Introduction
Gliomas are serious primary brain tumors, which
account for 80% of malignant tumors of the central nervous system
in the United States. Current therapeutic strategies for this type
of brain cancer are inadequate and the median survival rate for
patients with malignant glioma is 14 months due to their aggressive
nature, high recurrence rate, and gradual increase in radiotherapy
and chemotherapy resistance (1).
Therefore, it is vital to examine the mechanisms underlying the
progression of glioma and to urgently develop novel therapies with
improved therapeutic effects for the treatment of patients with
glioma.
Ouabain is a steroid, which is extracted from the
seeds and leaves of digitalis (2).
As a cardiac glycoside, ouabain is used in the treatment of
congestive heart disease due to its positive cardiac inotropic
effect by inhibiting Na+/K+-ATPase or
Na+/K+ pumps and increasing intracellular
calcium (3). Previous studies have
shown that the plasma transporter,
Na+/K+-ATPase, which is the unique receptor
established for ouabain, not only functions as an ion pump, but is
overexpressed in different types of tumor, including lung cancer,
hepatoma and gliomas (4–6), and its inhibitors are concerned with
the regulation of multiple intracellular signaling pathways,
including Akt/mammalian target of rapamycin (mTOR), Ras and protein
kinase C (PKC) (7).
Previous studies have reported that, as a specific
Na+/K+-ATPase inhibitor, ouabain may affect
the adhesion and metastasis of tumor cells, resulting in the
apoptosis of several tumor cells, including malignant breast
cancer, prostate cancer and human leukemia (8). In addition, epidemiological evidence
shows that tumor patients treated with cardiac glycosides have
lower mortality rates, compared with those treated without cardiac
glycosides, suggesting that ouabain has potential antitumor effects
(9). However, the mechanism
underlying its antitumor effect remains to be elucidated.
In the present study, the human glioblastoma U-87MG
cell line was selected to examine the effects of ouabain, as a
specific Na+/K+-ATPase inhibitor, on the
apoptosis of U87-MG cells, followed by preliminarily investigation
of the role of the Akt/mTOR signaling pathway in the process.
Materials and methods
Materials
Dulbecco's modified Eagle's medium (DMEM) and fetal
bovine serum (FBS) were obtained from Gibco; Thermo Fisher
Scientific, Inc. (Waltham, MA, USA). Ouabain was purchased from
Sigma-Aldrich (Merck KGaA, Darmstadt, Germany).
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT)
was purchased from (Promega Corporation, Madison, WI, USA).
Cell culture and grouping
The human U-87MG glioma cell line was obtained from
the American Type Culture Collection (Mannasas, VA, USA) and
cultured in DMEM containing 10% FBS and 1% penicillin/streptomycin
in a humidified incubator of 5% CO2 at 37°C. The cells
were randomized into two groupings: Ouabain-treated groups (0.05,
0.25, 0.5, 2.5, 5 and 25 µmol/l for the MTT assay; 0.05, 0.5, 2.5
and 25 µmol/l for other experiments) and a control group (cultured
with an equal volume of culture medium containing 0.1% DMSO).
MTT assay
For the MTT assays, 4×103 cells were
seeded into each well of 96-well plates. Following conventional
culture for 24 h, 200 µl of the different concentrations of ouabain
were added into each well and the cells were cultured for another
24 h at 37°C. In the control group, the cells were cultured in 200
µl of culture medium containing 0.1% DMSO only. Following culture,
10 µl of 5 mg/ml MTT was added to the cells in each well and
incubated for 4 h. Subsequently, 150 µl of DMSO (150 µl) was added
for 10 min and the plates were positioned in a microplate
autoreader (Thermo Fisher Scientific, Inc.) The optical density at
490 nm wavelength was read. On the basis of the optical density (A
value), cell viability was calculated as follows: Cell
viability=average A value of ouabain-treated cells/average A value
of control ×100%. The half maximal inhibitory concentration
(IC50) value was calculated as the concentration
required for 50% inhibition of cell growth.
Digital holographic microscopy (DHM)
assay
The DHM (HoloMonitor™ M4; Phase Holographic Imaging
AB, Lund, Sweden) was used to record the 3D structure of adherent
cells via interfering wave front induction when exposed to the 0.8
mW HeNe laser (633 nm). The DHM was based on measurements of how
the cells shift light, which passes through the cells without any
labeling or staining. The cells were plated in 6-well plates at
1×105/ml and cultured for 24 h, following which
different concentrations of ouabain were added into each well prior
to being moved to the DHM. This was placed in an incubator and was
used to capture time-lapse images of the 6-well plates at 5 min
intervals for 24 h. For each sample, a region for imaging was
randomly selected and changes in cell number were analyzed using
HoloStudio 2.6.2 software (Holographic Imaging AB). In addition, 10
cells were randomly selected per well to perform cell motion
tracking, and cell motility was determined as the distance of cell
movement throughout the 24 h of time-lapse video.
