|
1
|
Sakao S: Chronic obstructive pulmonary
disease and the early stage of cor pulmonale: A perspective in
treatment with pulmonary arterial hypertension-approved drugs.
Respir Investig. 57:325–329. 2019. View Article : Google Scholar
|
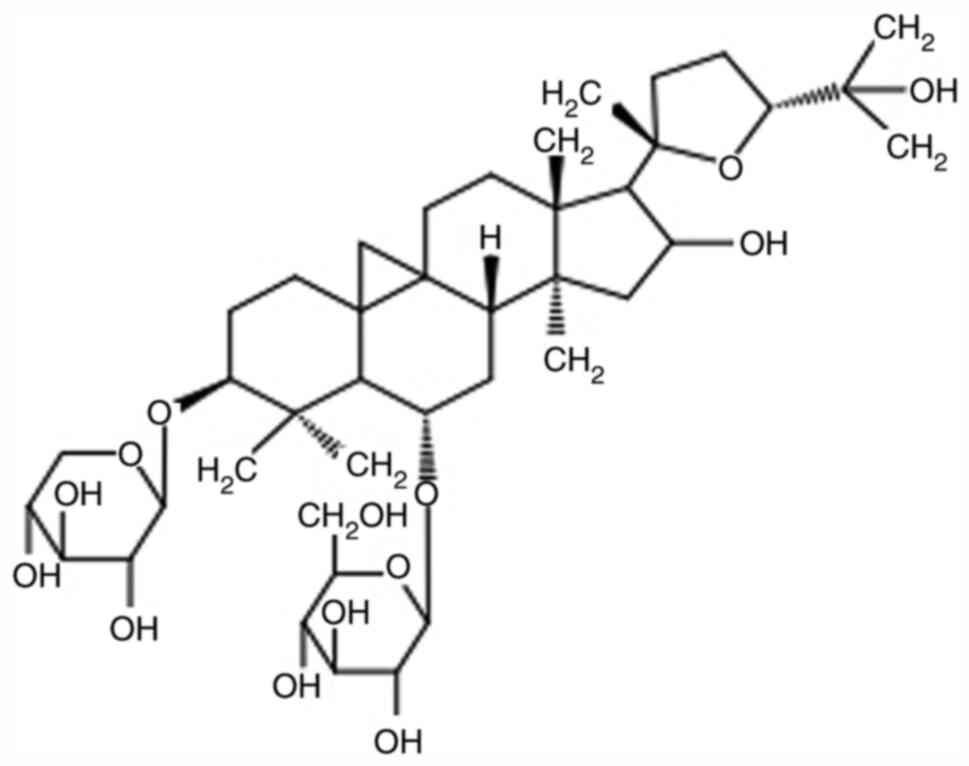
