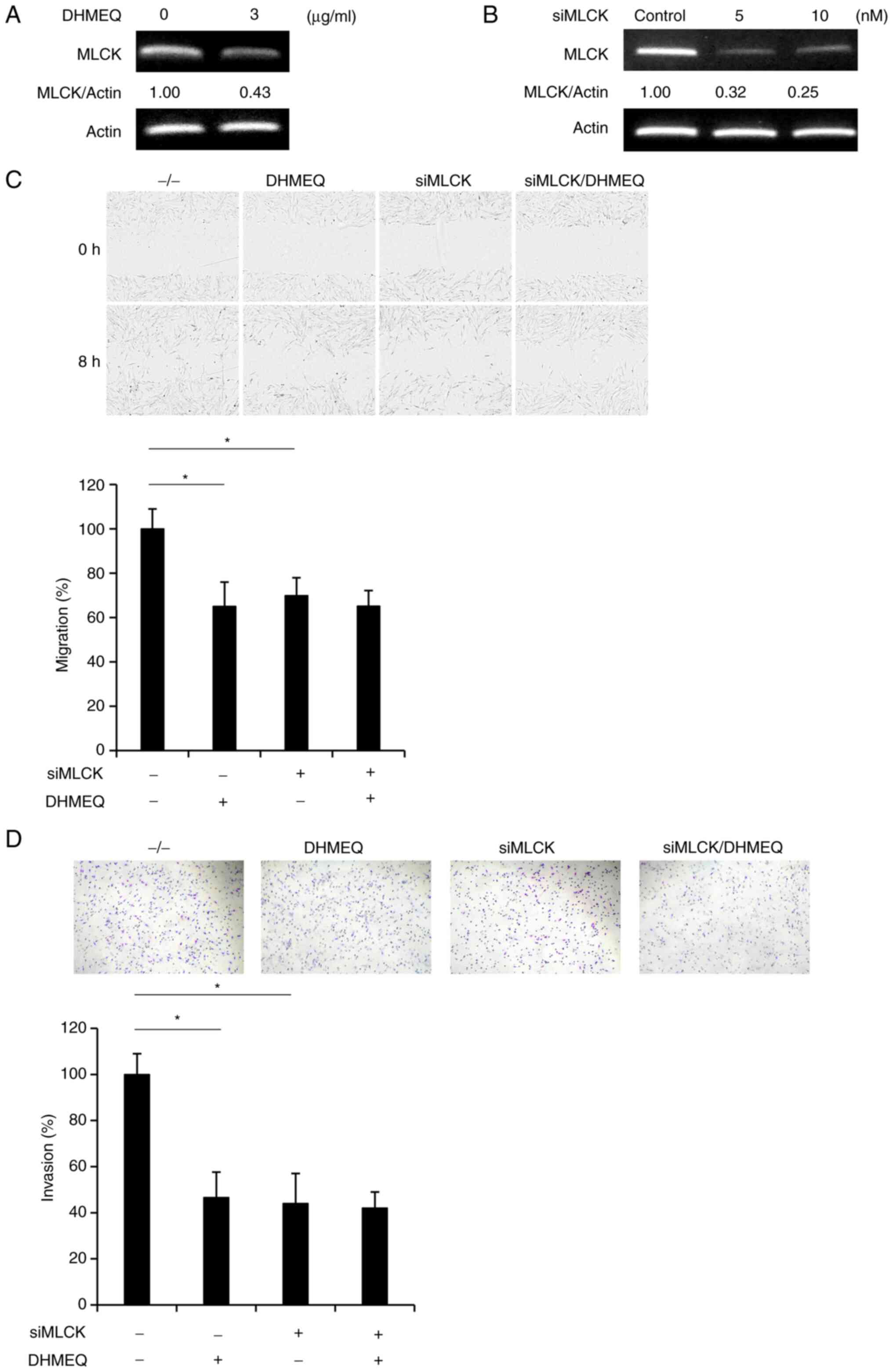
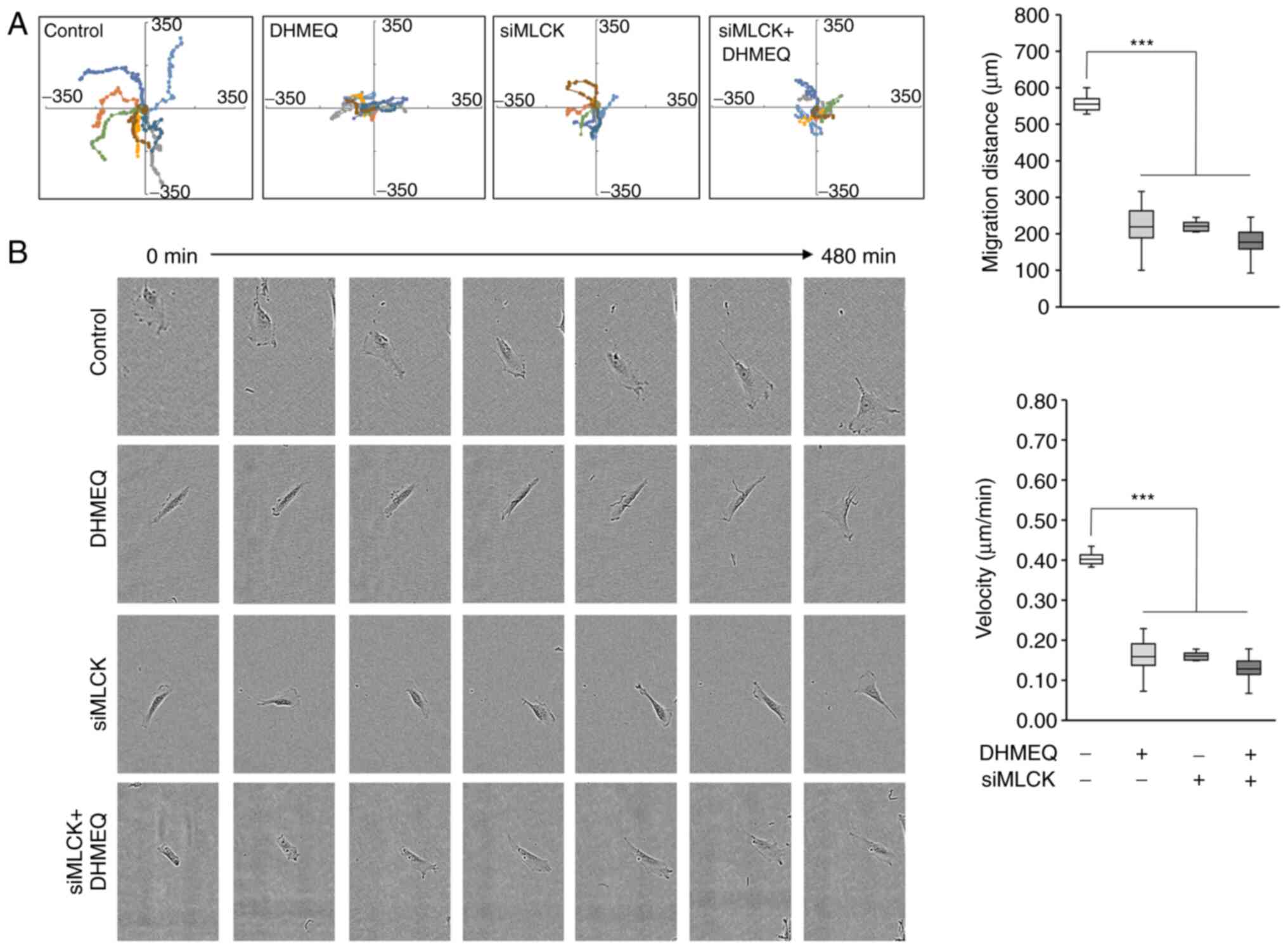
|
1
|
World Health Organization (WHO), .
International Classification of Diseases. 11th Revision (ICD-11).
