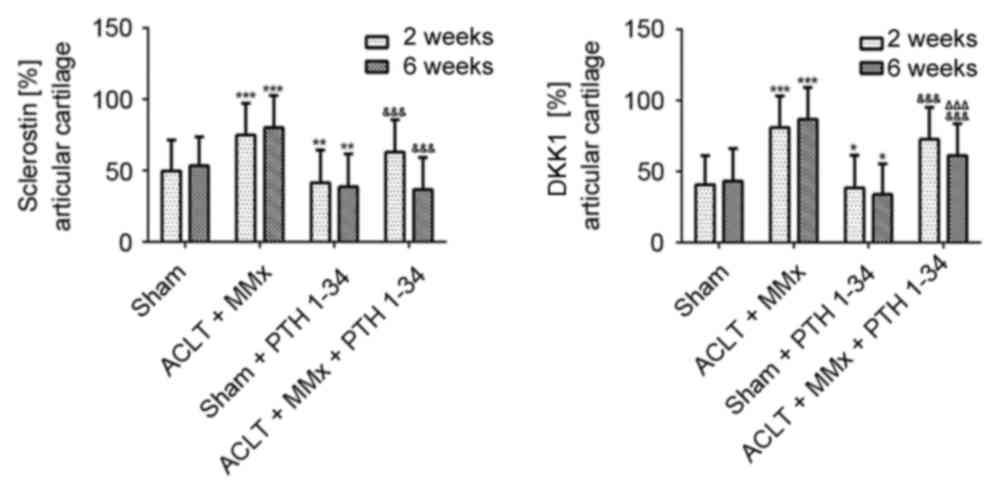
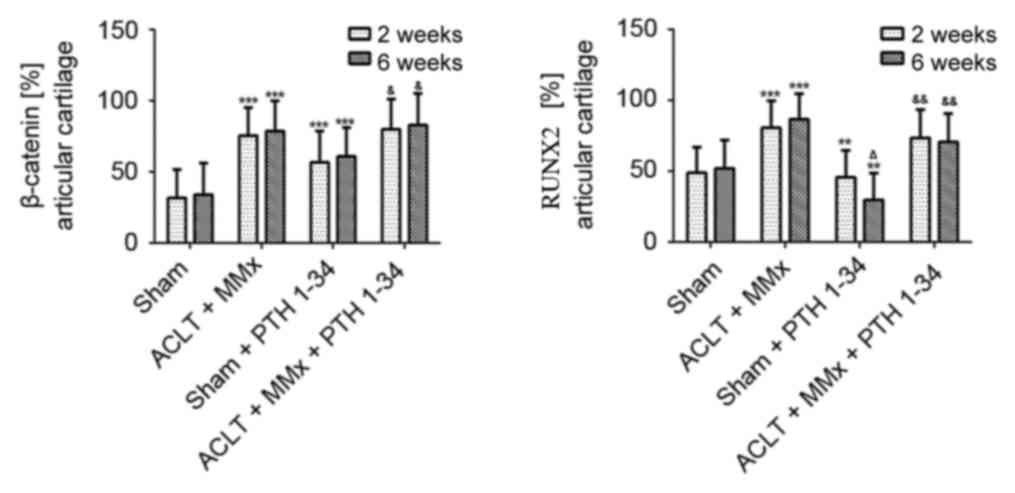
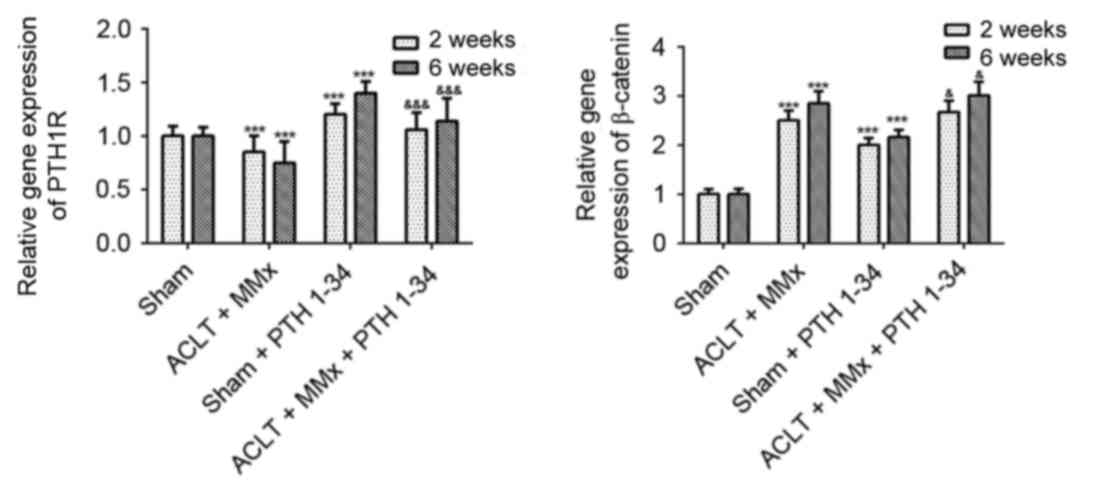
|
1
|
Allen KD and Golightly YM: State of the
evidence. Curr Opin Rheumatol. 27:276–283. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Pulsatelli L, Addimanda O, Brusi V,
Pavloska B and Meliconi R: New findings in osteoarthritis
pathogenesis: Therapeutic implications. Ther Adv Chronic Dis.
4:23–43. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Zhong L, Huang X, Karperien M and Post JN:
The regulatory role of signaling crosstalk in hypertrophy of MSCs
and human articular chondrocytes. Int J Mol Sci. 16:19225–19247.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Yuasa T, Otani T, Koike T, Iwamoto M and
Enomoto-Iwamoto M: Wnt/beta-catenin signaling stimulates matrix
catabolic genes and activity in articular chondrocytes: Its
possible role in joint degeneration. Lab Invest. 88:264–274. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Harrington EK, Roddy GW, West R and
Svoboda KK: Parathyroid hormone/parathyroid hormone-related peptide
modulates growth of avian sternal cartilage via chondrocytic
proliferation. Anat Rec (Hoboken). 290:155–167. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Neer RM, Arnaud CD, Zanchetta JR, Prince
R, Gaich GA, Reginster JY, Hodsman AB, Eriksen EF, Ish-Shalom S,
Genant HK, et al: Effect of parathyroid hormone (1–34) on fractures
and bone mineral density in postmenopausal women with osteoporosis.
N Engl J Med. 344:1434–1441. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Abou-Samra AB, Jüppner H, Force T, Freeman