Protein extraction and western blot
analysis
Following treatment with 0.05, 0.5, 2.5 and 25
µmol/l of ouabain for 24 h, the cells were rinsed in ice-cold PBS
three times, collected with a cell scraper, and lysed in lysis
buffer containing 50 mmol/l Tris-HCl (pH 7.2), 250 mmol/l NaCl,
0.1% NP-40, 2 mmol/l EDTA, 10% glycerol, 1 mmol/l PMSF, 5 µg/ml
aprotinin and 5 µg/ml leupeptin on ice. The concentrations of the
proteins of interest were quantified using the BCA method. Protein
samples (40 µg) of each group were separated by 10% polyacrylamide
gel electrophoresis and transferred onto nitrocellulose membranes,
which were blocked at room temperature by 5% nonfat milk for 1 h.
The membranes were then incubated with primary antibodies including
Akt (ab179463, 1:1,000; Abcam, Cambridge, MA, USA), p-Akt (ab81283,
1:1,000; Abcam, Cambridge, MA, USA), mTOR (ab32028, 1:1,000;
Abcam), p-mTOR (ab109268, 1:1,000; Abcam) and HIF-1α (ab51608,
1:1,000; Abcam) overnight at 4°C and were subsequently incubated
with secondary antibody (anti-rabbit IgG; 5210-0174; 1:5,000; KPL,
Inc., Gaithersburg, MD, USA) for 2 h at room temperature. β-actin
was used as the internal control for protein loading.
High-content screening (HCS)
assay
The cells were seeded in 96-well plates at
2×105/well, cultured for 24 h, and treated with
different concentrations of ouabain added into each well. The cells
were then fixed with 4% paraformaldehyde and permeabilized with
0.25% Triton-X 100. The fixed cells were nonspecifically blocked in
5% nonfat milk for 1 h and incubated with primary antibodies (Akt,
p-Akt, mTOR, p-mTOR, and HIF-1α) at 1:1,000 dilution overnight at
4°C. Following incubation, the cells were stained with FITC-labeled
anti-rabbit IgG secondary antibody (5230–0298; 1:200 dilution; KPL,