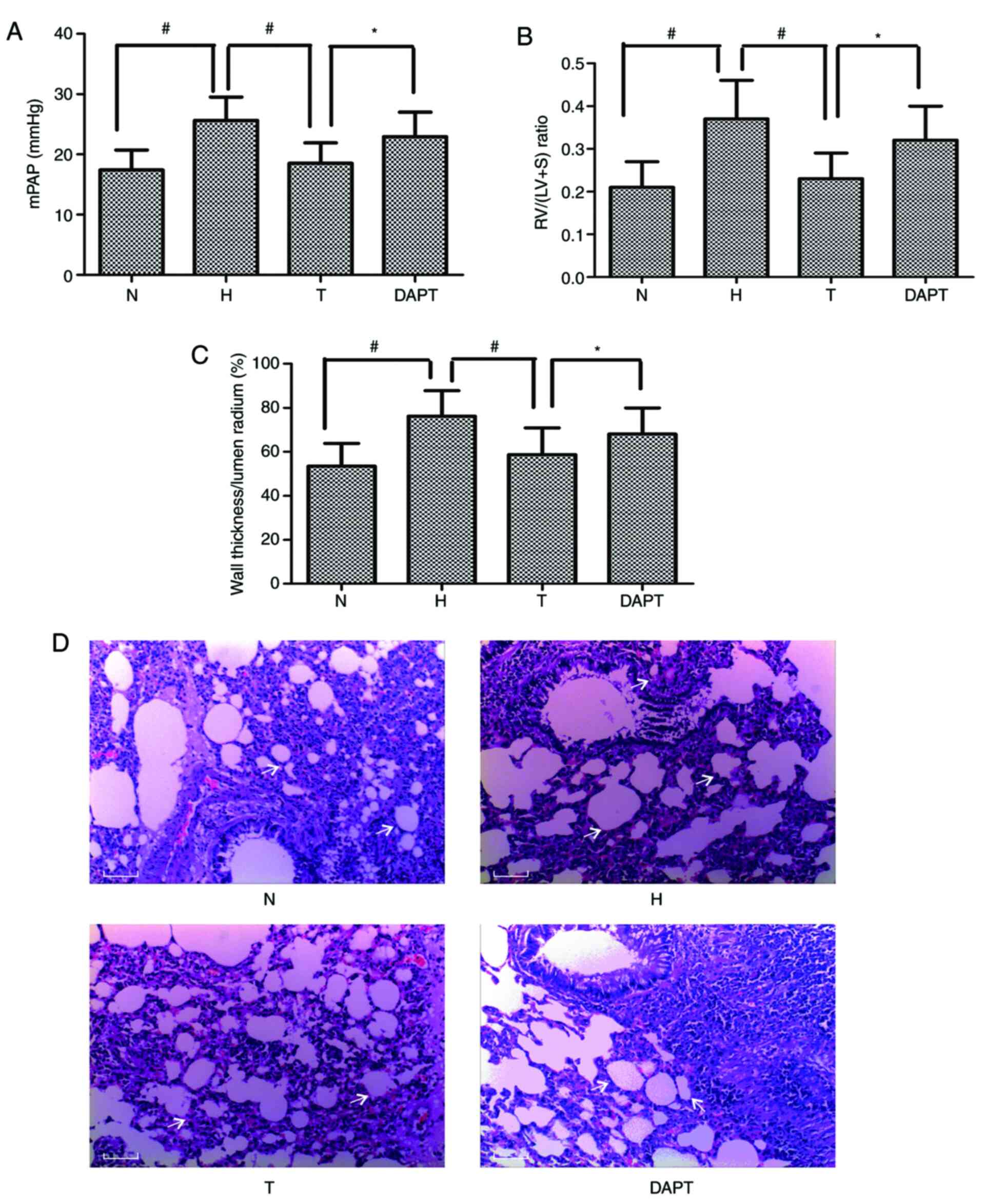
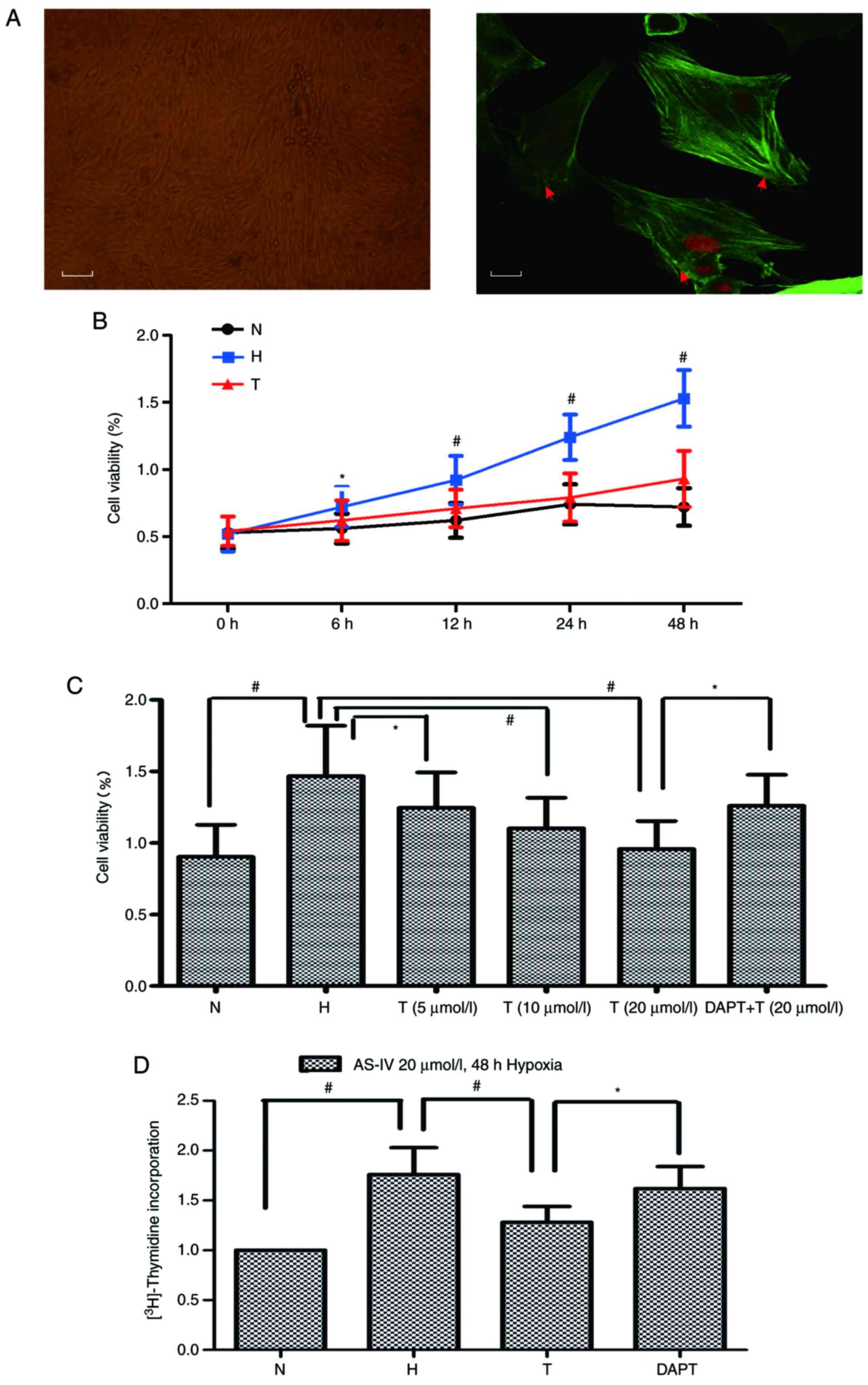
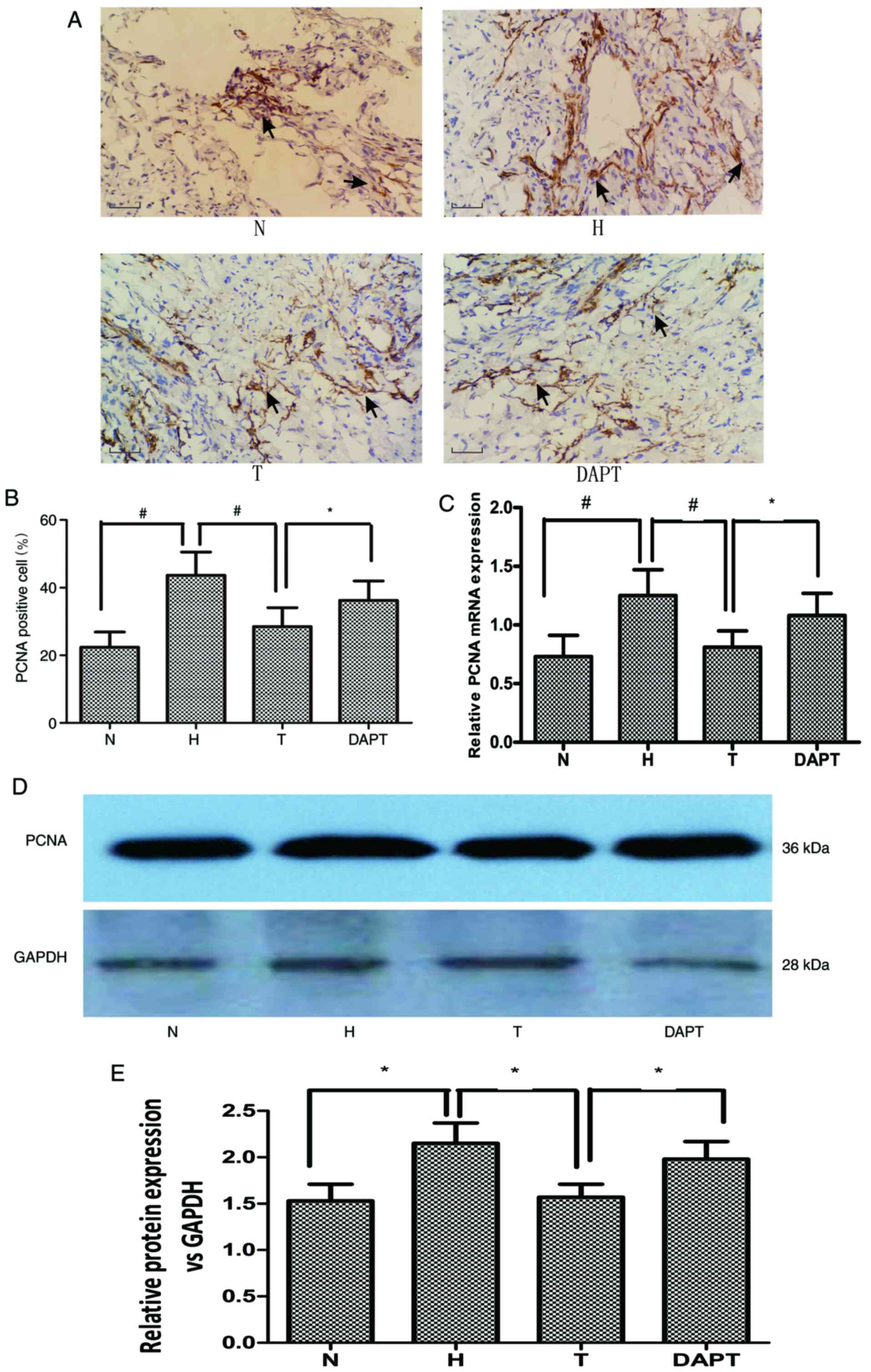
|
2
|
Samareh Fekri M, Torabi M, Azizi Shoul S
and Mirzaee M: Prevalence and predictors associated with severe
pulmonary hypertension in COPD. Am J Emerg Med. 36:277–280. 2018.
View Article : Google Scholar
|
|
3
|
Rowan SC, Keane MP, Gaine S and McLoughlin
P: Hypoxic pulmonary hypertension in chronic lung diseases: Novel
vasoconstrictor pathways. Lancet Respir Med. 4:225–236. 2016.
View Article : Google Scholar
|
|
4
|
Bunel V, Guyard A, Dauriat G, Danel C,
Montani D, Gauvain C, Thabut G, Humbert M, Castier Y, Dorfmüller P
and Mal H: Pulmonary arterial histologic lesions in patients with
COPD with severe pulmonary hypertension. Chest. 156:33–44. 2019.