MW, Kong XF, Schipani E, Urena P, Richards J, Bonventre JV, Potts
JT Jr, et al: Expression cloning of a common receptor for
parathyroid hormone and parathyroid hormone-related peptide from
rat osteoblast-like cells: A single receptor stimulates
intracellular accumulation of both cAMP and inositol trisphosphates
and increases intracellular free calcium. Proc Natl Acad Sci USA.
89:pp. 2732–2736. 1992; View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Yan JY, Tian FM, Wang WY, Cheng Y, Song
HP, Zhang YZ and Zhang L: Parathyroid hormone (1–34) prevents
cartilage degradation and preserves subchondral bone
micro-architecture in guinea pigs with spontaneous osteoarthritis.
Osteoarthritis Cartilage. 22:1869–1877. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Sampson ER, Hilton MJ, Tian Y, Chen D,
Schwarz EM, Mooney RA, Bukata SV, O'Keefe RJ, Awad H, Puzas JE, et
al: Teriparatide as a chondroregenerative therapy for
injury-induced osteoarthritis. Sci Transl Med. 3:101ra932011.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chang JK, Chang LH, Hung SH, Wu SC, Lee
HY, Lin YS, Chen CH, Fu YC, Wang GJ and Ho ML: Parathyroid hormone
1–34 inhibits terminal differentiation of human articular
chondrocytes and osteoarthritis progression in rats. Arthritis
Rheum. 60:3049–3060. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Regard JB, Zhong Z, Williams BO and Yang
Y: Wnt signaling in bone development and disease: Making stronger
bone with Wnts. Cold Spring Harb Perspect Biol. 4:pii: a0079972012.
