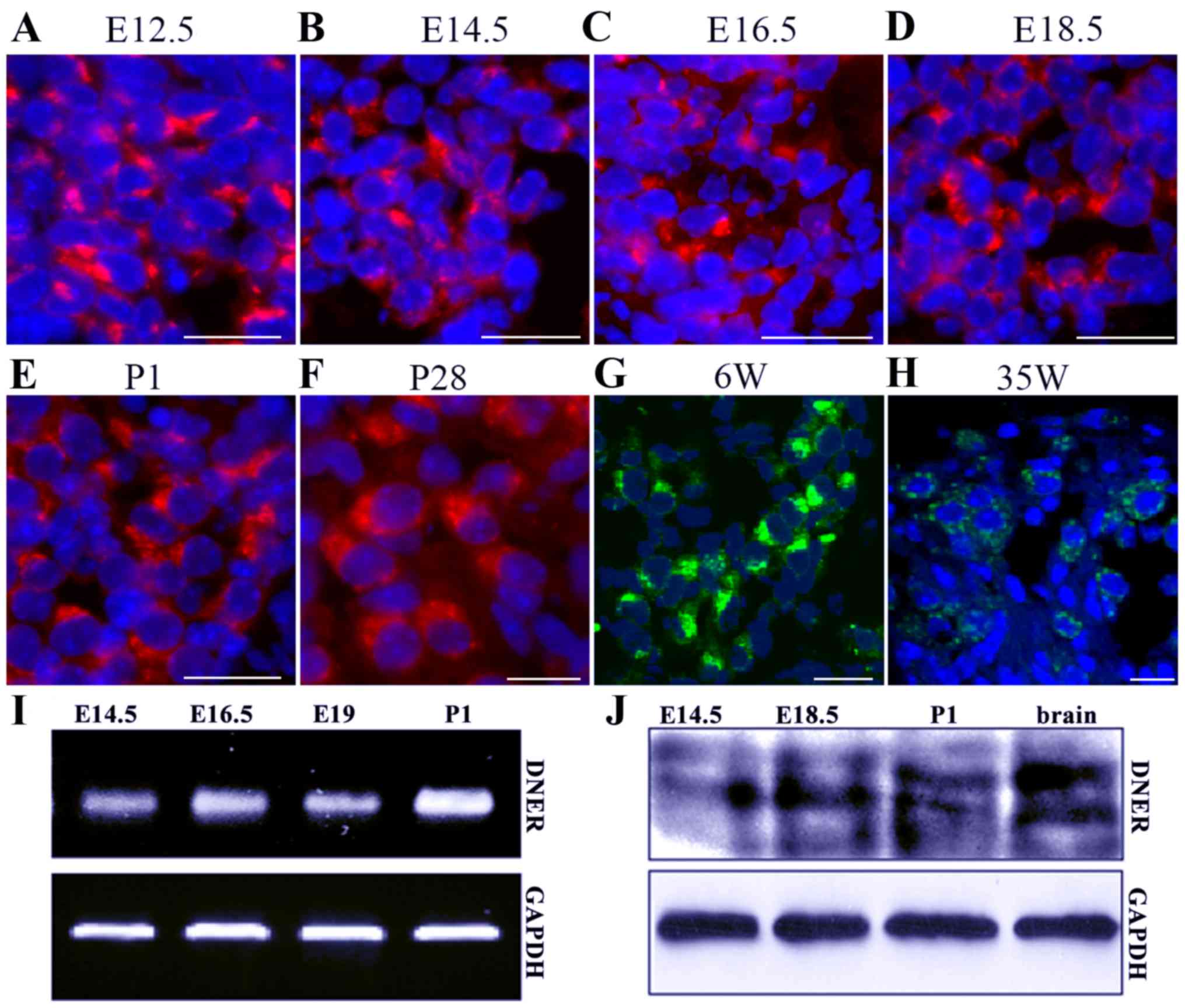
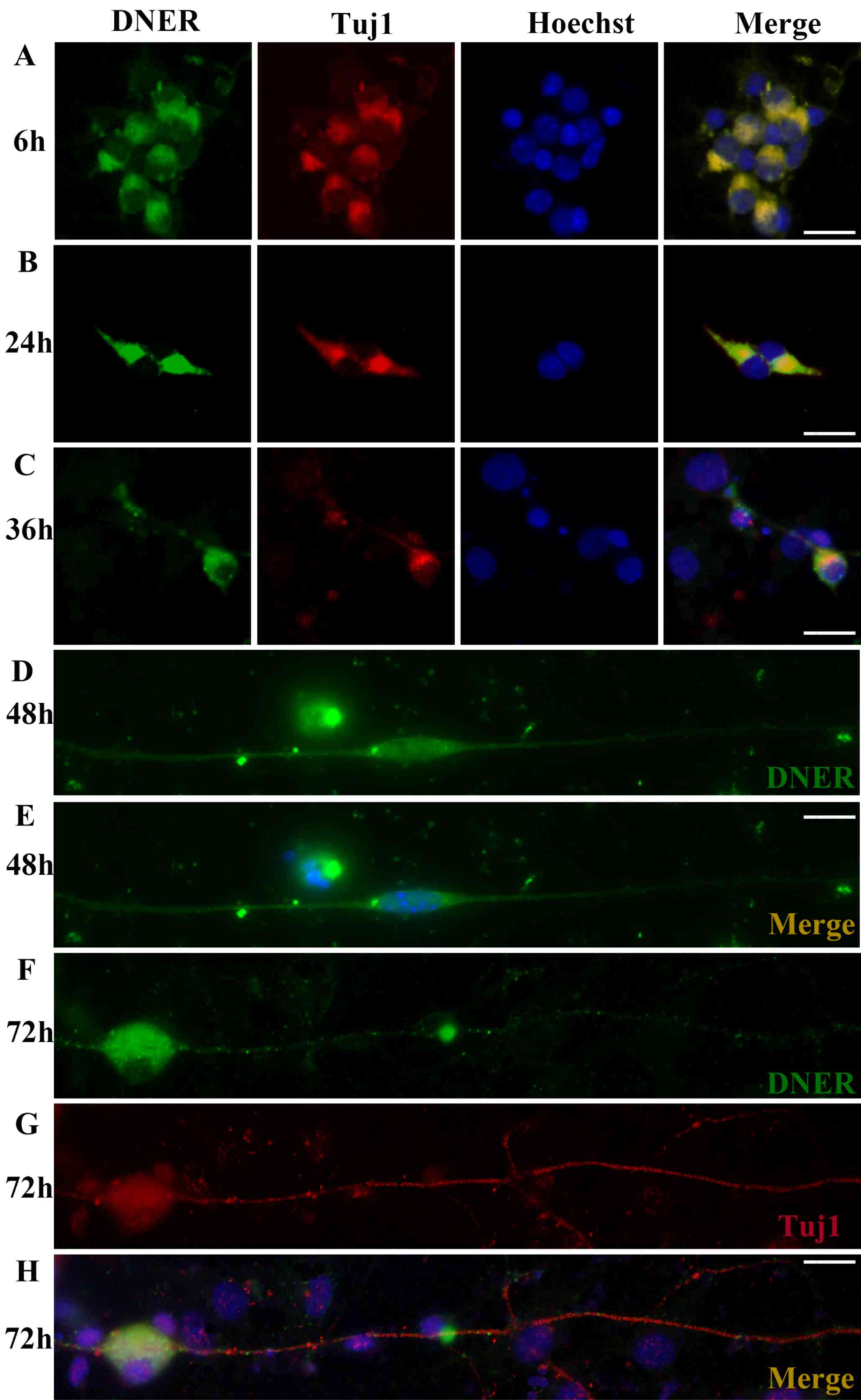
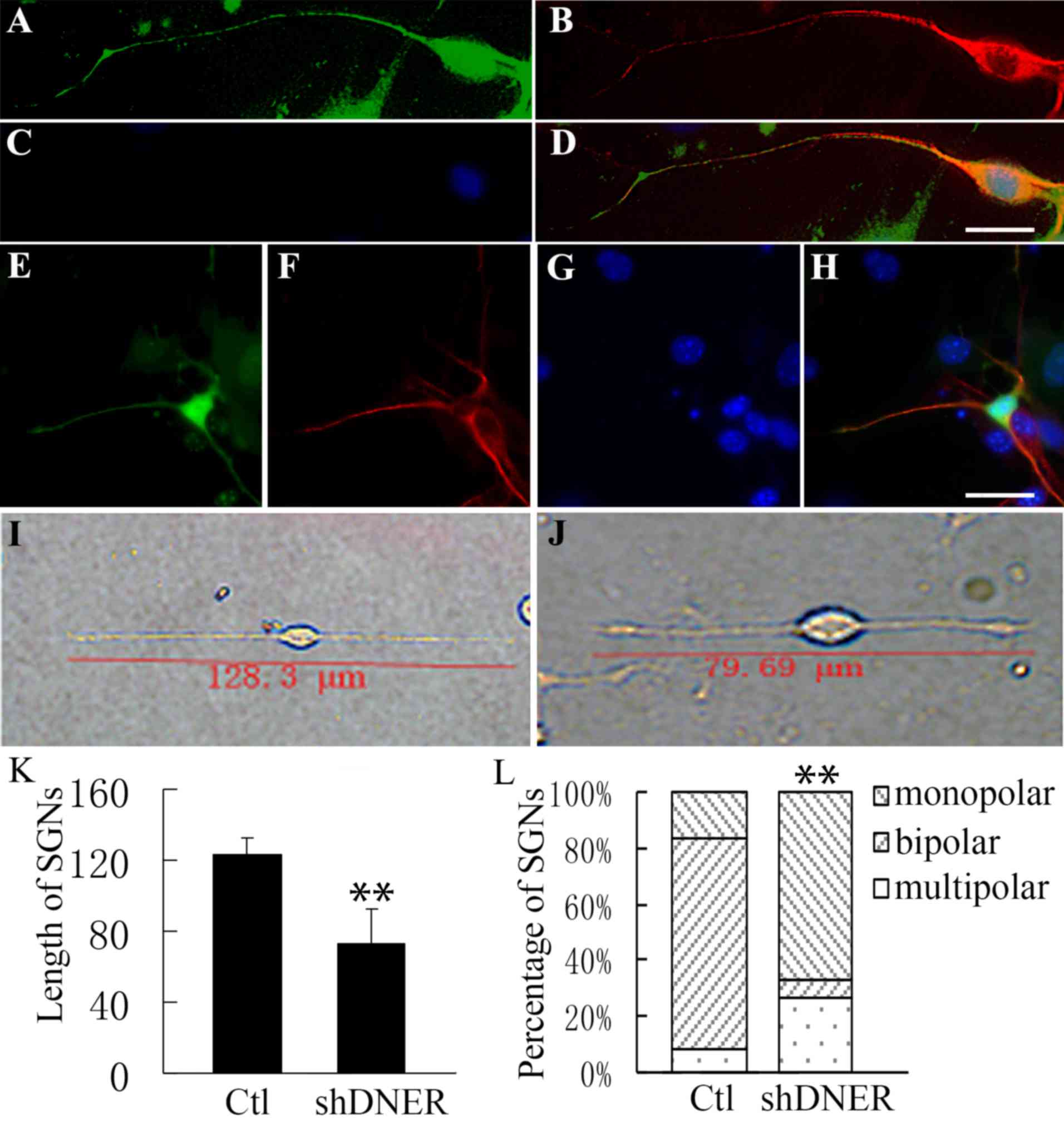
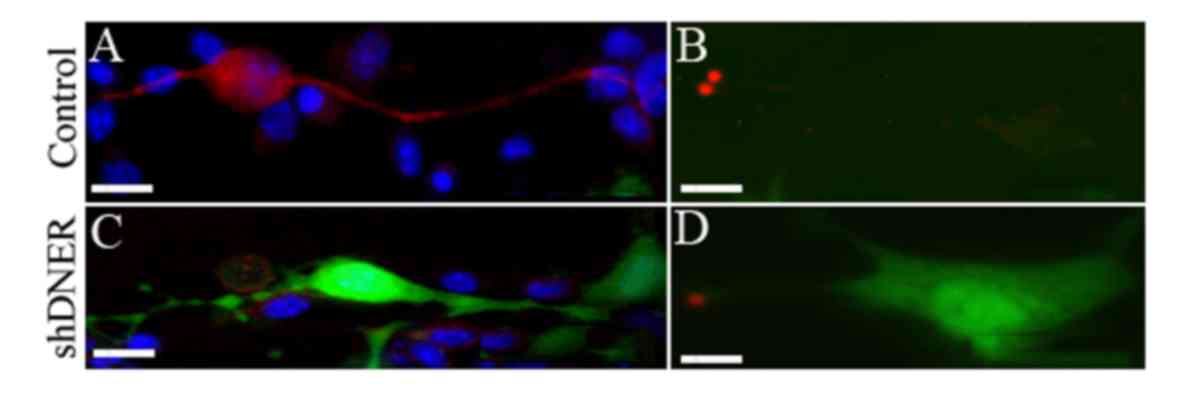
|
1
|
Xu HX, Kim GH, Snissarenko EP, Cureoglu S
and Paparella MM: Multi-channel cochlear implant histopathology:
Are fewer spiral ganglion cells really related to better clinical
performance? Acta Otolaryngol. 132:482–490. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Roehm PC and Hansen MR: Strategies to
preserve or regenerate spiral ganglion neurons. Curr Opin
Otolaryngol Head Neck Surg. 13:294–300. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Petrovic J, Formosa-Jordan P,
Luna-Escalante JC, Abelló G, Ibañes M, Neves J and Giraldez F:
Ligand-dependent Notch signaling strength orchestrates lateral
induction and lateral inhibition in the developing inner ear.
Development. 141:2313–2324. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kiernan AE: Notch signaling during cell
fate determination in the inner ear. Semin Cell Dev Biol.