Inc., Gaithersburg, MD, USA) for 1 h at room temperature, and 1
g/ml of DAPI for 10 min. The immunofluorescence of the stained
cells was quantified using the HCA cell analyzer 2200 (GE
Healthcare Life Sciences, Chalfont, UK).
Statistical analysis
SPSS 13.0 statistical software (SPSS, Inc., Chicago,
IL, ISA) was used for data analysis. All data are expressed as the
mean ± standard deviation and were analyzed using one-way analysis
of variance followed Tukey's post hoc analysis. P<0.05 was
considered to indicate a statistically significant difference.
Results
Ouabain suppresses the viability of
U-87MG cells
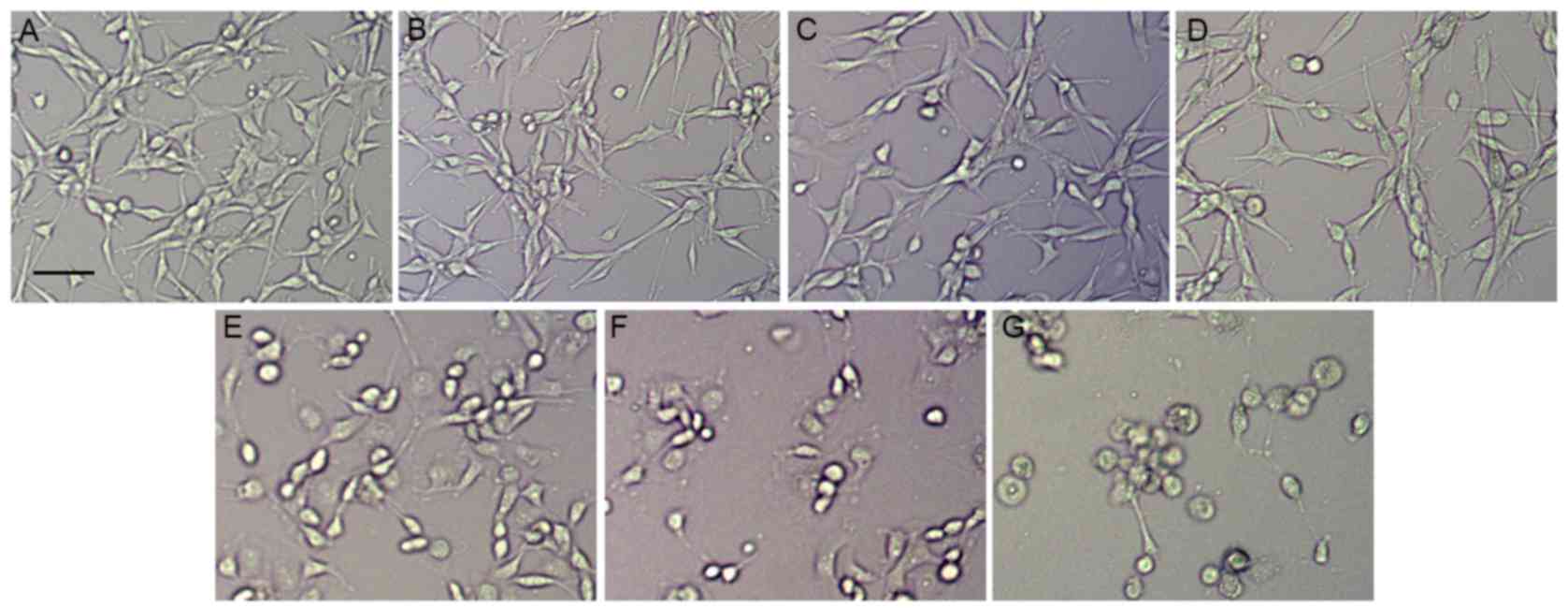
To assess the effect of ouabain on U-87MG cells, the
cultured U-87MG cells were exposed to different concentrations of
ouabain for 24 h. The U-87MG cells in the control group exhibited
orderly shapes, well-defined borders and satisfactory diopter. By
contrast, the cells treated with increasing concentrations of
ouabain gradually shrunk, and exhibited poorly defined margins and
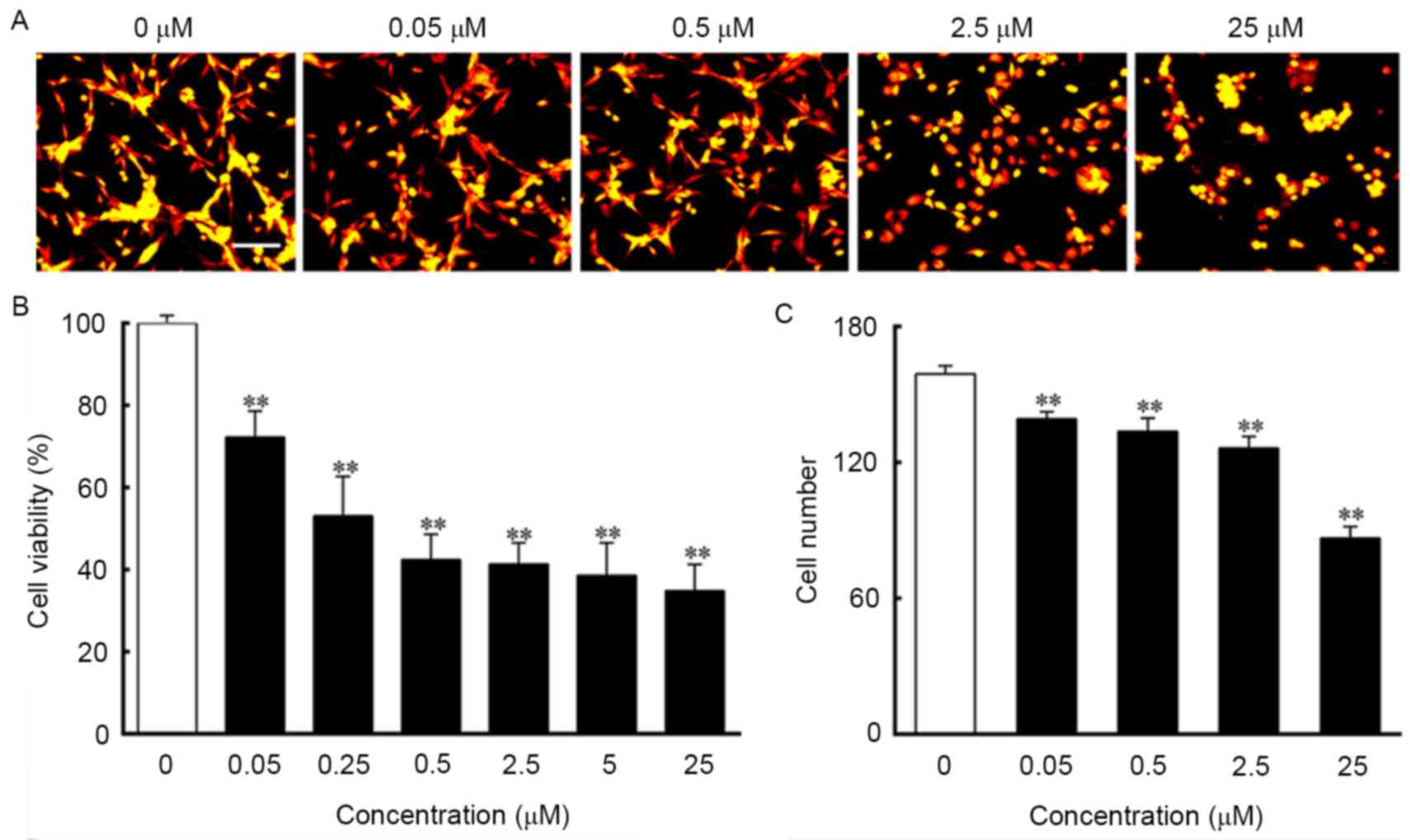
irregular shapes, with cell death observed (Fig. 1A-G). As shown in Fig. 2A, the control cells grew into
clusters with a normal morphology, whereas the cells treated with
ouabain exhibited increasing shrinkage as the concentration of
ouabain increased. The results of the MTT assay indicated that the
viability of cells treated with ouabain were significantly
decreased and in a dose-dependent manner, compared with those in
the control group. The IC50 of ouabain treatment for 24
h was 0.45 µmol/l (Fig. 2B). The
analysis of cell numbers suggested that ouabain treatment decreased
the number of cells in a dose-dependent manner, which was in line
with the morphological changes and the MTT results (Fig. 2C).