View Article : Google Scholar
|
|
12
|
Yasuhara R, Ohta Y, Yuasa T, Kondo N,
Hoang T, Addya S, Fortina P, Pacifici M, Iwamoto M and
Enomoto-Iwamoto M: Roles of β-catenin signaling in phenotypic
expression and proliferation of articular cartilage superficial
zone cells. Lab Invest. 91:1739–1752. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Loeser RF: Osteoarthritis Year in Review
2013: Biology. Osteoarthritis Cartilage. 21:1436–1442. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Dong YF, do Y Soung, Schwarz EM, O'Keefe
RJ and Drissi H: Wnt induction of chondrocyte hypertrophy through
the Runx2 transcription factor. J Cell Physiol. 208:77–86. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Pickarski M, Hayami T, Zhuo Y and Duong
LT: Molecular changes in articular cartilage and subchondral bone
in the rat anterior cruciate ligament transection and
meniscectomized models of osteoarthritis. BMC Musculoskelet Disord.
12:1972011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Rossi R, Dettoni F, Bruzzone M, Cottino U,
D'Elicio DG and Bonasia DE: Clinical examination of the knee: Know
your tools for diagnosis of knee injuries. Sports Med Arthrosc
Rehabil Ther Technol. 3:252011. View Article : Google Scholar : PubMed/NCBI
|
|
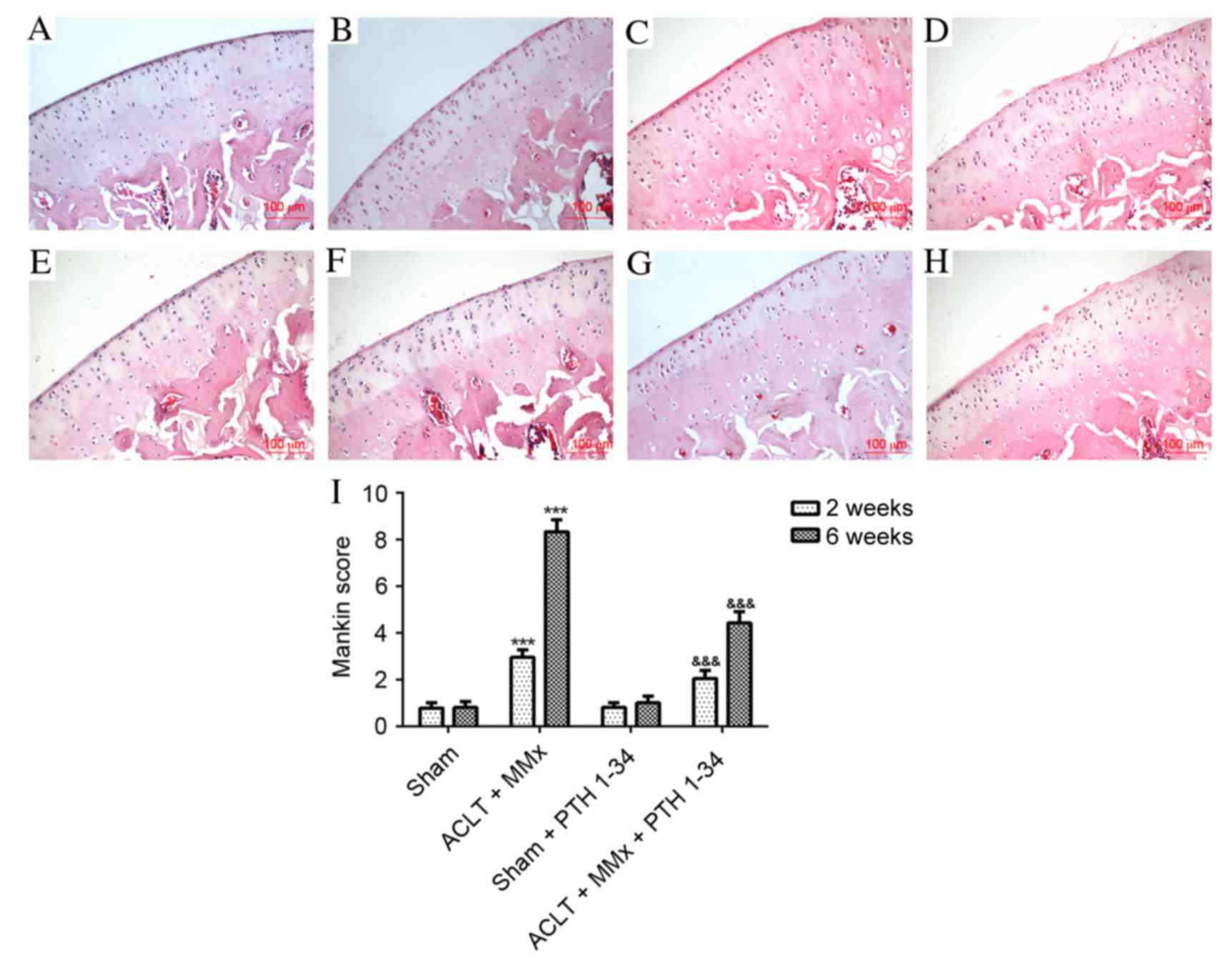
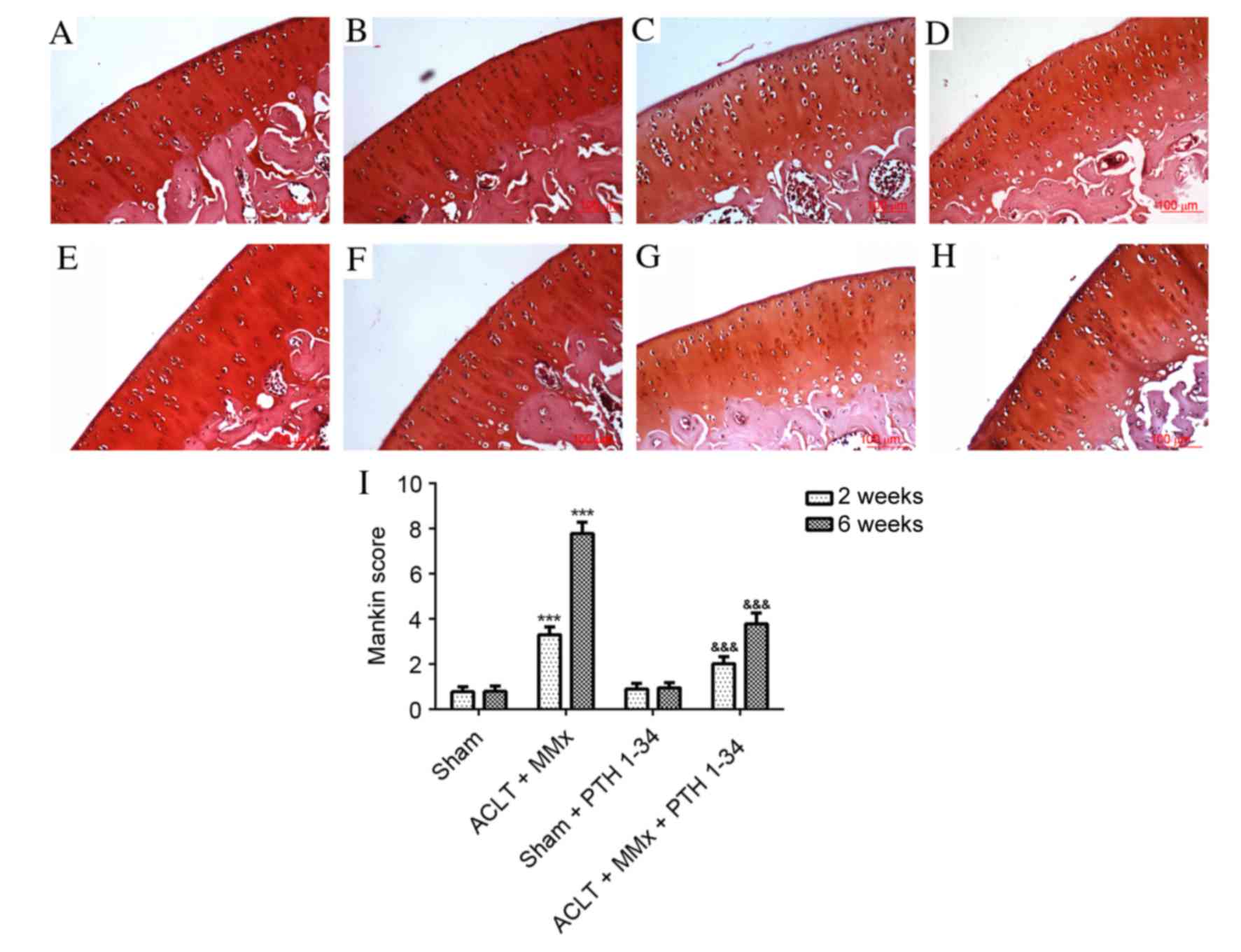
17
|
Mankin HJ, Dorfman H, Lippiello L and
Zarins A: Biochemical and metabolic abnormalities in articular
cartilage from osteo-arthritic human hips. II. Correlation of
morphology with biochemical and metabolic data. J Bone Joint Surg
Am. 53:523–537. 1971. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Longo UG, Loppini M, Fumo C, Rizzello G,
Khan WS, Maffulli N and Denaro V: Osteoarthritis: New insights in
animal models. Open Orthop J. 6:558–563. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Orth P, Cucchiarini M, Zurakowski D,
