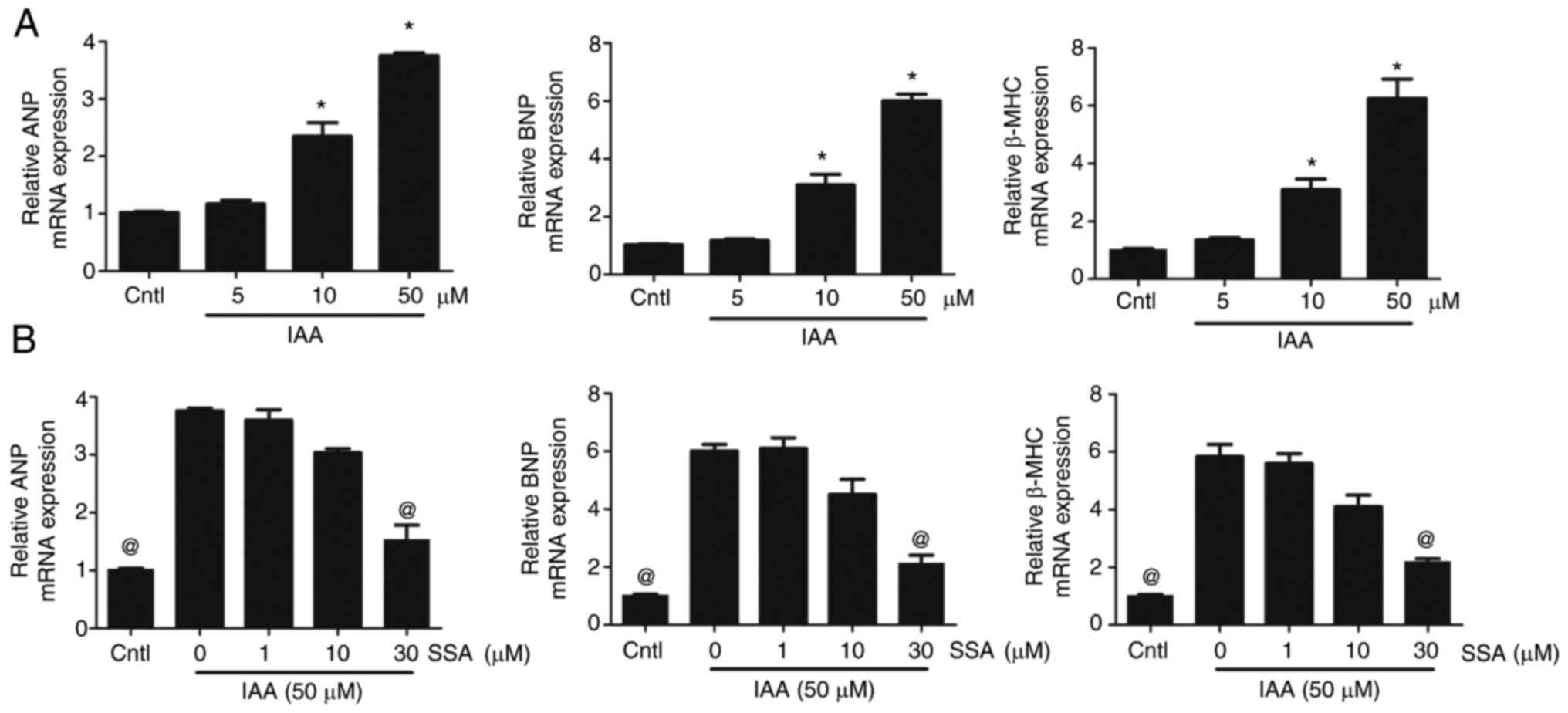
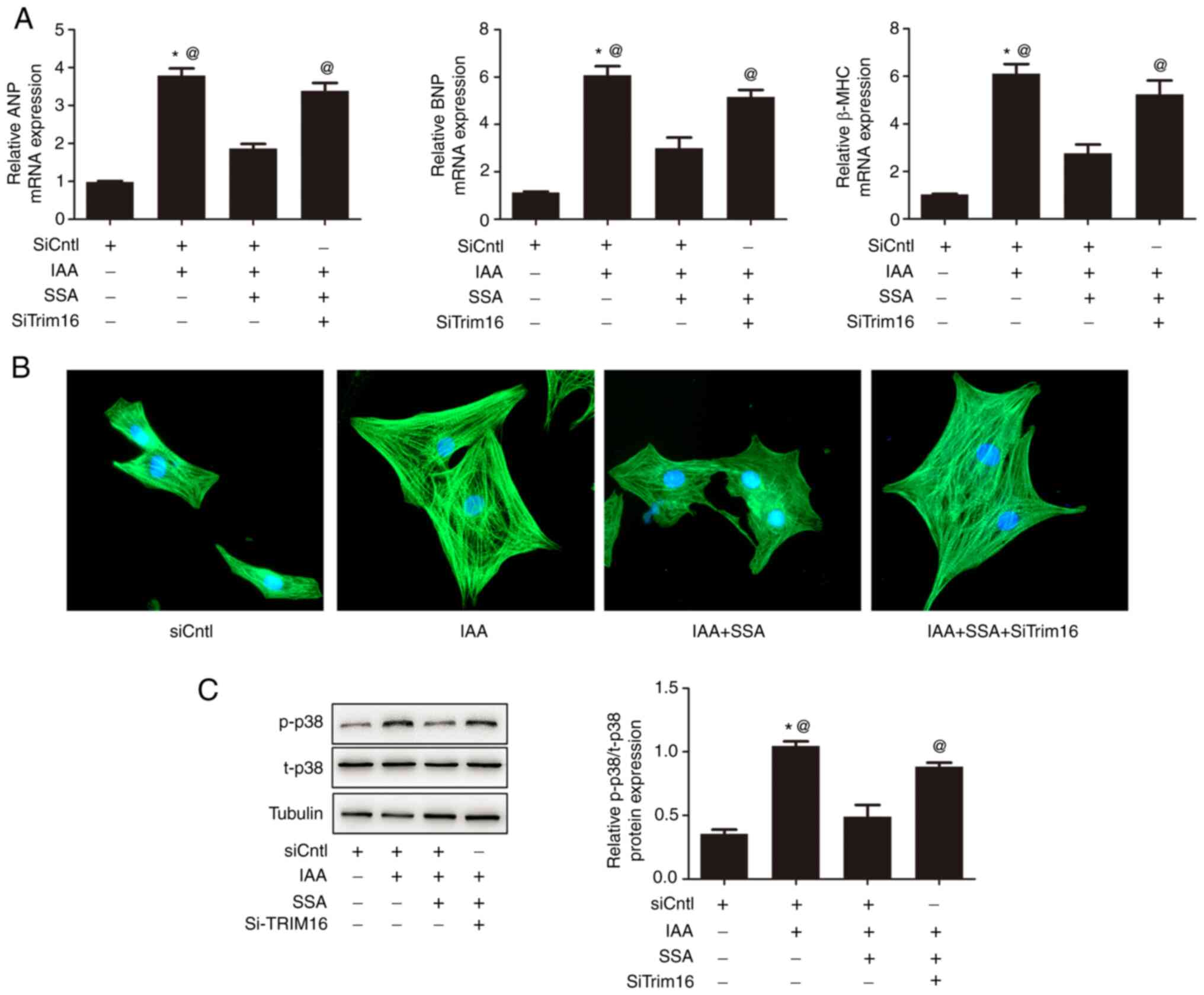
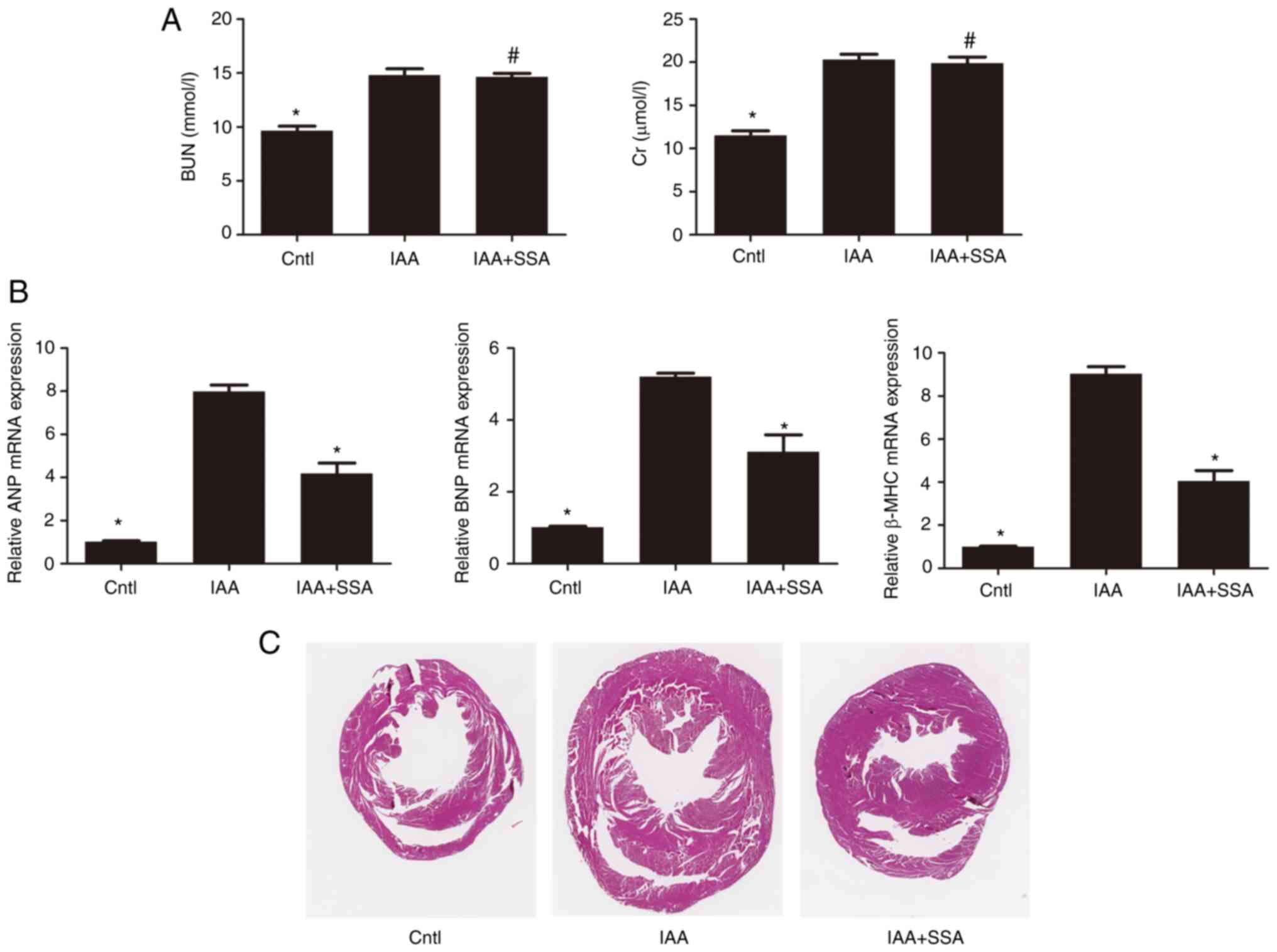
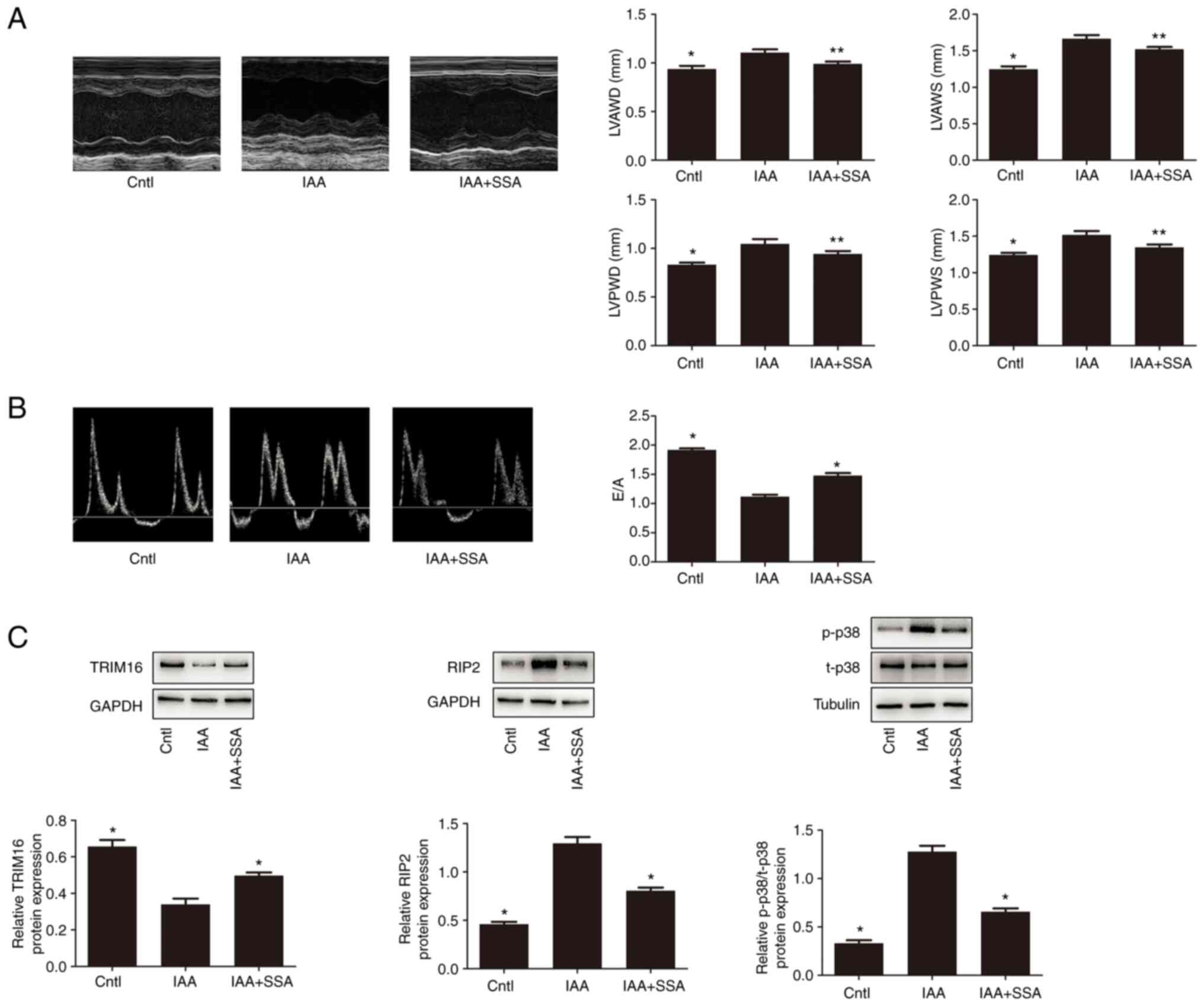
|
1
|
Lv JC and Zhang LX: Prevalence and disease
burden of chronic kidney disease. Adv Exp Med Biol. 1165:3–15.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Webster AC, Nagler EV, Morton RL and
Masson P: Chronic kidney disease. Lancet. 389:1238–1252. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Matsushita K, Ballew SH, Wang AY,
Kalyesubula R, Schaeffner E and Agarwal R: Epidemiology and risk of
cardiovascular disease in populations with chronic kidney disease.