Geneva: 2018
|
|
2
|
Zondervan KT, Becker CM and Missmer SA:
Endometriosis. N Engl J Med. 382:1244–1256. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Johnson NP and Hummelshoj L: World
endometriosis society montpellier consortium. Consensus on current
management of endometriosis. Hum Reprod. 28:1552–1568. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Matsumoto N, Ariga A, To-e S, Nakamura H,
Agata N, Hirano S, Inoue J and Umezawa K: Synthesis of NF-kappaB
activation inhibitors derived from epoxyquinomicin C. Bioorg Med
Chem Lett. 10:865–869. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Ariga A, Namekawa J, Matsumoto N, Inoue J
and Umezawa K: Inhibition of TNF-α-induced nuclear translocation
and activation of NF-κB by dehydroxymethyl-epoxyquinomicin. J Biol
Chem. 277:27625–27630. 2002. View Article : Google Scholar
|
|
6
|
Yamamoto M, Horie R, Takeiri M, Kozawa I
and Umezawa K: Inactivation of nuclear factor kappa B components by
covalent binding of (−)-dehydroxymethylepoxyquinomicin to specific
cysteine residues. J Med Chem. 51:5780–5788. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Takeiri M, Horie K, Takahashi D, Watanabe
M, Horie R, Simizu S and Umezawa K: Involvement of DNA binding
domain in the cellular stability and importin affinity of NF-κB
component RelB. Org Biomol Chem. 10:3053–3059. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ma J, Zhang Y, Sugai T, Kubota T, Keino H,
El-Salhy M, Ozaki M and Umezawa K: Inhibition of cellular and
animal inflammatory disease models by NF-κB inhibitor DHMEQ. Cells.
10:22712021. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Umezawa K, Breborowicz A and Gantsev S:
Anticancer activity of novel NF-κB inhibitor DHMEQ by
intraperitoneal administration. Oncol Res. 28:541–550. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Sosińska P, Maćkowiak B, Staniszewski R,
Umezawa K and Bręborowicz A: Inhibition of NF-κB with
dehydroxyepoxiquinomicin modifies function of human peritoneal
mesothelial cells. Am J Transl Res. 8:5756–5765. 2016.PubMed/NCBI
|
|
11
|
Ukaji T, Lin Y, Okada S and Umezawa K:
Inhibition of MMP-2-mediated cellular invasion by NF-κB inhibitor
DHMEQ in 3D culture of breast carcinoma MDA-MB-231 cells: A model
for early phase of metastasis. Biochem Biophys Res Commun.
485:76–81. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Umezawa K and Lin Y: Inhibition of matrix
metalloproteinase expression and cellular invasion by NF-κB
inhibitors of microbial origin. Biochim Biophys Acta Proteins
Proteom. 1868:1404122020. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Lin Y, Sidthipong K, Ma J, Koide N,
Umezawa K and Kubota T: Designed NF-κB inhibitor, DHMEQ, inhibits
KISS1R-mediated invasion and increases drug-sensitivity in mouse
plasmacytoma SP2/0 cells. Exp Ther Med. 22:10922021. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Noma N, Asagiri M, Takeiri M, Ohmae S,
Takemoto K, Iwaisako K, Simizu S and Umezawa K: Inhibition of
MMP-2-mediated mast cell invasion by NF-κB inhibitor DHMEQ in mast
cells. Int Arch Allergy Immunol. 166:84–90. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Sato M, Nakanishi K, Haga S, Fujiyoshi M,
Baba M, Mino K, Yimin, Niwa H, Yokoo H, Umezawa K, et al: Anoikis
induction and inhibition of peritoneal metastasis of pancreatic
cancer cells by a nuclear factor-kappa B inhibitor, (−)-DHMEQ.
Oncology Res. 21:333–343. 2014. View Article : Google Scholar
|
|
16
|
Sidthipong K, Ma J, Yu W, Wang Y,
Kobayashi S, Kishino S, Koide N, Yokochi T, Kato K, Okada S and
Umezawa K: Rational design, synthesis and in vitro evaluation of
novel exo-methylene butyrolactone salicyloylamide as NF-κB
inhibitor. Bioorg Med Chem Lett. 27:562–566. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Suzuki Y, Sugiyama C, Ohno O and Umezawa
K: Preparation and biological activities of optically active
dehydroxymethylepoxyquinomicin, a novel NF-κB inhibitor.
Tetrahedron. 60:7061–7066. 2004. View Article : Google Scholar
|
|
18
|
Lin Y, Chen Y, Ukaji T, Okada S and
Umezawa K: Isolation of ketomycin from Actinomycetes as an
inhibitor of 2D and 3D cancer cell invasion. J Antibiot (Tokyo).
72:148–157. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ito H, Tsunoda T, Riku M, Inaguma S, Inoko
A, Murakami H, Ikeda H, Matsuda M and Kasai K: Indispensable role
of STIL in the regulation of cancer cell motility through the
lamellipodial accumulation of ARHGEF7-PAK1 complex. Oncogene.