Menger MD, Kohn DM and Madry H: Parathyroid hormone [1-34] improves
articular cartilage surface architecture and integration and
subchondral bone reconstitution in osteochondral defects in vivo.
Osteoarthritis Cartilage. 21:614–624. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
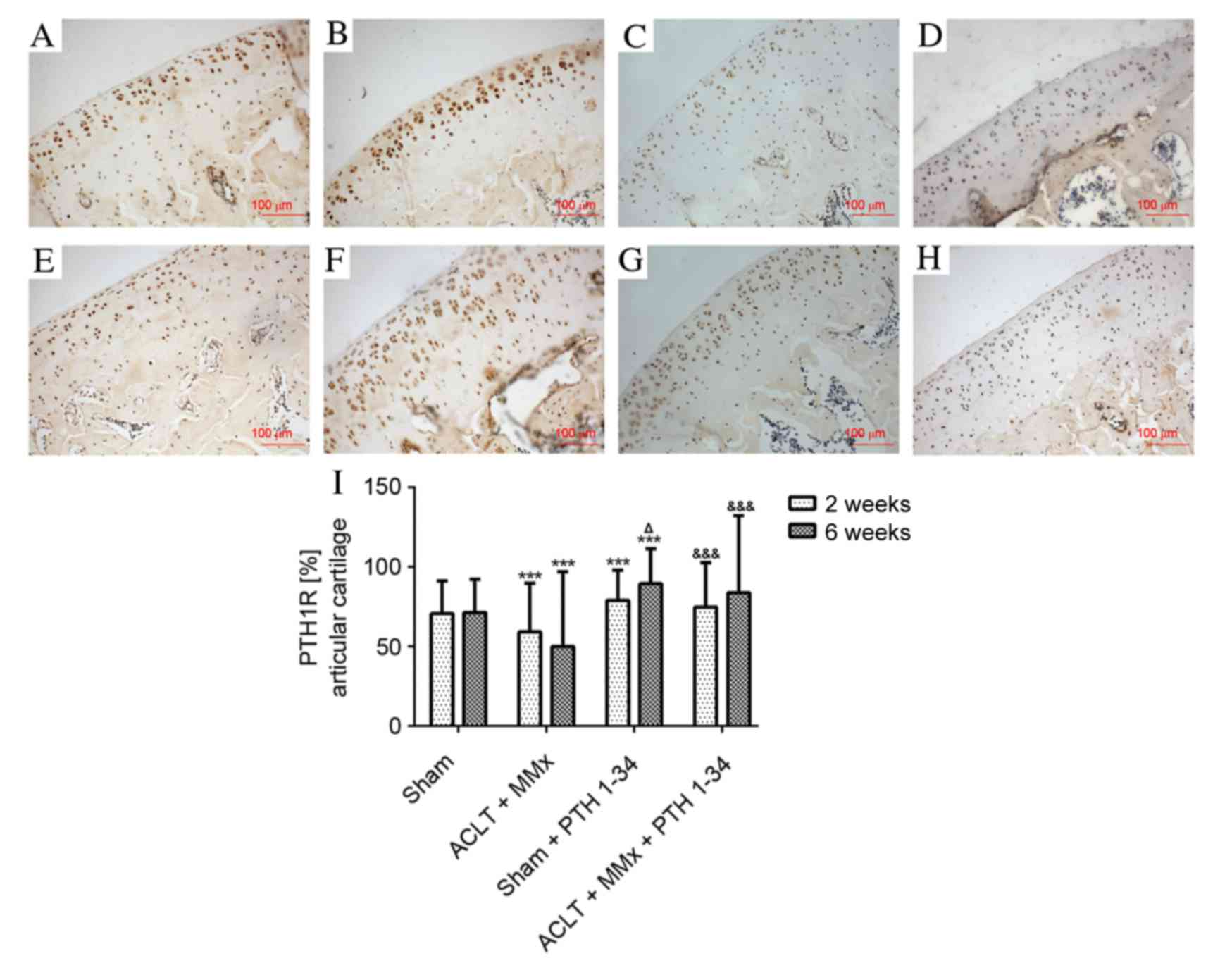
Roudier M, Li X, Niu QT, Pacheco E,
Pretorius JK, Graham K, Yoon BR, Gong J, Warmington K, Ke HZ, et
al: Sclerostin is expressed in articular cartilage but loss or
inhibition does not affect cartilage remodeling during aging or
following mechanical injury. Arthritis Rheum. 65:721–731. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
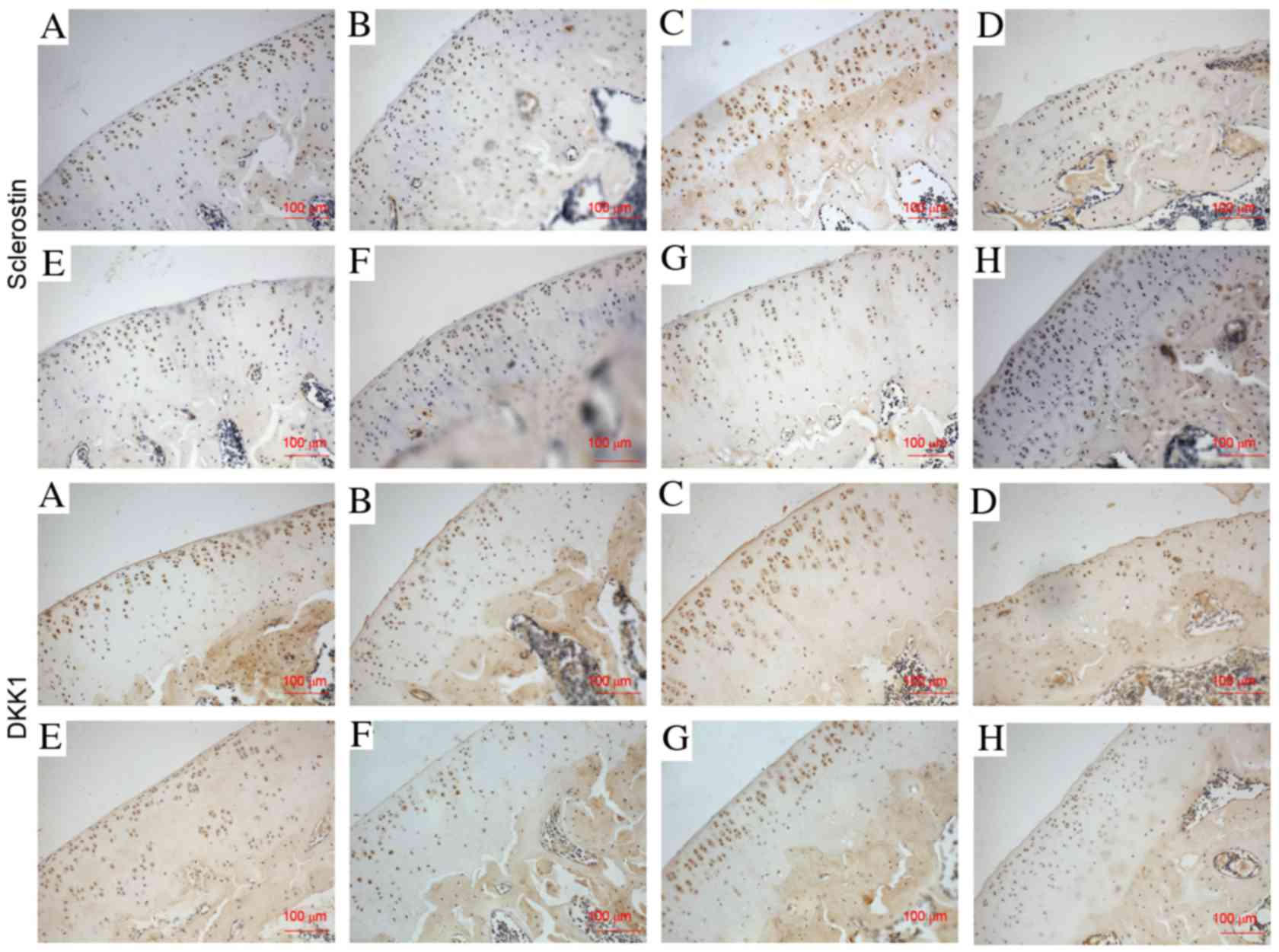
van Bezooijen RL, Roelen BA, Visser A, Van
der Wee-Pals L, de Wilt E, Karperien M, Hamersma H, Papapoulos SE,
ten Dijke P and Löwik CW: Sclerostin is an osteocyte-expressed
negative regulator of bone formation, but not a classical BMP