Ouabain inhibits the motility of
U-87MG cells
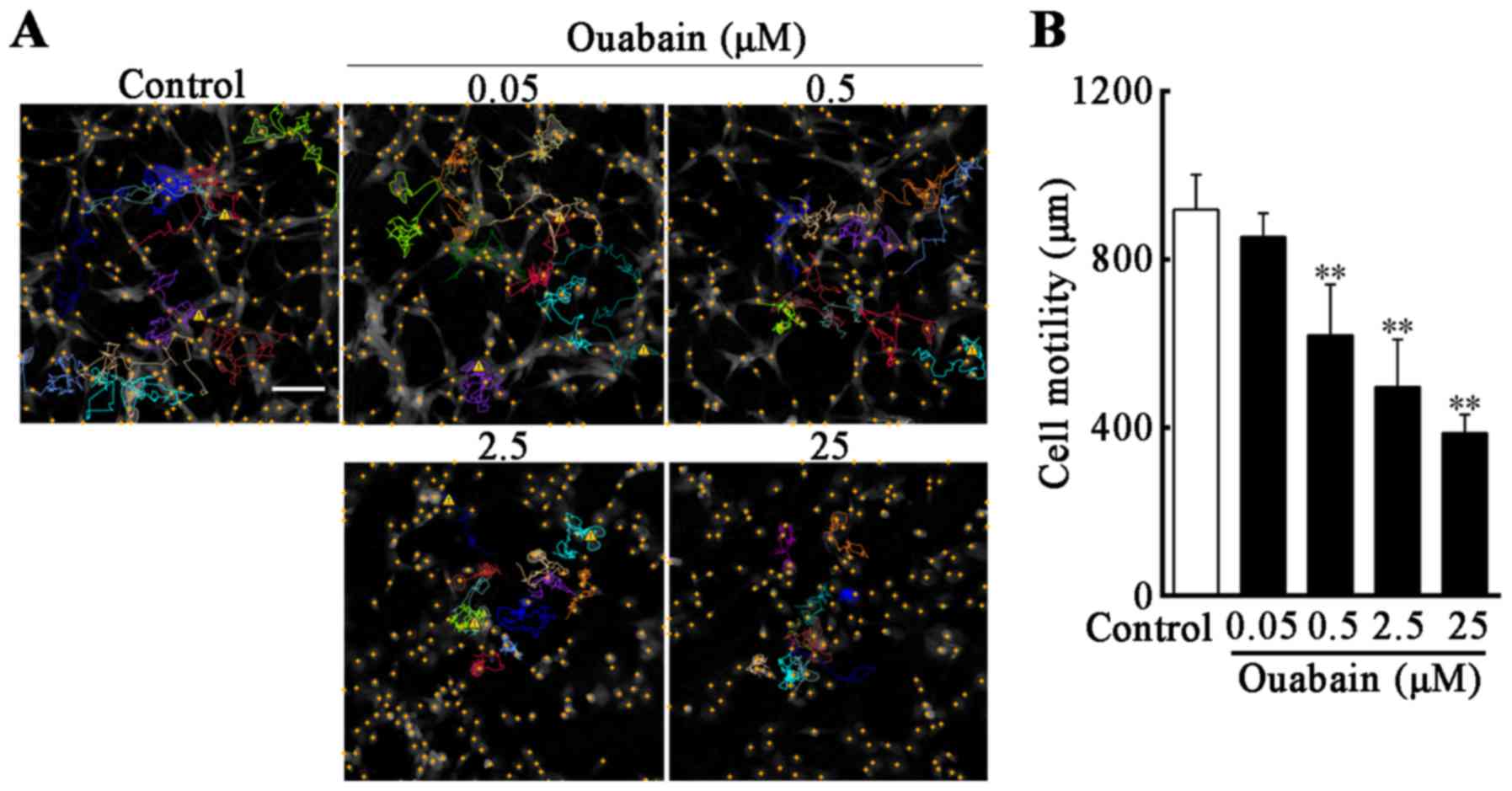
In order to investigate whether U-87MG cell motility
was reduced under ouabain treatment, compared with controls,
time-lapse videos were used to assess real-time cell motility, and
cell displacement in the control and under different concentrations
of ouabain were tracked. The cell motion tracking indicated that,
following treatment with ouabain for 24 h, the movement range of
U-87MG cells was decreased as the concentration of ouabain
increased, and the routes of selected cells were irregular and
multidirectional (Fig. 3A).
Compared with the control (919±79 µm), the migration ability of the
U-87MG cells in 0.5, 2.5 and 25 µmol/l ouabain treatment groups
were suppressed in a dose-dependent manner (0.5 µmol/l, 620±115 µm;
2.5 µmol/l, 498±107 µm; 25 µmol/l, 387±42 µm; P<0.01; Fig. 3B).