Nat Rev Nephrol. 18:696–707. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Patel N, Yaqoob MM and Aksentijevic D:
Cardiac metabolic remodelling in chronic kidney disease. Nat Rev
Nephrol. 18:524–537. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Chen C, Xie C, Xiong Y, Wu H, Wu L, Zhu J,
Xing C and Mao H: Damage of uremic myocardium by p-cresyl sulfate
and the ameliorative effect of Klotho by regulating SIRT6
ubiquitination. Toxicol Lett. 367:19–31. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Yang K, Wang C, Nie L, Zhao X, Gu J, Guan
X, Wang S, Xiao T, Xu X, He T, et al: Klotho protects against
indoxyl sulphate-induced myocardial hypertrophy. J Am Soc Nephrol.
26:2434–2446. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Han H, Zhu J, Zhu Z, Ni J, Du R, Dai Y,
Chen Y, Wu Z, Lu L and Zhang R: p-Cresyl sulfate aggravates cardiac
dysfunction associated with chronic kidney disease by enhancing
apoptosis of cardiomyocytes. J Am Heart Assoc. 4:e0018522015.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wu CC, Hsieh MY, Hung SC, Kuo KL, Tsai TH,
Lai CL, Chen JW, Lin SJ, Huang PH and Tarng DC: Serum indoxyl
sulfate associates with postangioplasty thrombosis of dialysis
grafts. J Am Soc Nephrol. 27:1254–1264. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Jourde-Chiche N, Dou L, Cerini C,
Dignat-George F, Vanholder R and Brunet P: Protein-bound
toxins-update 2009. Semin Dial. 22:334–339. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Dou L, Sallée M, Cerini C, Poitevin S,
Gondouin B, Jourde-Chiche N, Fallague K, Brunet P, Calaf R, Dussol
B, et al: The cardiovascular effect of the uremic solute indole-3
acetic acid. J Am Soc Nephrol. 26:876–887. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Shi Y, Tian H, Wang Y, Shen Y, Zhu Q and
Ding F: Improved dialysis removal of protein-bound uraemic toxins
with a combined displacement and adsorption technique. Blood Purif.
51:548–558. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Shuai-Cheng W, Xiu-Ling C, Jian-Qing S,
Zong-Mei W, Zhen-Jiang Y and Lian-Tao L: Saikosaponin A protects
chickens against pullorum disease via modulation of cholesterol.
Poult Sci. 98:3539–3547. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zhu Y, Chen X, Rao X, Zheng C and Peng X:
Saikosaponin a ameliorates l ipopolysaccharide and
d-galactosamine-induced liver injury via activating LXRα. Int
Immunopharmacol. 72:131–137. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Liu Y, Gao L, Zhao X, Guo S, Liu Y, Li R,
Liang C, Li L, Dong J, Li L and Yang H: Saikosaponin a protects
from pressure overload-induced cardiac fibrosis via inhibiting
fibroblast activation or endothelial cell endMT. Int J Biol Sci.
14:1923–1934. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Song Y, Sun H, Gao S, Tang K, Zhao Y, Xie
G and Gao H: Saikosaponin a attenuates lead-induced kidney injury
through activating Nrf2 signaling pathway. Comp Biochem Physiol C
Toxicol Pharmacol. 242:1089452021. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Liu J, Li W, Deng KQ, Tian S, Liu H, Shi
H, Fang Q, Liu Z, Chen Z, Tian T, et al: The E3 ligase TRIM16 is a
key suppressor of pathological cardiac hypertrophy. Circ Res.