antagonist. J Exp Med. 199:805–814. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Li X, Zhang Y, Kang H, Liu W, Liu P, Zhang
J, Harris SE and Wu D: Sclerostin binds to LRP5/6 and antagonizes
canonical Wnt signaling. J Biol Chem. 280:19883–19887. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Li X, Ominsky MS, Warmington KS, Morony S,
Gong J, Cao J, Gao Y, Shalhoub V, Tipton B, Haldankar R, et al:
Sclerostin antibody treatment increases bone formation, bone mass,
and bone strength in a rat model of postmenopausal osteoporosis. J
Bone Miner Res. 24:578–588. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Bouaziz W, Funck-Brentano T, Lin H, Marty
C, Ea HK, Hay E and Cohen-Solal M: Loss of sclerostin promotes
osteoarthritis in mice via beta-catenin-dependent and -independent
Wnt pathways. Arthritis Res Ther. 17:242015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Chan BY, Fuller ES, Russell AK, Smith SM,
Smith MM, Jackson MT, Cake MA, Read RA, Bateman JF, Sambrook PN and
Little CB: Increased chondrocyte sclerostin may protect against
cartilage degradation in osteoarthritis. Osteoarthritis Cartilage.
19:874–885. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Mao B, Wu W, Davidson G, Marhold J, Li M,
Mechler BM, Delius H, Hoppe D, Stannek P, Walter C, et al: Kremen
proteins are Dickkopf receptors that regulate Wnt/beta-catenin
signalling. Nature. 417:664–667. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Niehrs C: Function and biological roles of
the Dickkopf family of Wnt modulators. Oncogene. 25:7469–7481.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Honsawek S, Tanavalee A, Yuktanandana P,
Ngarmukos S, Saetan N and Tantavisut S: Dickkopf-1 (Dkk-1) in
plasma and synovial fluid is inversely correlated with radiographic
severity of knee osteoarthritis patients. BMC Musculoskelet Disord.
