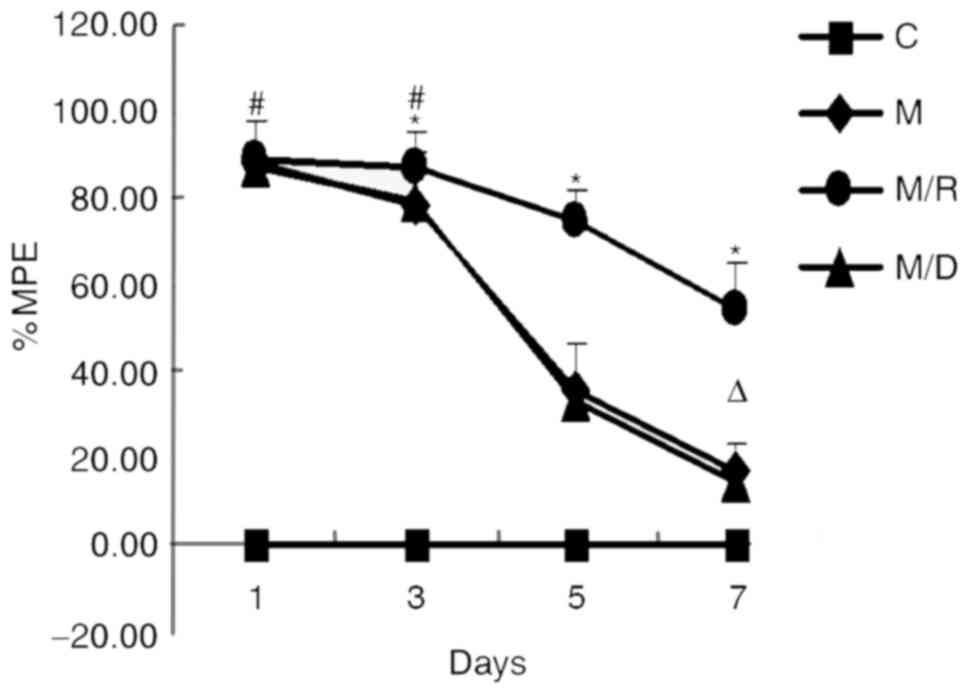
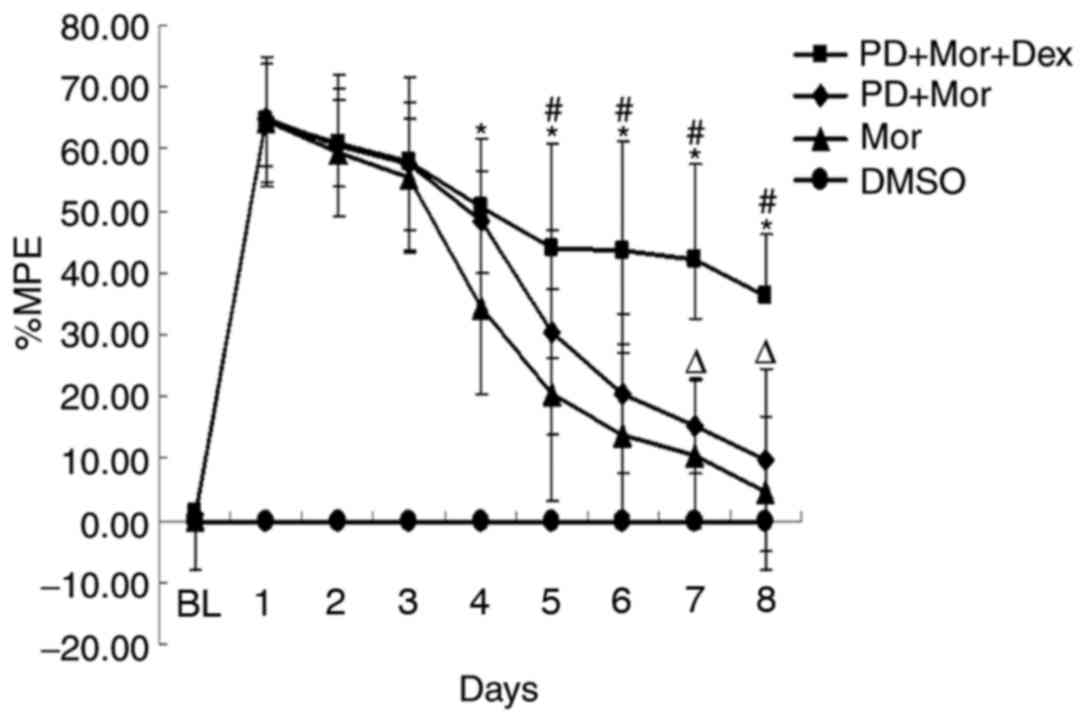
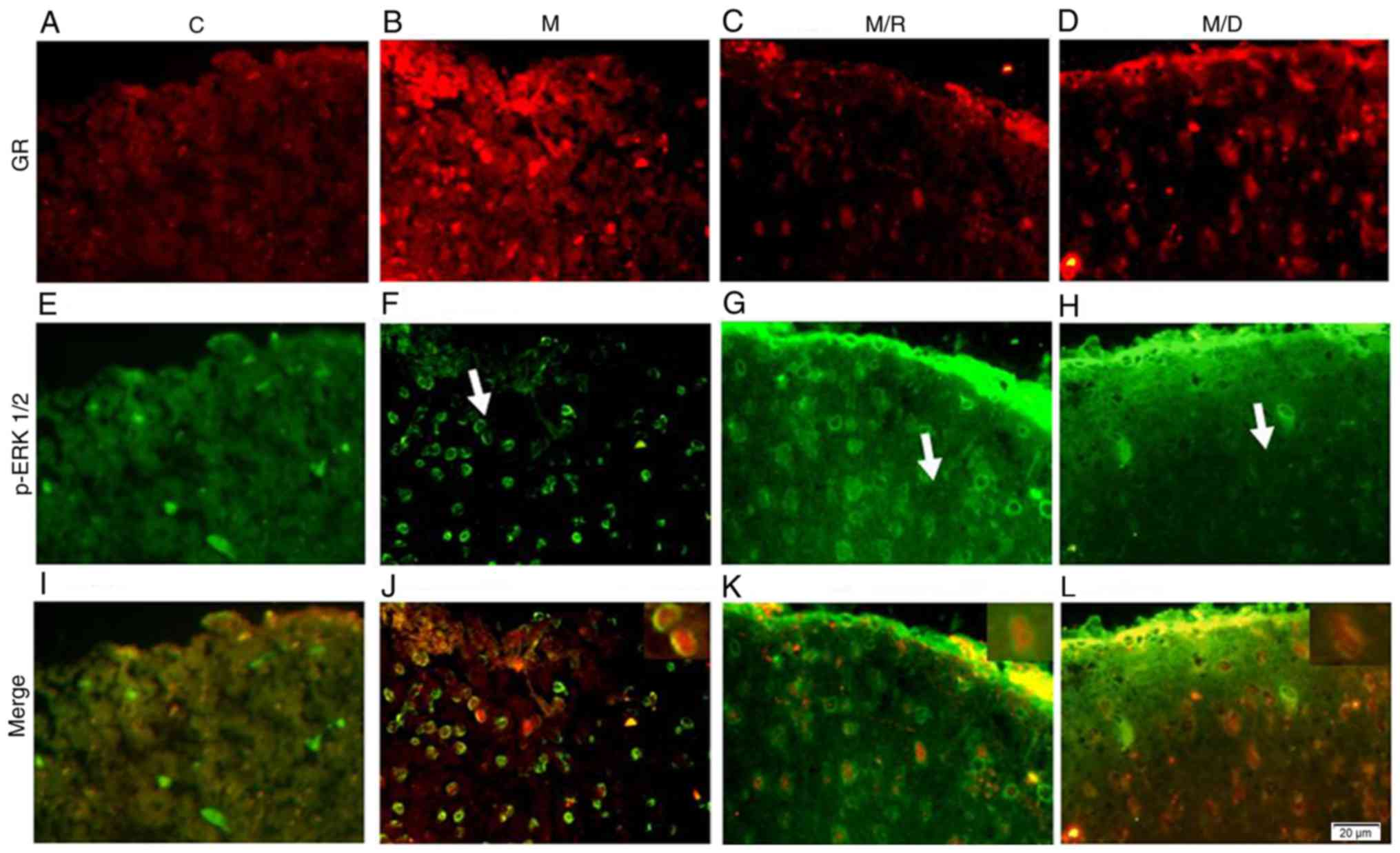
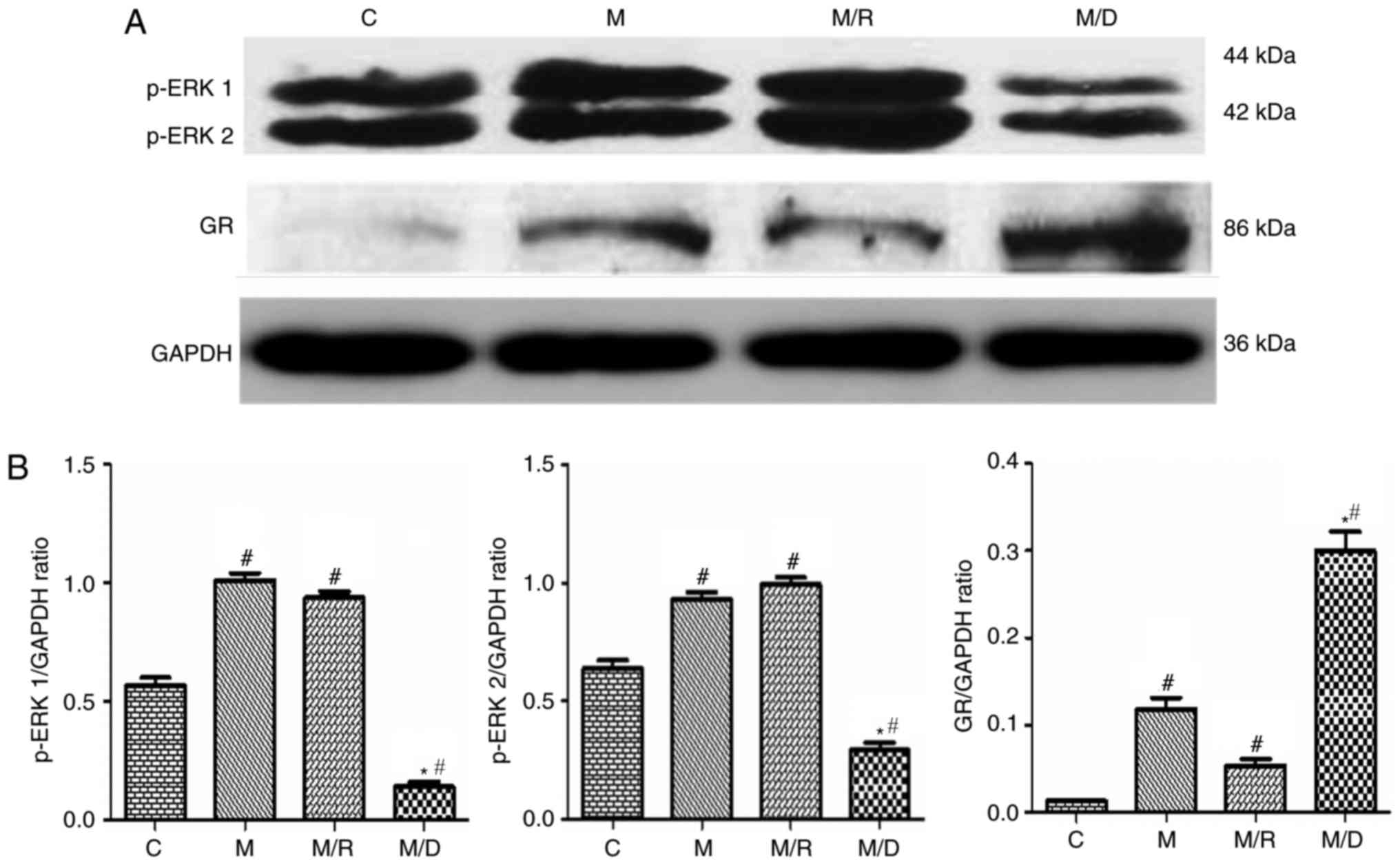
|
1
|
Bailey CP, Llorente J, Gabra BH, Smith FL,
Dewey WL, Kelly E and Henderson G: Role of protein kinase C and
mu-opioid receptor (MOPr) desensitization in tolerance to morphine
in rat locus coeruleus neurons. Eur J Neurosci. 29:307–318. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Chu J, Zheng H, Loh HH and Law PY:
Morphine-induced mu-opioid receptor rapid desensitization is
independent of receptor phosphorylation and beta-arrestins. Cell