130:1586–1600. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Roshanazadeh MR, Adelipour M, Sanaei A,
Chenane H and Rashidi M: TRIM3 and TRIM16 as potential tumor
suppressors in breast cancer patients. BMC Res Notes. 15:3122022.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Humphries F, Yang S, Wang B and Moynagh
PN: RIP kinases: Key decision makers in cell death and innate
immunity. Cell Death Differ. 22:225–236. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Jacquet S, Nishino Y, Kumphune S, Sicard
P, Clark JE, Kobayashi KS, Flavell RA, Eickhoff J, Cotton M and
Marber MS: The role of RIP2 in p38 MAPK activation in the stressed
heart. J Biol Chem. 283:11964–1171. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ravi V, Jain A, Taneja A, Chatterjee K and
Sundaresan NR: Isolation and culture of neonatal murine primary
cardiomyocytes. Curr Protoc. 1:e1962021.PubMed/NCBI
|
|
21
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Oh JK, Appleton CP, Hatle LK, Nishimura
RA, Seward JB and Tajik AJ: The noninvasive assessment of left
ventricular diastolic function with two-dimensional and Doppler
echocardiography. J Am Soc Echocardiogr. 10:246–270. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Glassock RJ, Warnock DG and Delanaye P:
The global burden of chronic kidney disease: Estimates, variability
and pitfalls. Nat Rev Nephrol. 13:104–114. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Chen TK, Knicely DH and Grams ME: Chronic
kidney disease diagnosis and management: A review. JAMA.
322:1294–1304. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Thomas R, Kanso A and Sedor JR: Chronic
kidney disease and its complications. Prim Care. 35:329–344. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
House AA, Wanner C, Sarnak MJ, Piña IL,
McIntyre CW, Komenda P, Kasiske BL, Deswal A, deFilippi CR, Cleland
JGF, et al: Heart failure in chronic kidney disease: Conclusions
from a kidney disease: Improving global outcomes (KDIGO)
controversies conference. Kidney Int. 95:1304–1317. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Chen C, Xie C, Wu H, Wu L, Zhu J, Mao H
and Xing C: Uraemic cardiomyopathy in different mouse models. Front
Med (Lausanne). 8:6905172021. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Cao XS, Chen J, Zou JZ, Zhong YH, Teng J,
Ji J, Chen ZW, Liu ZH, Shen B, Nie YX, et al: Association of
indoxyl sulfate with heart failure among patients on hemodialysis.
Clin J Am Soc Nephrol. 10:111–119. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Yamaguchi K, Yisireyili M, Goto S, Cheng
XW, Nakayama T, Matsushita T, Niwa T, Murohara T and Takeshita K:
Indoxyl sulfate activates NLRP3 inflammasome to induce cardiac
contractile dysfunction accompanied by myocardial fibrosis and
hypertrophy. Cardiovasc Toxicol. 22:365–377. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Fernandez-Prado R, Esteras R, Perez-Gomez
MV, Gracia-Iguacel C, Gonzalez-Parra E, Sanz AB, Ortiz A and
Sanchez-Niño MD: Nutrients turned into toxins: Microbiota
modulation of nutrient properties in chronic kidney disease.
Nutrients. 9:4892017. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Liabeuf S, Laville SM, Glorieux G,
Cheddani L, Brazier F, Beauport DT, Valholder R, Choukroun G and
Massy ZA: Difference in profiles of the gut-derived tryptophan
metabolite indole acetic acid between transplanted and
non-transplanted patients with chronic kidney disease. Int J Mol
Sci. 21:20312020. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Claro LM, Moreno-Amaral AN, Gadotti AC,
Dolenga CJ, Nakao LS, Azevedo MLV, de Noronha L, Olandoski M, de
Moraes TP, Stinghen AEM and Pécoits-Filho R: The impact of uremic
toxicity induced inflammatory response on the cardiovascular burden
in chronic kidney disease. Toxins (Basel). 10:3842018. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Chinnappa S, Tu YK, Yeh YC, Glorieux G,
Vanholder R and Mooney A: Association between protein-bound uremic
toxins and asymptomatic cardiac dysfunction in patients with
chronic kidney disease. Toxins (Basel). 10:5202018. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Stockler-Pinto MB, Soulage CO, Borges NA,
Cardozo LFM, Dolenga CJ, Nakao LS, Pecoits-Filho R, Fouque D and
Mafra D: From bench to the hemodialysis clinic: Protein-bound
uremic toxins modulate NF-κB/Nrf2 expression. Int Urol Nephrol.