39:1931–1943. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Lin Y, Sidthipong K, Ma J, Koide N,
Umezawa K and Kubota T: The designed NF-κB inhibitor, DHMEQ,
inhibits KISS1R-mediated invasion and increases drug-sensitivity in
mouse plasmacytoma SP2/0 cells. Exp Ther Med. 22:10922021.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Karnan S, Ota A, Murakami H, Rahman ML,
Hasan MN, Wahiduzzaman M, Hanamura I, Quang Vu L, Inoko A, Hyodo T,
et al: Identification of CD24 as a potential diagnostic and
therapeutic target for malignant pleural mesothelioma. Cell Death
Discovry. 6:1272020. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Vandesompele J, De Preter K, Pattyn F,
Poppe B, Van Roy N, De Paepe A and Speleman F: Accurate
normalization of real-time quantitative RT-PCR data by geometric
averaging of multiple internal control genes. Genome Biol.
3:RESEARCH00342002. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Banu SK, Lee J, Speights VO Jr,
Starzinski-Powitz A and Arosh JA: Selective inhibition of
prostaglandin E2 receptors EP2 and EP4 induces apoptosis of human
endometriotic cells through suppression of ERK1/2, AKT, NFkappaB,
and beta-catenin pathways and activation of intrinsic apoptotic
mechanisms. Mol Endocrinol. 23:1291–1305. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kaponis A, Uwabe T, Taniguchi F, Ito M,
Deura I, Decavalas G, Terakawa N and Harada T: The role of NF-κB in
endometriosis. Front Biosci (Schol Ed). 4:1213–1234.
2012.PubMed/NCBI
|
|
25
|
Somlyo A and Somlyo A: Signal transduction
by G-proteins, rho-kinase and protein phosphatase to smooth muscle
and non-muscle myosin II. J Physiol. 522:177–185. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Tan J, Ravid S and Spudich J: Control of
nonmuscle myosins by phosphorylation. Annu Rev Biochem. 61:721–759.
1992. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Tohtong R, Phattarasakul K and
Jiraviriyakul A: Dependence of metastatic cancer cell invasion on
MLCK-catalyzed phosphorylation of myosin regulatory light chain.
Prostate Cancer Prostatic Dis. 6:212–216. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Chen C, Tao T, Wen C, He W, Qiao Y, Gao Y,
Chen X, Wang P, Chen C, Zhao W, et al: Myosin light chain kinase
(MLCK) regulates cell migration in a myosin regulatory light chain
phosphorylation-independent mechanism. J Biol Chem.
289:28478–28488. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Su Z, Gong Y, Yang H, Deng D and Liang Z:
Activation of the nuclear factor-kappa B signaling pathway damages
the epithelial barrier in the human pancreatic ductal
adenocarcinoma cell line HPAF-II. Pancreas. 48:1380–1385. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wang F, Graham W, Wang Y, Witkowski E,
Schwarz B and Turner J: Interferon-gamma and tumor necrosis
factor-alpha synergize to induce intestinal epithelial barrier
dysfunction by up-regulating myosin light chain kinase expression.
Am J Pathol. 166:409–419. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Ma T, Boivin M, Ye D, Pedram A and Said H:
Mechanism of TNF-(alpha) modulation of Caco-2 intestinal epithelial
tight junction barrier: Role of mysion light-chanin kinase protein
expression. Am J Physiol Gastrointest Liver Physiol. 288:G422–G430.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Ma T, Iwamoto G, Hoa N, Akotia V, Pedram
A, Boivin M and Said H: TNF-alpha-induced increase in intestinal
epithelial tight junction permeability requires NF-kappa b
activation. Am J Physiol Gastrointest Liver Physiol. 286:G367–G376.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Graham W, Wang F, Clayburgh D, Cheng J,
Yoon B, Wang Y, Lin A and Turner J: Tumor necrosis factor-induced
long myosin light chain kinase transcription is regulated by
differentiation-dependent signaling events. Characterization of the
human long myosin light chain kinase promoter. J Biol Chem.
281:26205–26215. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Ye D and Ma TY: Cellular and molecular
mechanisms that mediate basal and tumour necrosis
factor-alpha-induced regulation of myosin light chain kinase gene
activity. J Cell Mol Med. 12:1331–1346. 2008. View Article : Google Scholar : PubMed/NCBI
|