Signal. 20:1616–1624. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Sim-Selley LJ, Scoggins KL, Cassidy MP,
Smith LA, Dewey WL, Smith FL and Selley DE: Region-dependent
attenuation of mu opioid receptor-mediated G-protein activation in
mouse CNS as a function of morphine tolerance. Br J Pharmacol.
151:1324–1333. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bull FA, Baptista-Hon DT, Sneddon C,
Wright L, Walwyn W and Hales TG: Src kinase inhibition attenuates
morphine tolerance without affecting reinforcement or psychomotor
stimulation. Anesthesiology. 127:878–889. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Wilson-Poe AR, Jeong HJ and Vaughan CW:
Chronic morphine reduces the readily releasable pool of GABA, a
presynaptic mechanism of opioid tolerance. J Physiol.
595:6541–6555. 2017. View
Article : Google Scholar : PubMed/NCBI
|
|
6
|
Lim G, Wang S, Zeng Q, Sung B and Mao J:
Spinal glucocorticoid receptors contribute to the development of
morphine tolerance in rats. Anesthesiology. 102:832–837. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lim G, Wang S, Zeng Q, Sung B and Mao J:
Evidence for a long-term influence on morphine tolerance after
previous morphine exposure: Role of neuronal glucocorticoid
receptors. Pain. 114:81–92. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Narita M, Ioka M, Suzuki M, Narita M and
Suzuki T: Effect of repeated administration of morphine on the
activity of extracellular signal regulated kinase in the mouse
brain. Neurosci Lett. 324:97–100. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Robbins TW and Everitt BJ: Drug addiction:
Bad habits add up. Nature. 398:567–570. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Williams JT, Christie MJ and Manzoni O:
Cellular and synaptic adaptations mediating opioid dependence.
Physiol Rev. 81:299–343. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Deng XT, Han Y, Liu WT and Song XJ: B
vitamins potentiate acute morphine antinociception and attenuate
the development of tolerance to chronic morphine in mice. Pain Med.