View Article : Google Scholar
|
|
5
|
Sauler M, Fares WH and Trow TK: Standard
nonspecific therapies in the management of pulmonary arterial
hypertension. Clin Chest Med. 34:799–810. 2013. View Article : Google Scholar
|
|
6
|
Shimoda LA, Yun X and Sikka G: Revisiting
the role of hypoxia-inducible factors in pulmonary hypertension.
Curr Opin Physiol. 7:33–40. 2019. View Article : Google Scholar
|
|
7
|
Liu S, Shergis J, Chen X, Yu X, Guo X,
Zhang AL, Lu C and Xue CC: Chinese herbal medicine (weijing
decoction) combined with pharmacotherapy for the treatment of acute
exacerbations of chronic obstructive pulmonary disease. Evid Based
Complement Alternat Med. 2014:2570122014. View Article : Google Scholar
|
|
8
|
Ren S, Zhang H, Mu Y, Sun M and Liu P:
Pharmacological effects of Astragaloside IV: A literature review. J
Tradit Chin Med. 33:413–416. 2013. View Article : Google Scholar
|
|
9
|
Yuan X, Sun S, Wang S and Sun Y: Effects
of astragaloside IV on IFN-gamma level and prolonged airway
dysfunction in a murine model of chronic asthma. Planta Med.
77:328–333. 2011. View Article : Google Scholar
|
|
10
|
Leng B, Tang F, Lu M, Zhang Z, Wang H and
Zhang Y: Astragaloside IV improves vascular endothelial dysfunction
by inhibiting the TLR4/NF-κB signaling pathway. Life Sci.
209:111–121. 2018. View Article : Google Scholar
|
|
11
|
Chen Z, Cai Y, Zhang W, Liu X and Liu S:
Astragaloside IV inhibits platelet-derived growth
factor-BB-stimulated proliferation and migration of vascular smooth
muscle cells via the inhibition of p38 MAPK signaling. Exp Ther
Med. 8:1253–1258. 2014. View Article : Google Scholar
|
|
12
|
Song Z, Wei D, Chen Y, Chen L, Bian Y,
Shen Y, Chen J and Pan Y: Association of astragaloside IV-inhibited
autophagy and mineralization in vascular smooth muscle cells with
lncRNA H19 and DUSP5-mediated ERK signaling. Toxicol Appl
Pharmacol. 364:45–54. 2019. View Article : Google Scholar
|
|
13
|
Spiekerkoetter E, Goncharova EA,
Guignabert C, Stenmark K, Kwapiszewska G, Rabinovitch M, Voelkel N,
Bogaard HJ, Graham B, Pullamsetti SS and Kuebler WM: Hot topics in
the mechanisms of pulmonary arterial hypertension disease:
Cancer-like pathobiology, the role of the adventitia, systemic
involvement, and right ventricular failure. Pulm Circ.
9:20458940198897752019. View Article : Google Scholar
|
|
14
|
Wang X, Xiao D, Ma C, Zhang L, Duan Q,
Zheng X, Mao M, Zhu D and Li Q: The effect of honokiol on pulmonary
artery endothelium cell autophagy mediated by cyclophilin A in
hypoxic pulmonary arterial hypertension. J Pharmacol Sci.
139:158–165. 2019. View Article : Google Scholar
|
|
15
|
Wang S, Cao W, Gao S, Nie X, Zheng X, Xing
Y, Chen Y, Bao H and Zhu D: TUG1 regulates pulmonary arterial
smooth muscle cell proliferation in pulmonary arterial
hypertension. Can J Cardiol. 35:1534–1545. 2019. View Article : Google Scholar
|
|
16
|
Borggrefe T, Lauth M, Zwijsen A,
Huylebroeck D, Oswald F and Giaimo BD: The Notch intracellular
domain integrates signals from Wnt, Hedgehog, TGFβ/BMP and hypoxia
pathways. Biochim Biophys Acta. 1863:303–313. 2016. View Article : Google Scholar
|
|
17
|
Bigas A and Espinosa L: The multiple
usages of Notch signaling in development, cell differentiation and
cancer. Curr Opin Cell Biol. 55:1–7. 2018. View Article : Google Scholar
|
|
18
|
Wang Y, Dai S, Cheng X, Prado E, Yan L, Hu
J, He Q, Lv Y, Lv Y and Du L: Notch3 signaling activation in smooth
muscle cells promotes extrauterine growth restriction-induced
pulmonary hypertension. Nutr Metab Cardiovasc Dis. 29:639–651.
