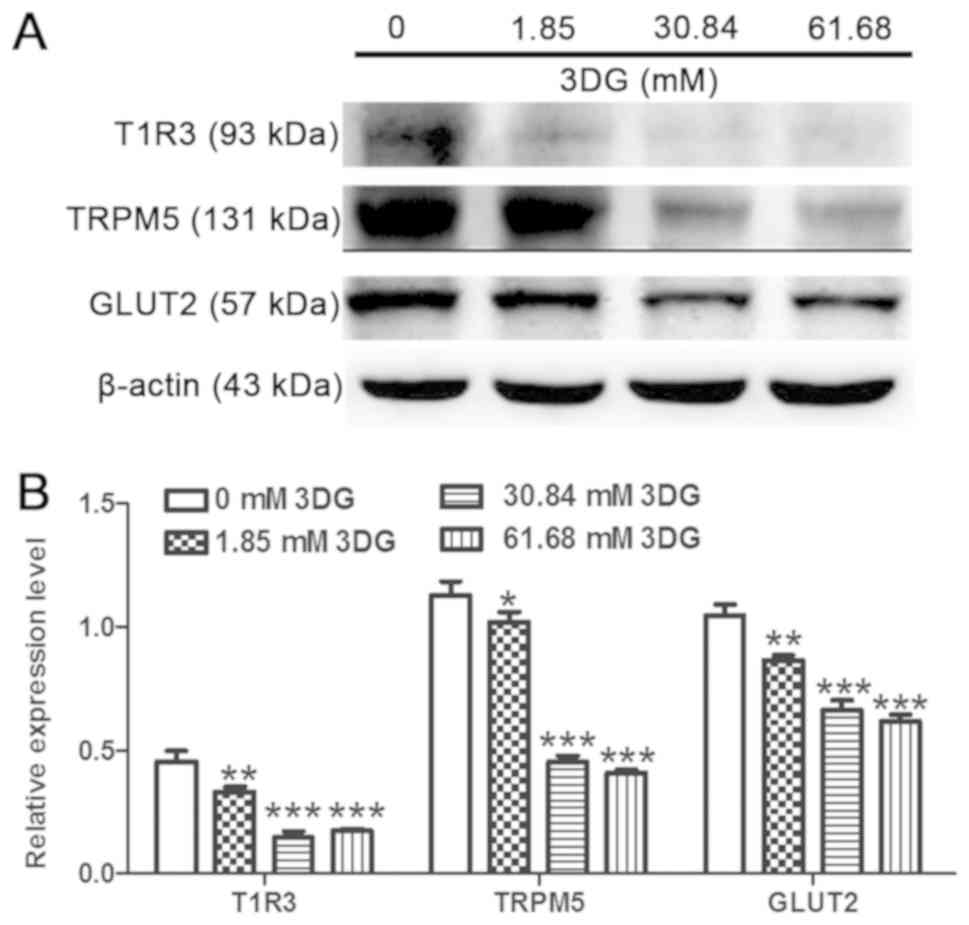
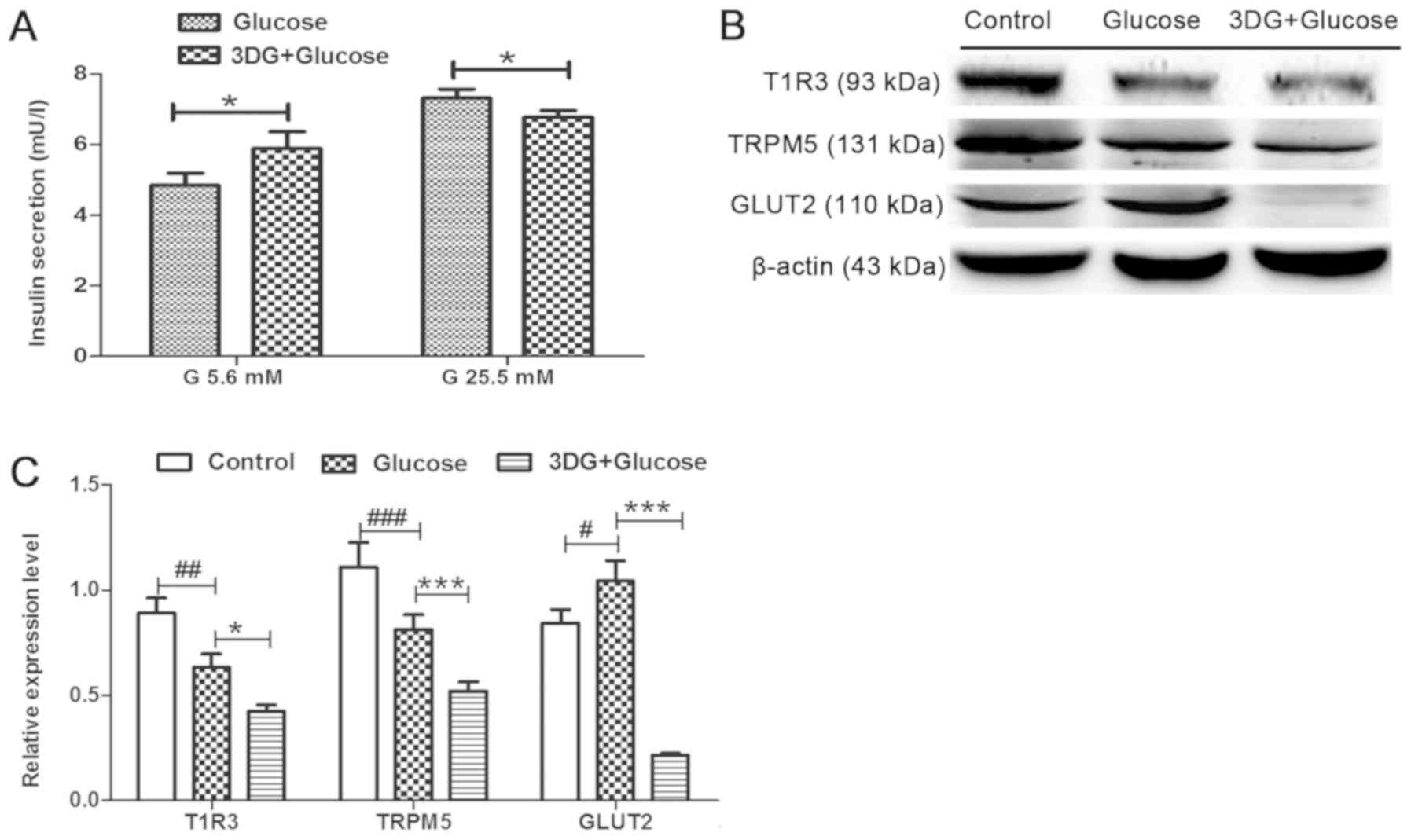
|
1
|
Schuit FC, Huypens P, Heimberg H and
Pipeleers DG: Glucose sensing in pancreatic beta-cells: A model for
the study of other glucose-regulated cells in gut, pancreas, and
hypothalamus. Diabetes. 50:1–11. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Kojima I, Medina J and Nakagawa Y: Role of
the glucose-sensing receptor in insulin secretion. Diabetes Obes
Metab. 1 (Suppl 19):S54–S62. 2017. View Article : Google Scholar
|
|
3
|
Nelson G, Hoon MA, Chandrashekar J, Zhang
Y, Ryba NJ and Zuker CS: Mammalian sweet taste receptors. Cell.
106:381–390. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Margolskee RF: Molecular mechanisms of
bitter and sweet taste transduction. J Biol Chem. 277:1–4. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Depoortere I: Taste receptors of the gut:
Emerging roles in health and disease. Gut. 63:179–190. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kyriazis GA, Soundarapandian MM and
Tyrberg B: Sweet taste receptor signaling in beta cells mediates
fructose-induced potentiation of glucose-stimulated insulin
secretion. Proc Natl Acad Sci USA. 109:E524–E532. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Nakagawa Y, Nagasawa M, Yamada S, Hara A,
Mogami H, Nikolaev VO, Lohse MJ, Shigemura N, Ninomiya Y and Kojima
I: Sweet taste receptor expressed in pancreatic beta-cells
activates the calcium and cyclic AMP signaling systems and
stimulates insulin secretion. PLoS One. 4:e51062009. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Nakagawa Y, Nagasawa M, Mogami H, Lohse M,
Ninomiya Y and Kojima I: Multimodal function of the sweet taste
receptor expressed in pancreatic β-cells: Generation of diverse
patterns of intracellular signals by sweet agonists. Endocr J.