18:1961–1974. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Pan Y, Sun X, Jiang L, Hu L, Kong H, Han
Y, Qian C, Song C, Qian Y and Liu W: Metformin reduces morphine
tolerance by inhibiting microglial-mediated neuroinflammation. J
Neuroinflammation. 13:2942016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Bao Y, Gao Y, Yang L, Kong X, Yu J, Hou W
and Hua B: The mechanism of mu-opioid receptor (MOR)-TRPV1
crosstalk in TRPV1 activation involves morphine anti-nociception,
tolerance and dependence. Channels (Austin). 9:235–243. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Jiang C, Xu L, Chen L, Han Y, Tang J, Yang
Y, Zhang G and Liu W: Selective suppression of microglial
activation by paeoniflorin attenuates morphine tolerance. Eur J
Pain. 19:908–919. 2015. View
Article : Google Scholar : PubMed/NCBI
|
|
15
|
Gutstein HB, Rubie EA, Mansour A, Akil H
and Woodgett JR: Opioid effects on mitogen-activated protein kinase
signaling cascades. Anesthesiology. 87:1118–1126. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Fukuda K, Kato S, Morikawa H, Shoda T and
Mori K: Functional coupling of the delta-, mu-, and kappa-opioid
receptors to mitogen-activated protein kinase and arachidonate
release in Chinese hamster ovary cells. J Neurochem. 67:1309–1316.
1996. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Cameron SA and Dutia MB: Lesion-induced
plasticity in rat vestibular nucleus neurones dependent on
glucocorticoid receptor activation. J Physiol. 518:151–158. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Garabedian MJ, Harris CA and Jeanneteau F:
Glucocorticoid receptor action in metabolic and neuronal function.
F1000Res. 6:12082017. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Takahashi M, Sugimachi K and Kaneto H:
Role of adrenal glucocorticoids in the blockade of the development
of analgesic tolerance to morphine by footshock stress exposure in
mice. Jpn J Pharmacol. 51:329–336. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Clark AR and Lasa M: Crosstalk between
glucocorticoids and mitogen-activated protein kinase signalling
pathways. Curr Opin Pharmacol. 3:404–411. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wang S, Lim G, Zeng Q, Sung B, Ai Y, Guo
G, Yang L and Mao J: Expression of central glucocorticoid receptors
after peripheral nerve injury contributes to neuropathic pain
behaviors in rats. J Neurosci. 24:8595–8605. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Ma W and Quirion R: The ERK/MAPK pathway,
as a target for the treatment of neuropathic pain. Expert Opin Ther
Targets. 9:699–713. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Mayer DJ, Mao J, Holt J and Price DD:
Cellular mechanisms of neuropathic pain, morphine tolerance, and
their interactions. Proc Natl Acad Sci USA. 96:7731–7736. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
LoPachin RM, Rudy TA and Yaksh TL: An
improved method for chronic catheterization of the rat spinal
subarachnoid space. Physiol Behav. 27:559–561. 1981. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Mao J: Opioid-induced abnormal pain
sensitivity: Implications in clinical opioid therapy. Pain.
100:213–217. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Cui Y, Chen Y, Zhi JL, Guo RX, Feng JQ and
Chen PX: Activation of p38 mitogen-activated protein kinase in
spinal microglia mediates morphine antinociceptive tolerance. Brain
Res. 1069:235–243. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Mao J, Price DD and Mayer DJ: Experimental
mononeuropathy reduces the antinociceptive effects of morphine:
Implications for common intracellular mechanisms involved in
morphine tolerance and neuropathic pain. Pain. 61:353–364. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Gardell LR, Wang R, Burgess SE, Ossipov
MH, Vanderah TW, Malan TP Jr, Lai J and Porreca F: Sustained
morphine exposure induces a spinal dynorphin-dependent enhancement
of excitatory transmitter release from primary afferent fibers. J
Neurosci. 22:6747–6755. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Trang T, Quirion R and Jhamandas K: The
spinal basis of opioid tolerance and physical dependence:
Involvement of calcitonin gene-related peptide, substance P, and
arachidonic acid-derived metabolites. Peptides. 26:1346–1355. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Guo D and Hu J: Spinal presynaptic
inhibition in pain control. Neuroscience. 283:95–106. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
DeLander GE, Portoghese PS and Takemori
AE: Role of spinal mu opioid receptors in the development of
morphine tolerance and dependence. J Pharmacol Exp Ther. 231:91–96.