11:2572010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Oh H, Chun CH and Chun JS: Dkk-1
expression in chondrocytes inhibits experimental osteoarthritic
cartilage destruction in mice. Arthritis Rheum. 64:2568–2578. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Weng LH, Wang CJ, Ko JY, Sun YC and Wang
FS: Control of Dkk-1 ameliorates chondrocyte apoptosis, cartilage
destruction, and subchondral bone deterioration in osteoarthritic
knees. Arthritis Rheum. 62:1393–1402. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Guo J, Liu M, Yang D, Bouxsein ML, Saito
H, Galvin RJ, Kuhstoss SA, Thomas CC, Schipani E, Baron R, et al:
Suppression of Wnt signaling by Dkk1 attenuates PTH-mediated
stromal cell response and new bone formation. Cell Metab.
11:161–171. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Hwang SG, Ryu JH, Kim IC, Jho EH, Jung HC,
Kim K, Kim SJ and Chun JS: Wnt-7a causes loss of differentiated
phenotype and inhibits apoptosis of articular chondrocytes via
different mechanisms. J Biol Chem. 279:26597–26604. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Voronkov A and Krauss S: Wnt/beta-catenin
signaling and small molecule inhibitors. Curr Pharm Des.
19:634–664. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Zhang M, Xie R, Hou W, Wang B, Shen R,
Wang X, Wang Q, Zhu T, Jonason JH and Chen D: PTHrP prevents
chondrocyte premature hypertrophy by inducing cyclin-D1-dependent
Runx2 and Runx3 phosphorylation, ubiquitylation and proteasomal
degradation. J Cell Sci. 122:1382–1389. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Wan M, Yang C, Li J, Wu X, Yuan H, Ma H,
He X, Nie S, Chang C and Cao X: Parathyroid hormone signaling
through low-density lipoprotein-related protein 6. Genes Dev.
22:2968–2979. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Tobimatsu T, Kaji H, Sowa H, Naito J,
Canaff L, Hendy GN, Sugimoto T and Chihara K: Parathyroid hormone
increases beta-catenin levels through Smad3 in mouse osteoblastic
cells. Endocrinology. 147:2583–2590. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Romero G, Sneddon WB, Yang Y, Wheeler D,
Blair HC and Friedman PA: Parathyroid hormone receptor directly
interacts with dishevelled to regulate beta-Catenin signaling and
osteoclastogenesis. J Biol Chem. 285:14756–14763. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Kim IS, Otto F, Zabel B and Mundlos S:
Regulation of chondrocyte differentiation by Cbfa1. Mech Dev.
80:159–170. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Takeda S, Bonnamy JP, Owen MJ, Ducy P and
Karsenty G: Continuous expression of Cbfa1 in nonhypertrophic
chondrocytes uncovers its ability to induce hypertrophic
chondrocyte differentiation and partially rescues Cbfa1-deficient
mice. Genes Dev. 15:467–481. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Kamekura S, Kawasaki Y, Hoshi K, Shimoaka
T, Chikuda H, Maruyama Z, Komori T, Sato S, Takeda S, Karsenty G,
et al: Contribution of runt-related transcription factor 2 to the
pathogenesis of osteoarthritis in mice after induction of knee
joint instability. Arthritis Rheum. 54:2462–2470. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Ueta C, Iwamoto M, Kanatani N, Yoshida C,
Liu Y, Enomoto-Iwamoto M, Ohmori T, Enomoto H, Nakata K, Takada K,
et al: Skeletal malformations caused by overexpression of Cbfa1 or
its dominant negative form in chondrocytes. J Cell Biol.
153:87–100. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Li TF, Dong Y, Ionescu AM, Rosier RN,
Zuscik MJ, Schwarz EM, O'Keefe RJ and Drissi H: Parathyroid
hormone-related peptide (PTHrP) inhibits Runx2 expression through
the PKA signaling pathway. Exp Cell Res. 299:128–136. 2004.
View Article : Google Scholar : PubMed/NCBI
|