60:1191–1206. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Nakagawa Y, Ohtsu Y, Nagasawa M, Shibata H
and Kojima I: Glucose promotes its own metabolism by acting on the
cell-surface glucose-sensing receptor T1R3. Endocr J. 61:119–131.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Niwa T: 3-Deoxyglucosone: Metabolism,
analysis, biological activity, and clinical implication. J
Chromatogr B Biomed Sci Appl. 731:23–36. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Degen J, Beyer H, Heymann B, Hellwig M and
Henle T: Dietary influence on urinary excretion of 3-deoxyglucosone
and its metabolite 3-deoxyfructose. J Agric Food Chem.
62:2449–2456. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Brings S, Fleming T, Freichel M,
Muckenthaler MU, Herzig S and Nawroth PP: Dicarbonyls and advanced
glycation end-products in the development of diabetic complications
and targets for intervention. Int J Mol Sci. 18:E9842017.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Lal S, Kappler F, Walker M, Orchard TJ,
Beisswenger PJ, Szwergold BS and Brown TR: Quantitation of
3-deoxyglucosone levels in human plasma. Arch Biochem Biophys.
342:254–260. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Hamada Y, Nakamura J, Fujisawa H, Yago H,
Nakashima E, Koh N and Hotta N: Effects of glycemic control on
plasma 3-deoxyglucosone levels in NIDDM patients. Diabetes Care.
20:1466–1469. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Jiang G, Zhang L, Ji Q, Wang F, Xu H,
Huang F and Wang C: Accumulation of plasma 3-deoxyglucosone
impaired glucose regulation in Chinese seniors: Implication for
senile diabetes? Diabetes Metab Syndr. 6:140–145. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhang L, Song X, Zhou L, Liang G, Xu H,
Wang F, Huang F and Jiang G: Accumulation of intestinal tissue
3-deoxyglucosone attenuated GLP-1 secretion and its insulinotropic
effect in rats. Diabetol Metab Syndr. 8:782016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wang F, Zhou L, Song X, Liang G, Xu H,
Zhang L and Jiang G: Acute reduction of incretin effect and glucose
intolerance in rats by single intragastric administration of
3-deoxyglucosone. Exp Clin Endocrinol Diabetes. 125:4–11.
2017.PubMed/NCBI
|
|
18
|
Zhang L, Zhou L, Song X, Liang G, Xu Z,
Wang F, Huang F and Jiang G: Involvement of exogenous
3-deoxyglucosone in β-cell dysfunction induces impaired glucose
regulation. Mol Med Rep. 16:2976–2984. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Liang G, Song X, Xu H, Wang F, Zhang L,
Zhou L and Jiang G: 3-Deoxyglucosone induced acute glucose
intolerance in sprague-dawley rats: Involvement of insulin
resistance and impaired β-cell function. Exp Clin Endocrinol
Diabetes. 124:431–436. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Maessen DE, Hanssen NM, Scheijen JL, van
der Kallen CJ, van Greevenbroek MM, Stehouwer CD and Schalkwijk CG:
Post-glucose load plasma α-dicarbonyl concentrations are increased
in individuals with impaired glucose metabolism and type 2
diabetes: The CODAM study. Diabetes Care. 38:913–920. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Feng R, Qian C, Liu Q, Jin Y, Liu L, Li S,
Liao Y, Zhou H, Liu W, Rayner CK and Ma J: Expression of sweet
taste receptor and gut hormone secretion in modeled type 2 diabete.
Gen Comp Endocrinol. 252:142–149. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Medina A, Nakagawa Y, Ma J, Li L, Hamano
K, Akimoto T, Ninomiya Y and Kojima I: Expression of the
glucose-sensing receptor T1R3 in pancreatic islet: Changes in the
expression levels in various nutritional and metabolic states.
Endocr J. 61:797–805. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kyriazis GA, Smith KR, Tyrberg B, Hussain
T and Pratley RE: Sweet taste receptors regulate basal insulin
secretion and contribute to compensatory insulin hypersecretion
during the development of diabetes in male mice. Endocrinology.