Ouabain affects the expression of
Akt/mTOR signaling pathway proteins
It has been shown that the survival and
proliferation of glioma cells can be enhanced by activation of the
Akt/mTOR signaling pathway, which has been targeted for glioma
treatment (10). To obtain
additional evidence for the ouabain-inhibited viability and
motility of U-87MG cells, the expression of Akt/mTOR signaling
pathway regulators were analyzed using western blot analysis and
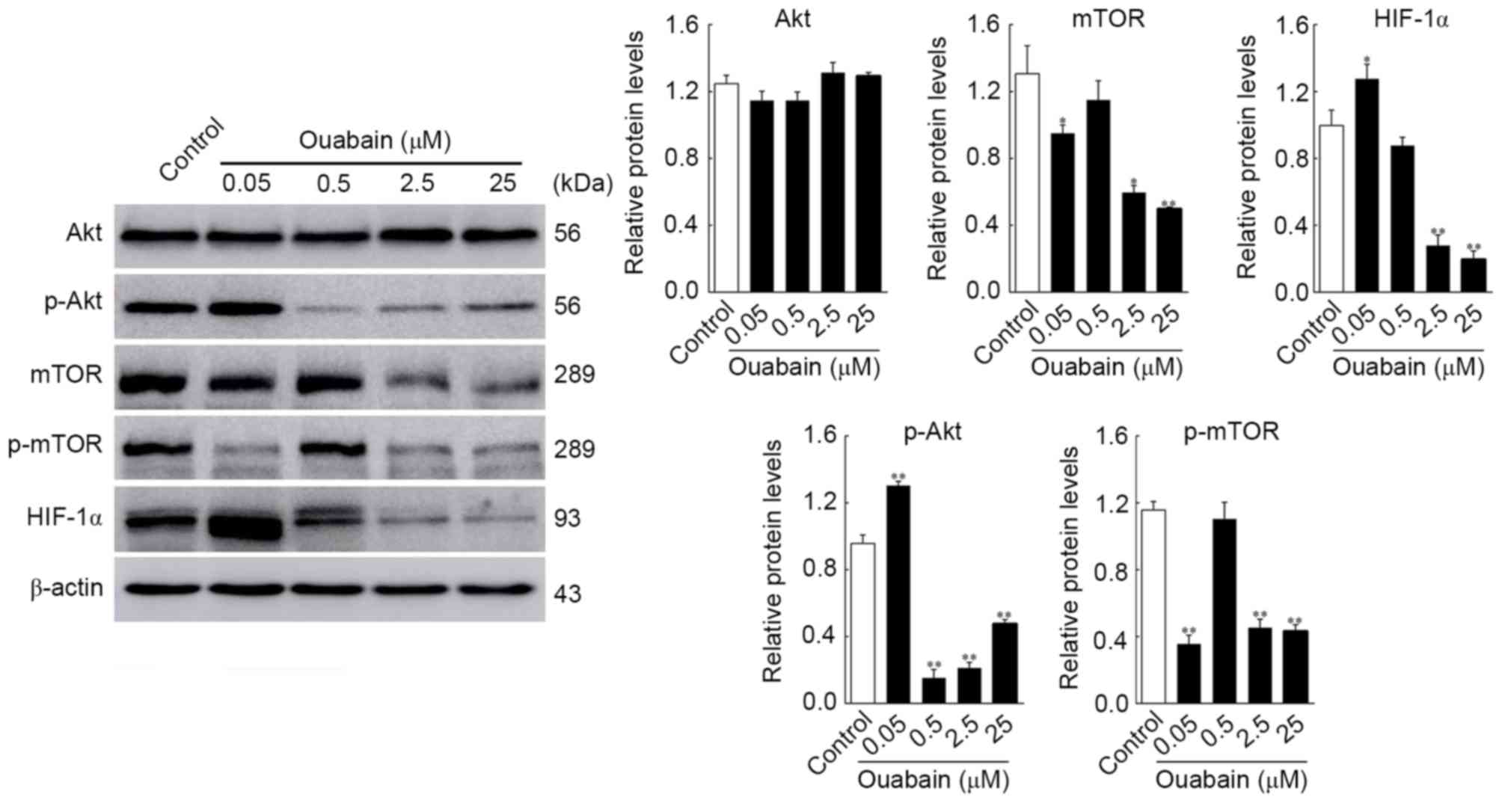
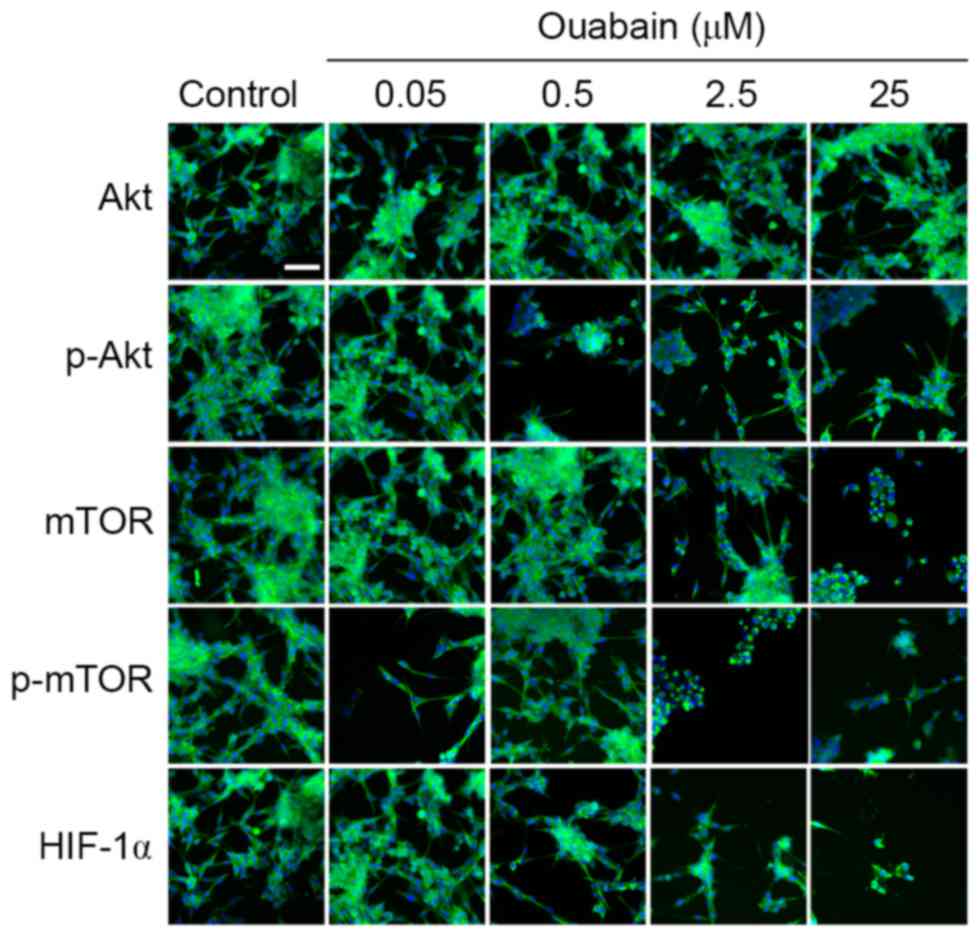
HCS. As shown in Fig. 4, the
protein expression levels of p-Akt and HIF-1α in the 0.05 µmol/l
ouabain-treated group was significantly altered, compared with that
in the control group (p-Akt, 1.30±0.02, vs. 0.96±0.04, P<0.01;
HIF-1α, 1.32±0.24, vs. 1.06±0.16, P<0.05;), whereas that of mTOR
and p-mTOR decreased rapidly (mTOR, 0.95±0.04, vs. 1.31±0.13,
P<0.05; p-mTOR, 0.36±0.04, vs. 1.16±0.04, P<0.01). Treatment
with 0.5 µmol/l ouabain reduced only the expression of p-Akt
(0.15±0.04, vs. 0.96±0.04, P<0.01). In addition, the expression
levels of Akt, mTOR, p-mTOR and HIF-1α were decreased by 2.5 and 25
µmol/l ouabain: p-Akt, 0.21±0.02 and 0.48±0.02, vs. 0.96±0.04;
mTOR, 0.60±0.03 and 0.50±0.01, vs. 1.31±0.14; p-mTOR, 0.45±0.04 and
0.43±0.03, vs. 1.16±0.04; HIF-1α, 0.28±0.05 and 0.20±0.04, vs.
1.06±0.16 (P<0.05 or P<0.01). However, no significant
differences were found in the expression of Akt following treatment
with ouabain (P>0.05). These results were corroborated by HCS
assays (Fig. 5). Therefore, it was
hypothesized that ouabain mediated the suppression of U-87MG cell
viability and motility via downregulating proteins of the Akt/mTOR
signaling pathway.
Discussion
There has been important progress in therapeutic
strategies for gliomas in previous decades, as one of the most
malignant types of brain tumor with high relapse and mortality
rates. In the present study, ouabain intervention of glioma U-87MG
cells was performed for the experimental investigation of
therapeutic strategies for glioma, as U-87MG cells are more