2019. View Article : Google Scholar
|
|
19
|
Harrison OJ, Visan AC, Moorjani N, Modi A,
Salhiyyah K, Torrens C, Ohri S and Cagampang FR: Defective NOTCH
signaling drives increased vascular smooth muscle cell apoptosis
and contractile differentiation in bicuspid aortic valve
aortopathy: A review of the evidence and future directions. Trends
Cardiovasc Med. 29:61–68. 2019. View Article : Google Scholar
|
|
20
|
Yu YR, Mao L, Piantadosi CA and Gunn MD:
CCR2 deficiency, dysregulation of Notch signaling, and spontaneous
pulmonary arterial hypertension. Am J Respir Cell Mol Biol.
48:647–654. 2013. View Article : Google Scholar
|
|
21
|
Song Y, Zhang Y, Jiang H, Zhu Y, Liu L,
Feng W, Yang L, Wang Y and Li M: Activation of Notch3 promotes
pulmonary arterial smooth muscle cells proliferation via
Hes1/p27Kip1 signaling pathway. FEBS Open Bio. 5:656–660. 2015.
View Article : Google Scholar
|
|
22
|
Chen X, Yao JM, Fang X, Zhang C, Yang YS,
Hu CP, Chen Q and Zhong GW: Hypoxia promotes pulmonary vascular
remodeling via HIF-1α to regulate mitochondrial dynamics. J Geriatr
Cardiol. 16:855–871. 2019.
|
|
23
|
Yu X, Li T, Liu X, Yu H, Hao Z, Chen Y,
Zhang C, Liu Y, Li Q, Mao M and Zhu D: Modulation of pulmonary
vascular remodeling in hypoxia: Role of 15-LOX-2/15-HETE-MAPKs
pathway. Cell Physiol Biochem. 35:2079–2097. 2015. View Article : Google Scholar
|
|
24
|
Crnkovic S, Marsh LM, El Agha E,
Voswinckel R, Ghanim B, Klepetko W, Stacher-Priehse E, Olschewski
H, Bloch W, Bellusci S, et al: Resident cell lineages are preserved
in pulmonary vascular remodeling. J Pathol. 244:485–498. 2018.
View Article : Google Scholar
|
|
25
|
Yan J, Chen R, Liu P and Gu Y:
Docosahexaenoic acid inhibits development of hypoxic pulmonary
hypertension: In vitro and in vivo studies. Int J Cardiol.
168:4111–4116. 2013. View Article : Google Scholar
|
|
26
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
27
|
Roels S, Tilmant K, Van Daele A, Van Marck
E and Ducatelle R: Proliferation, DNA ploidy, p53 overexpression
and nuclear DNA fragmentation in six equine melanocytic tumours. J
Vet Med A Physiol Pathol Clin Med. 47:439–48. 2000. View Article : Google Scholar
|
|
28
|
Heidebrecht F, Heidebrecht A, Schulz I,
Behrens SE and Bader A: Improved semiquantitative western blot
technique with increased quantification range. J Immunol Methods.