155:2112–2121. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Wang F, Song X, Zhou L, Liang G, Huang F,
Jiang G and Zhang L: The downregulation of sweet taste receptor
signaling in enteroendocrine L-cells mediates
3-deoxyglucosone-induced attenuation of high glucose-stimulated
GLP-1 secretion. Arch Physiol Biochem. 124:430–435. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kato H, van Chuyen N, Shinoda T, Sekiya F
and Hayase F: Metabolism of 3-deoxyglucosone, an intermediate
compound in the maillard reaction, administered orally or
intravenously to rats. Biochim Biophys Acta. 1035:71–76. 1990.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Liang G, Wang F, Song X, Zhang L, Qian Z
and Jiang G: 3-Deoxyglucosone induces insulin resistance by
impairing insulin signaling in HepG2 cells. Mol Med Rep.
13:4506–4512. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Chlebus M, Guillen J and Prins JB:
Directive 2010/63/EU: Facilitating full and correct implementation.
Lab Anim. 50:1512016. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Yamada H, Miyata S, Igaki N, Yatabe H,
Miyauchi Y, Ohara T, Sakai M, Shoda H, Oimomi M and Kasuga M:
Increase in 3-deoxyglucosone levels in diabetic rat plasma.
Specific in vivo determination of intermediate in advanced Maillard
reaction. J Biol Chem. 269:20275–20280. 1994.PubMed/NCBI
|
|
29
|
Asfari M, Janjic D, Meda P, Li G, Halban
PA and Wollheim CB: Establishment of 2-mercaptoethanol-dependent
differentiated insulin-secreting cell lines. Endocrinology.
130:167–178. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kang MY, Oh TJ and Cho YM: Glucagon-like
peptide-1 increases mitochondrial biogenesis and function in INS-1
rat insulinoma cells. Endocrinol Metab (Seoul). 30:216–220. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Bo J, Xie S, Guo Y, Zhang C, Guan Y, Li C,
Lu J and Meng QH: Methylglyoxal impairs insulin secretion of
pancreatic β-Cells through increased production of ROS and
mitochondrial dysfunction mediated by upregulation of UCP2 and
MAPKs. J Diabetes Res. 2016:20298542016. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Hamano K, Nakagawa Y, Ohtsu Y, Li L,
Medina J, Tanaka Y, Masuda K, Komatsu M and Kojima I: Lactisole
inhibits the glucose-sensing receptor T1R3 expressed in mouse
pancreatic β-cells. J Endocrinol. 226:57–66. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Zhou L, Huang W, Xu Y, Gao C, Zhang T, Guo
M, Liu Y, Ding J, Qin L, Xu Z, et al: Sweet taste receptors
mediated ROS-NLRP3 inflammasome signaling activation: Implications
for diabetic nephropathy. J Diabetes Res. 2018:70782142018.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Elmhiri G, Barella LF, Vieau D, Camous S,
Mathias PC and Abdennebi-Najar L: Acute exposure to a precursor of
advanced glycation end products induces a dual effect on the rat
pancreatic islet function. Int J Endocrinol. 2014:3782842014.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Mace OJ, Affleck J, Patel N and Kellett
GL: Sweet taste receptors in rat small intestine stimulate glucose
absorption through apical GLUT2. J Physiol. 582:379–392. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Ashraf JM, Shahab U, Tabrez S, Lee EJ,
Choi I, Aslam Yusuf M and Ahmad S: DNA glycation from
3-deoxyglucosone leads to the formation of AGEs: Potential role in
cancer auto-antibodies. Cell Biochem Biophys. 74:67–77. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Hecker M and Wagner AH: Role of protein
carbonylation in diabetes. J Inherit Metab Dis. 41:29–38. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Aldini G, Dalle-Donne I, Facino RM,
Milzani A and Carini M: Intervention strategies to inhibit protein
carbonylation by lipoxidation-derived reactive carbonyls. Med Res
Rev. 27:817–868. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Thorens B: GLUT2, glucose sensing and
glucose homeostasis. Diabetologia. 58:221–232. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Song Z, Wang H, Zhu L, Han M, Gao Y, Du Y
and Wen Y: Curcumin improves high glucose-induced INS-1 cell
insulin resistance via activation of insulinsignaling. Food Funct.
6:461–469. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Guillam MT, Hümmler E, Schaerer E, Yeh JI,
Birnbaum MJ, Beermann F, Schmidt A, Dériaz N and Thorens B: Early
diabetes and abnormal postnatal pancreatic islet development in
mice lacking Glut-2. Nat Genet. 17:327–330. 1997. View Article : Google Scholar : PubMed/NCBI
|