susceptible to cardiac glycosides, and ouabain can cross the blood
brain barrier as a liquid, soluble cardiac glycoside.
Na+/K+-ATPase comprises an α
subunit and β subunit, and the α subunit, which is essential in the
transport activity of Na+/K+-ATPase, contains
multiple binding sites of ATP and the enzyme inhibitor ouabain
(11). Increasing evidence
suggests that Na+/K+-ATPase is essential in
the regulation of ion homeostasis and formation of the pathogenesis
of glioma. Ahmed et al (12) reported that the inhibition of
Na+/K+-ATPase regulated cell adhesion and
migration, inhibited intracellular PKC and the NF-κB signaling
pathway, and consequently resulted in the apoptosis of tumor cells.
Additionally, Lin et al (13) found that ouabain inhibited the
α-subunit activity of Na+/K+-ATPase,
suppressed tumor cell growth, and eventually caused tumor cell
death. The present study revealed that ouabain intervention
inhibited U-87MG cell viability and cell motility in a
dose-dependent manner, suggesting that ouabain promoted the
apoptosis of U87-MG cells, which may be associated with alterations
in certain intracellular signaling pathways.
In previous years, there has been an improved
understanding of the role of Na+/K+-ATPase in
multiple signaling pathways, including Akt/mTOR, Ras and PKC,
resulting in the promotion of tumor invasion and metastasis
(14). Notably, the Akt/mTOR
signaling pathway is crucial for tumor progression according to the
regulation of cell survival, proliferation, invasion and stem cell
function (15). The upregulation
of Akt/mTOR can directly or indirectly inhibit the phosphorylation
of B-cell lymphoma 2-associated X protein, caspase-9 and other
apoptosis-associated proteins, which attenuates apoptosis and leads
to the deterioration of physiological functions (16). Liu et al (17) confirmed the effect of inhibition of
the Akt/mTOR pathway against apoptosis in glioma; the
pharmacological suppression of Akt/mTOR and their phosphorylation
upregulates downstream proteins correlated with apoptosis, inducing
an increase in the apoptosis of tumor cells.