50:347–354. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Bataille S, Pelletier M, Sallée M, Berland
Y, McKay N, Duval A, Gentile S, Mouelhi Y, Brunet P and Burtey S:
Indole 3-acetic acid, indoxyl sulfate and paracresyl-sulfate do not
influence anemia parameters in hemodialysis patients. BMC Nephrol.
18:2512017. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Addi T, Poitevin S, McKay N, El Mecherfi
KE, Kheroua O, Jourde-Chiche N, de Macedo A, Gondouin B, Cerini C,
Brunet P, et al: Mechanisms of tissue factor induction by the
uremic toxin indole-3 acetic acid through aryl hydrocarbon
receptor/nuclear factor-kappa B signaling pathway in human
endothelial cells. Arch Toxicol. 93:121–136. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Hager THI: Assessment toxic effects of
exposure to 3-indoleacetic acid via hemato-biochemical, hormonal,
and histopathological screening in rats. Environ Sci Pollut Res
Int. 29:90703–90718. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Wang X, Wang Q, Burczynski FJ, Kong W and
Gong Y: Saikosaponin A of Bupleurum chinense (Chaihu) elevates bone
morphogenetic protein 4 (BMP-4) during hepatic stellate cell
activation. Phytomedicine. 20:1330–1335. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Wu SJ, Tam KW, Tsai YH, Chang CC and Chao
JC: Curcumin and saikosaponin a inhibit chemical-induced liver
inflammation and fibrosis in rats. Am J Chin Med. 38:99–111. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Zhou F, Wang N, Yang L, Zhang LC, Meng LJ
and Xia YC: Saikosaponin A protects against dextran sulfate
sodium-induced colitis in mice. Int Immunopharmacol. 72:454–458.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Du ZA, Sun MN and Hu ZS: Saikosaponin a
Ameliorates LPS-induced acute lung injury in mice. Inflammation.
41:193–198. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Wang X and Yang G: Saikosaponin A
attenuates neural injury caused by ischemia/reperfusion. Transl
Neurosci. 11:227–235. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Fu Y, Hu X, Cao Y, Zhang Z and Zhang N:
Saikosaponin a inhibits lipopolysaccharide-oxidative stress and
inflammation in Human umbilical vein endothelial cells via
preventing TLR4 translocation into lipid rafts. Free Radic Biol
Med. 89:777–785. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
He D, Wang H, Xu L, Wang X, Peng K, Wang
L, Liu P and Qu P: Saikosaponin-a attenuates oxidized LDL uptake
and prompts cholesterol efflux in THP-1 cells. J Cardiovasc
Pharmacol. 67:510–518. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Zhang YJ, Wu SS, Chen XM, Pi JK, Cheng YF,
Zhang Y, Wang XJ, Luo D, Zhou JH, Xu JY, et al: Saikosaponin D
alleviates DOX-induced cardiac injury in vivo and in vitro. J
Cardiovasc Pharmacol. 79:558–567. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Huang M, Yan Y, Deng Z, Zhou L, She M,
Yang Y, Zhang M and Wang D: Saikosaponin A and D attenuate skeletal
muscle atrophy in chronic kidney disease by reducing oxidative
stress through activation of PI3K/AKT/Nrf2 pathway. Phytomedicine.