345:40–48. 2009. View Article : Google Scholar
|
|
29
|
Yang C, Mo Y, Xu E, Wen H, Wei R, Li S,
Zheng J, Li W, Le B, Chen Y, et al: Astragaloside IV ameliorates
motor deficits and dopaminergic neuron degeneration via inhibiting
neuroinflammation and oxidative stress in a Parkinson's disease
mouse model. Int Immunopharmacol. 75:1056512019. View Article : Google Scholar
|
|
30
|
Du J, Liu J, Zhen J, Yang ST, Zheng EL and
Leng JY: Astragaloside IV protects cardiomyocytes from
hypoxia-induced injury by down-regulation of lncRNA GAS5. Biomed
Pharmacother. 116:1090282019. View Article : Google Scholar
|
|
31
|
Liu ZH, Liu HB and Wang J: Astragaloside
IV protects against the pathological cardiac hypertrophy in mice.
Biomed Pharmacother. 97:1468–1478. 2018. View Article : Google Scholar
|
|
32
|
Yao Y, Li H, Da X, He Z, Tang B, Li Y, Hu
C, Xu C, Chen Q and Wang QK: SUMOylation of Vps34 by SUMO1 promotes
phenotypic switching of vascular smooth muscle cells by activating
autophagy in pulmonary arterial hypertension. Pulm Pharmacol Ther.
55:38–49. 2019. View Article : Google Scholar
|
|
33
|
Zhang DQ, Li JS, Zhang YM, Gao F and Dai
RZ: Astragaloside IV inhibits Angiotensin II-stimulated
proliferation of rat vascular smooth muscle cells via the
regulation of CDK2 activity. Life Sci. 105–109. 2018. View Article : Google Scholar
|
|
34
|
Gu L, Tao X, Xu Y, Han X, Qi Y, Xu L, Yin
L and Peng J: Dioscin alleviates BDL- and DMN-induced hepatic
fibrosis via Sirt1/Nrf2-mediated inhibition of p38MAPK pathway.
Toxicol Appl Pharmacol. 2921:19–29. 2016. View Article : Google Scholar
|
|
35
|
Liu D, Chen L, Zhao H, Vaziri ND, Ma SC
and Zhao YY: Small molecules from natural products targeting the
Wnt/β-catenin pathway as a therapeutic strategy. Biomed
Pharmacother. 117:1089902019. View Article : Google Scholar
|
|
36
|
Wang SG, Xu Y, Chen JD, Yang CH and Chen
XH: Astragaloside IV stimulates angiogenesis and increases nitric
oxide accumulation via JAK2/STAT3 and ERK1/2 pathway. Molecules.
18:12809–12819. 2013. View Article : Google Scholar
|
|
37
|
Kostina A, Semenova D, Kostina D, Uspensky
V, Kostareva A and Malashicheva A: Human aortic endothelial cells
have osteogenic Notch-dependent properties in co-culture with
aortic smooth muscle cells. Biochem Biophys Res Commun.
514:462–468. 2019. View Article : Google Scholar
|
|
38
|
Yamamura H, Yamamura A, Ko EA, Pohl NM,
Smith KA, Zeifman A, Powell FL, Thistlethwaite PA and Yuan JX:
Activation of Notch signaling by short-term treatment with Jagged-1
enhances store-operated Ca(2+) entry in human pulmonary arterial
smooth muscle cells. Am J Physiol Cell Physiol. 306:C871–C878.
2014. View Article : Google Scholar
|
|
39
|
Zhang X, Chen J, Xu P and Tian X:
Protective effects of Astragaloside IV against hypoxic pulmonary
hypertension. Medchemcomm. 9:1715–1721. 2018. View Article : Google Scholar
|
|
40
|
Wang W, Liu J, Ma A, Miao R, Jin Y, Zhang
H, Xu K, Wang C and Wang J: mTORC1 is involved in hypoxia-induced
pulmonary hypertension through the activation of Notch3. J Cell
Physiol. 229:2117–2125. 2014. View Article : Google Scholar
|
|
41
|
Liang C, Ni GX, Shi XL, Jia L and Wang YL:
Astragaloside IV regulates the HIF/VEGF/Notch signaling pathway
through miRNA-210 to promote angiogenesis after ischemic stroke.
Restor Neurol Neurosci. 38:271–282. 2020.
|