Previous studies have confirmed that ouabain
depresses the phosphorylation of Akt and mTOR via regulating
phosphoinositide 3-kinase (PI3K), Ras and PKC signaling pathways,
which causes cell growth depression and the enhancement of
apoptosis (13). In the present
study, it was found that the phosphorylation of Akt and mTOR
significantly reduced cell viability, survival rate and cell
mobility when treated with 2.5 and 25 µmol/l of ouabain, which
confirmed that the antitumor property of ouabain involved the
alteration of certain intracellular signaling pathways.
The overexpression of HIF-1α has been reported in
several types of human tumor and metastasis, including glioma,
which is closely associated with tumor invasion and metastasis,
resistance to chemotherapy and radiotherapy, and poor prognosis
(18). As an important regulator
in hypoxic microenvironments, resulting from insufficient oxygen
and nutrient supply for rapidly growing tumors, the activation of
HIF-1α promotes tumor adaption to hypoxia through the promotion of
cell survival, motility and angiogenesis (19,20).
Soeda et al (21)
demonstrated that the hypoxic induction of HIF-1α facilitated the
self-renewal and proliferation of glioma stem cells, and maintained
glioma stem cells in an undifferentiated state with multilineage
potential. This assisted in glioma progression and metastasis.
In addition, previous studies have documented that
the Akt/mTOR pathway is associated with hypoxia signaling
transduction. The phosphorylation of Akt and mTOR can intensify the
hypoxic induction of HIF-1α at the translational level, and
promotes the stabilization and activation of HIF-1α, which
regulates intracellular glucose utilization and angiogenesis,
eventually promoting the growth of tumor cells (19,22).
García-Maceira and Mateo demonstrated that inhibiting the
phosphorylation of Akt and mTOR occurred with loss of the
expression of HIF-1α, whereas inhibition of the Akt/mTOR pathway
decreased the translation of HIF-1α and ultimately led to apoptosis
of tumor cells (23).
Consistently, the present study demonstrated that the
downregulation of HIF-1α was simultaneous with the reduced
phosphorylation of Akt and mTOR at high concentrations of ouabain
(2.5 and 25 µmol/l), suggesting that the regulation of HIF-1α by
the Akt/mTOR pathway was involved in tumor suppressive effect of
ouabain.
In conclusion, the data obtained in the present
study suggested that high concentrations of ouabain may depress
human glioma U-87MG cell growth, attenuate cell mobility and lead
to apoptosis via inhibiting the intracellular Akt/mTOR pathway and
associated HIF-1α. These data identify ouabain as a potential
target for the treatment of glioma in the future.
Acknowledgements
Not applicable.
Funding
This study was supported by grants from the Tianjin
Natural Science Foundation (grant no. 17JCYBJC25700 and
15ZXLCSY00040), the National Natural Science Foundation of China
(grant nos. 31200809, 81273552 and 81673651) and the Open Research
Funding Program from Logistics University of PAP (grant no.
2015ZXKF09).
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
XSY, ZWX, TLY, YT and SXC conceived and designed the
study. XSY, ZWX and TLY performed the experiments; RCX, JL, WBZ,
SZ, HTS and ZQY analyzed the results. XSY and SXC wrote the paper.
XSY, ZWX and TLY contributed equally to this study.
Co-corresponding authors of YT and SXC contributed equally to this
study. All authors reviewed and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
All authors declared that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
FBS
|
fetal bovine serum
|
|
DHM
|
digital holographic microscopy
|
|
HCS
|
high-content screening
|
References
|
1
|
Jain A, Lai JC and Bhushan A: Biochanin A
inhibits endothelial cell functions and proangiogenic pathways:
Implications in glioma therapy. Anticancer Drugs. 26:323–330. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bagrov AY, Shapiro JI and Fedorova OV:
Endogenous cardiotonic steroids: Physiology, pharmacology, and
novel therapeutic targets. Pharmacol Rev. 61:9–38. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kulikov A, Eva A, Kirch U, Boldyrev A and
Scheiner-Bobis G: Ouabain activates signaling pathways associated
with cell death in human neuroblastoma. Biochim Biophys Acta.
1768:1691–1702. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kim N, Yim HY, He N, Lee CJ, Kim JH, Choi
JS, Lee HS, Kim S, Jeong E, Song M, et al: Cardiac glycosides
display selective efficacy for STK11 mutant lung cancer. Sci Rep.
6:297212016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Xu Z, Wang F, Fan F, Gu Y, Shan N, Meng X,
Cheng S, Liu Y, Wang C, Song Y and Xu R: Quantitative proteomics
reveals that the inhibition of Na+/K+-ATPase
activity affects S-phase progression leading to a chromosome
segregation disorder by attenuating the Aurora A function in
hepatocellular carcinoma cells. J Proteome Res. 14:4594–4602. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Yan X, Liang F, Li D and Zheng J: Ouabain
elicits human glioblastoma cells apoptosis by generating reactive
oxygen species in ERK-p66SHC-dependent pathway. Mol Cell Biochem.
