
non-syndromic hearing loss (DFNB4). Hum Mol Genet. 9:1709–1715.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Bizhanova A, Chew TL, Khuon S and Kopp P:
Analysis of cellular localization and function of carboxy-terminal
mutants of pendrin. Cell Physiol Biochem. 28:423–434. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Frischmeyer PA and Dietz HC:
Nonsense-mediated mRNA decay in health and disease. Hum Mol Genet.
8:1893–1900. 1999. View Article : Google Scholar : PubMed/NCBI
|