114:1547662023. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Ozato K, Shin DM, Chang TH and Morse HC
III: TRIM family proteins and their emerging roles in innate
immunity. Nat Rev Immunol. 8:849–860. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Spirina LV, Yunusova NV, Kondakova IV and
Tarasenko NV: Transcription factors Brn-3α and TRIM16 in cancers,
association with hormone reception. Heliyon. 5:e020902019.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Bell JL, Malyukova A, Holien JK, Koach J,
Parker MW, Kavallaris M, Marshall GM and Cheung BB: TRIM16 acts as
an E3 ubiquitin ligase and can Heterodimerize with other TRIM
family members. PLoS One. 7:e374702012. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Jena KK, Mehto S, Kolapalli SP, Nath P,
Sahu R, Chauhan NR, Sahoo PK, Dhar K, Das SK and Chauhan S and
Chauhan S: TRIM16 governs the biogenesis and disposal of
stress-induced protein aggregates to evade cytotoxicity:
Implication for neurodegeneration and cancer. Autophagy.
15:924–926. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Kim PY, Tan O, Liu B, Trahair T, Liu T,
Haber M, Norris MD, Marshall GM and Cheung BB: High TDP43
expression is required for TRIM16-induced inhibition of cancer cell
growth and correlated with good prognosis of neuroblastoma and
breast cancer patients. Cancer Lett. 374:315–323. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Chauhan S, Kumar S, Jain A, Ponpuak M,
Mudd MH, Kimura T, Choi SW, Peters R, Mandell M, Bruun JA, et al:
TRIMs and galectins globally cooperate and TRIM16 and galectin-3
co-direct autophagy in endomembrane damage homeostasis. Dev Cell.
39:13–27. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Di Rienzo M, Romagnoli A, Antonioli M,
Piacentini M and Fimia GM: TRIM proteins in autophagy: Selective
sensors in cell damage and innate immune responses. Cell Death
Differ. 27:887–902. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Borlepawar A, Rangrez AY, Bernt A,
Christen L, Sossalla S, Frank D and Frey N: TRIM24 protein promotes
and TRIM32 protein inhibits cardiomyocyte hypertrophy via
regulation of dysbindin protein levels. J Biol Chem.
292:10180–10196. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Chen L, Huang J, Ji Y, Zhang X, Wang P,
Deng K, Jiang X, Ma G and Li H: Tripartite motif 32 prevents
pathological cardiac hypertrophy. Clin Sci. 130:813–828. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Zhao Y, Liu H, Xi X, Chen S and Liu D:
TRIM16 protects human periodontal ligament stem cells from
oxidative stress-induced damage via activation of PICOT. Exp Cell
Res. 397:1123362020. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Jena KK, Kolapalli SP, Mehto S, Nath P,
Das B, Sahoo PK, Ahad A, Syed GH, Raghav SK, Senapati S, et al:
TRIM16 controls assembly and degradation of protein aggregates by
modulating the p62-NRF2 axis and autophagy. EMBO J. 37:e983582018.
View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Andersson L, Täng MS, Lundqvist A, Lindbom
M, Mardani I, Fogelstrand P, Shahrouki P, Redfors B, Omerovic E,
Levin M, et al: Rip2 modifies VEGF-induced signalling and vascular
permeability in myocardial ischaemia. Cardiovasc Res. 107:478–486.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Lin Z, Liao HH, Zhou ZY, Zhang N, Li WJ
and Tang QZ: RIP2 inhibition alleviates lipopolysaccharide-induced
septic cardiomyopathy via regulating TAK1 signaling. Eur J
Pharmacol. 947:1756792023. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Yang JJ, Zhang N, Zhou ZY, Ni J, Feng H,
Li WJ, Mou SQ, Wu HM, Deng W, Liao HH and Tang QZ:
Cardiomyocyte-specific RIP2 overexpression exacerbated pathologic
remodeling and contributed to spontaneous cardiac hypertrophy.
Front Cell Dev Biol. 9:6882382021. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Yang S, Wang B, Humphries F, Jackson R,
Healy ME, Bergin R, Aviello G, Hall B, McNamara D, Darby T, et al:
Pellino3 ubiquitinates RIP2 and mediates Nod2-induced signaling and
protective effects in colitis. Nat Immunol. 14:927–936. 2013.
View Article : Google Scholar : PubMed/NCBI
|