1984.PubMed/NCBI
|
|
32
|
Pitcher GM, Yashpal K, Coderre TJ and
Henry JL: Mechanisms underlying antinociception provoked by
heterosegmental noxious stimulation in the rat tail-flick test.
Neuroscience. 65:273–281. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Cintra A, Molander C and Fuxe K:
Colocalization of Fos- and glucocorticoid
receptor-immunoreactivities is present only in a very restricted
population of dorsal horn neurons of the rat spinal cord after
nociceptive stimulation. Brain Res. 632:334–338. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
De Nicola AF, Moses DF, Gonzalez S and
Orti E: Adrenocorticoid action in the spinal cord: Some unique
molecular properties of glucocorticoid receptors. Cell Mol
Neurobiol. 9:179–192. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Capasso A, Di Giannuario A, Loizzo A,
Pieretti S and Sorrentino L: Central interaction of dexamethasone
and RU-38486 on morphine antinociception in mice. Life Sci.
51:PL139–PL143. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Wang Z, Ma W, Chabot JG and Quirion R:
Cell-type specific activation of p38 and ERK mediates calcitonin
gene-related peptide involvement in tolerance to morphine-induced
analgesia. FASEB J. 23:2576–2586. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Croxtall JD, Choudhury Q and Flower RJ:
Glucocorticoids act within minutes to inhibit recruitment of
signalling factors to activated EGF receptors through a
receptor-dependent, transcription-independent mechanism. Br J
Pharmacol. 130:289–298. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Mazzucchelli C, Vantaggiato C, Ciamei A,
Fasano S, Pakhotin P, Krezel W, Welzl H, Wolfer DP, Pages G,
Valverde O, et al: Knockout of ERK1 MAP kinase enhances synaptic
plasticity in the striatum and facilitates striatal-mediated
learning and memory. Neuron. 34:807–820. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Tai YH, Wang YH, Wang JJ, Tao PL, Tung CS
and Wong CS: Amitriptyline suppresses neuroinflammation and
up-regulates glutamate transporters in morphine-tolerant rats.
Pain. 124:77–86. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Wen ZH, Wu GJ, Chang YC, Wang JJ and Wong
CS: Dexamethasone modulates the development of morphine tolerance
and expression of glutamate transporters in rats. Neuroscience.
133:807–817. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Wong CS, Hsu MM, Chou R, Chou YY and Tung
CS: Intrathecal cyclooxygenase inhibitor administration attenuates
morphine antinociceptive tolerance in rats. Br J Anaesth.
85:747–751. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Chen Y, Yu YH, Zhai ML, Wang Z and Wang
GL: Participation of glucocorticoid receptors in morphine tolerance
development through the signal pathway of extracellular
signal-regulated kinase. Zhonghua Yi Xue Za Zhi. 91:1272–1275.
2011.(In Chinese). PubMed/NCBI
|
|
43
|
Martel MA, Wyllie DJ and Hardingham GE: In
developing hippocampal neurons, NR2B-containing
N-methyl-D-aspartate receptors (NMDARs) can mediate signaling to
neuronal survival and synaptic potentiation, as well as neuronal
death. Neuroscience. 158:334–343. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Urakubo H, Honda M, Froemke RC and Kuroda
S: Requirement of an allosteric kinetics of NMDA receptors for
spike timing-dependent plasticity. J Neurosci. 28:3310–3323. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Ota KT, Pierre VJ, Ploski JE, Queen K and
Schafe GE: The NO-cGMP-PKG signaling pathway regulates synaptic
plasticity and fear memory consolidation in the lateral amygdala
via activation of ERK/MAP kinase. Learn Mem. 15:792–805. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Toyoda H, Zhao MG, Xu H, Wu LJ, Ren M and
Zhuo M: Requirement of extracellular signal-regulated
kinase/mitogen-activated protein kinase for long-term potentiation
in adult mouse anterior cingulate cortex. Mol Pain. 3:362007.
View Article : Google Scholar : PubMed/NCBI
|