398:95–104. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Zhang L, Zhang Z, Guo H and Wang Y:
Na+/K+-ATPase-mediated signal transduction
and Na+/K+- ATPase regulation. Fundam Clin
Pharmacol. 22:615–621. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Desfrere L, Karlsson M, Hiyoshi H,
Malmersjö S, Nanou E, Estrada M, Miyakawa A, Lagercrantz H, El
Manira A, Lal M and Uhlén P: Na, K-ATPase signal transduction
triggers CREB activation and dendritic growth. Proc Natl Acad Sci
USA. 106:pp. 2212–2217. 2009; View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Prassas I and Diamandis EP: Novel
therapeutic applications of cardiac glycosides. Nat Rev Drug
Discov. 7:926–935. 2008. View
Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wei L, Su YK, Lin CM, Chao TY, Huang SP,
Huynh TT, Jan HJ, Whang-Peng J, Chiou JF, Wu AT and Hsiao M:
Preclinical investigation of ibrutinib, a Bruton's kinase tyrosine
(Btk) inhibitor, in suppressing glioma tumorigenesis and stem cell
phenotypes. Oncotarget. 7:69961–69975. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Xie Z: Molecular mechanisms of
Na/K-ATPase-mediated signal transduction. Ann N Y Acad Sci.
986:497–503. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ahmed Z, Deyama Y, Yoshimura Y and Suzuki
K: Cisplatin sensitivity of oral squamous carcinoma cells is
regulated by Na+, K+-ATPase activity rather
than copper-transporting P-type ATPases, ATP7A and ATP7B. Cancer
Chemother Pharmacol. 63:643–650. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Lin Y, Ho DH and Newman RA: Human tumor
cell sensitivity to oleandrin is dependent on relative expression
of Na+, K+-ATPase subunitst. J Exp Ther
Oncol. 8:271–286. 2010.PubMed/NCBI
|
|
14
|
Wu J, Akkuratov EE, Bai Y, Gaskill CM,
Askari A and Liu L: Cell signaling associated with
Na+/K+-ATPase: Activation of
phosphatidylinositide 3-kinase IA/Akt by ouabain is independent of
Src. Biochemistry. 52:9059–9067. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Joy A, Kapoor M, Georges J, Butler L,
Chang Y, Li C, Crouch A, Smirnov I, Nakada M, Hepler J, et al: The
role of AKT isoforms in glioblastoma: AKT3 delays tumor
progression. J Neurooncol. 130:43–52. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Clark PA, Bhattacharya S, Elmayan A,
Darjatmoko SR, Thuro BA, Yan MB, van Ginkel PR, Polans AS and Kuo
JS: Resveratrol targeting of AKT and p53 in glioblastoma and
glioblastoma stem-like cells to suppress growth and infiltration. J
Neurosurg. 126:1448–1460. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Liu Y, Zheng J, Zhang Y, Wang Z, Yang Y,
Bai M and Dai Y: Fucoxanthin activates apoptosis via inhibition of
PI3K/Akt/mTOR pathway and suppresses invasion and migration by
restriction of p38-MMP-2/9 pathway in human glioblastoma cells.
Neurochem Res. 41:2728–2751. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Rapisarda A, Uranchimeg B, Sordet O,
Pommier Y, Shoemaker RH and Melillo G: Topoisomerase I-mediated
inhibition of hypoxia-inducible factor 1: Mechanism and therapeutic
implications. Cancer Res. 64:1475–1482. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Thomas GV, Tran C, Mellinghoff IK, Welsbie
DS, Chan E, Fueger B, Czernin J and Sawyers CL: Hypoxia-inducible
factor determines sensitivity to inhibitors of mTOR in kidney
cancer. Nat Med. 12:122–127. 2006. View
Article : Google Scholar : PubMed/NCBI
|
|
20
|
Jensen RL, Ragel BT, Whang K and Gillespie
D: Inhibition of hypoxia inducible factor-1alpha (HIF-1alpha)
decreases vascular endothelial growth factor (VEGF) secretion and
tumor growth in malignant gliomas. J Neurooncol. 78:233–247. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Soeda A, Park M, Lee D, Mintz A,
Androutsellis-Theotokis A, McKay RD, Engh J, Iwama T, Kunisada T,
Kassam AB, et al: Hypoxia promotes expansion of the CD133-positive
glioma stem cells through activation of HIF-1alpha. Oncogene.
28:3949–3959. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Lum JJ, Bui T, Gruber M, Gordan JD,
DeBerardinis RJ, Covello KL, Simon MC and Thompson CB: The
transcription factor HIF-1alpha plays a critical role in the growth
factor-dependent regulation of both aerobic and anaerobic
glycolysis. Genes Dev. 21:1037–1049. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
García-Maceira P and Mateo J: Silibinin
inhibits hypoxia-inducible factor-1alpha and mTOR/p70S6K/4E-BP1
signalling pathway in human cervical and hepatoma cancer cells:
Implications for anticancer therapy. Oncogene. 28:313–324. 2009.
View Article : Google Scholar : PubMed/NCBI
|