24:470–479. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Brooker R, Hozumi K and Lewis J: Notch
ligands with contrasting functions: Jagged1 and Delta1 in the mouse
inner ear. Development. 133:1277–1286. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Haddon C, Jiang YJ, Smithers L and Lewis
J: Delta-Notch signalling and the patterning of sensory cell
differentiation in the zebrafish ear: Evidence from the mind bomb
mutant. Development. 125:4637–4644. 1998.PubMed/NCBI
|
|
7
|
Eiraku M, Hirata Y, Takeshima H, Hirano T
and Kengaku M: Delta/notch-like epidermal growth factor
(EGF)-related receptor, a novel EGF-like repeat-containing protein
targeted to dendrites of developing and adult central nervous
system neurons. J Biol Chem. 277:25400–25407. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Nishizumi H, Komiyama T, Miyabayashi T,
Sakano S and Sakano H: BET, a novel neuronal transmembrane protein
with multiple EGF-like motifs. Neuroreport. 13:909–915. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Hsieh FY, Ma TL, Shih HY, Lin SJ, Huang
CW, Wang HY and Cheng YC: Dner inhibits neural progenitor
proliferation and induces neuronal and glial differentiation in
zebrafish. Dev Biol. 375:1–12. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Eiraku M, Tohgo A, Ono K, Kaneko M,
Fujishima K, Hirano T and Kengaku M: DNER acts as a neuron-specific
Notch ligand during Bergmann glial development. Nat Neurosci.
8:873–880. 2005. View
Article : Google Scholar : PubMed/NCBI
|
|
11
|
Saito SY and Takeshima H: DNER as key
molecule for cerebellar maturation. Cerebellum. 5:227–231. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Tohgo A, Eiraku M, Miyazaki T, Miura E,
Kawaguchi SY, Nishi M, Watanabe M, Hirano T, Kengaku M and
Takeshima H: Impaired cerebellar functions in mutant mice lacking
DNER. Mol Cell Neurosci. 31:326–333. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hartman BH, Nelson BR, Reh TA and
Bermingham-McDonogh O: Delta/notch-like EGF-related receptor (DNER)
is expressed in hair cells and neurons in the developing and adult
mouse inner ear. J Assoc Res Otolaryngol. 11:187–201. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kowalik L and Hudspeth AJ: A search for
factors specifying tonotopy implicates DNER in hair-cell
development in the chick's cochlea. Dev Biol. 354:221–231. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Fukazawa N, Yokoyama S, Eiraku M, Kengaku
M and Maeda N: Receptor type protein tyrosine phosphatase
zeta-pleiotrophin signaling controls endocytic trafficking of DNER
that regulates neuritogenesis. Mol Cell Biol. 28:4494–4506. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Richardson L, Venkataraman S, Stevenson P,
Yang Y, Moss J, Graham L, Burton N, Hill B, Rao J, Baldock RA and
Armit C: EMAGE mouse embryo spatial gene expression database: 2014
update. Nucleic Acids Res. 42(Database issue): D835–D844. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Oshima K, Grimm CM, Corrales CE, Senn P,
Martinez Monedero R, Géléoc GS, Edge A, Holt JR and Heller S:
Differential distribution of stem cells in the auditory and
vestibular organs of the inner ear. J Assoc Res Otolaryngol.
8:18–31. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zhao LD, Guo WW, Lin C, Li LX, Sun JH, Wu
N, Ren LL, Li XX, Liu HZ, Young WY, et al: Effects of DAPT and
Atoh1 overexpression on hair cell production and hair bundle
orientation in cultured organ of corti from neonatal rats. PLoS
One. 6:e237292011. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Sage C, Huang M, Karimi K, Gutierrez G,
Vollrath MA, Zhang DS, Garcia-Añoveros J, Hinds PW, Corwin JT,
Corey DP and Chen ZY: Proliferation of functional hair cells in
vivo in the absence of the retinoblastoma protein. Science.
307:1114–1118. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Madden TA, Barrow D, McClelland RA, Gee JM
and Nicholson RI: Modulation of oestrogen action by receptor gene
inhibition. Eur J Cancer. 36 Suppl 4:S34–S35. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Artavanis-Tsakonas S, Rand MD and Lake RJ:
Notch signaling: Cell fate control and signal integration in
development. Science. 284:770–776. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Berezovska O, McLean P, Knowles R, Frosh
M, Lu FM, Lux SE and Hyman BT: Notch1 inhibits neurite outgrowth in
postmitotic primary neurons. Neuroscience. 93:433–439. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Redmond L, Oh SR, Hicks C, Weinmaster G
and Ghosh A: Nuclear Notch1 signaling and the regulation of
dendritic development. Nat Neurosci. 3:30–40. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Redmond L and Ghosh A: The role of Notch
and Rho GTPase signaling in the control of dendritic development.
Curr Opin Neurobiol. 11:111–117. 2001. View Article : Google Scholar : PubMed/NCBI
|