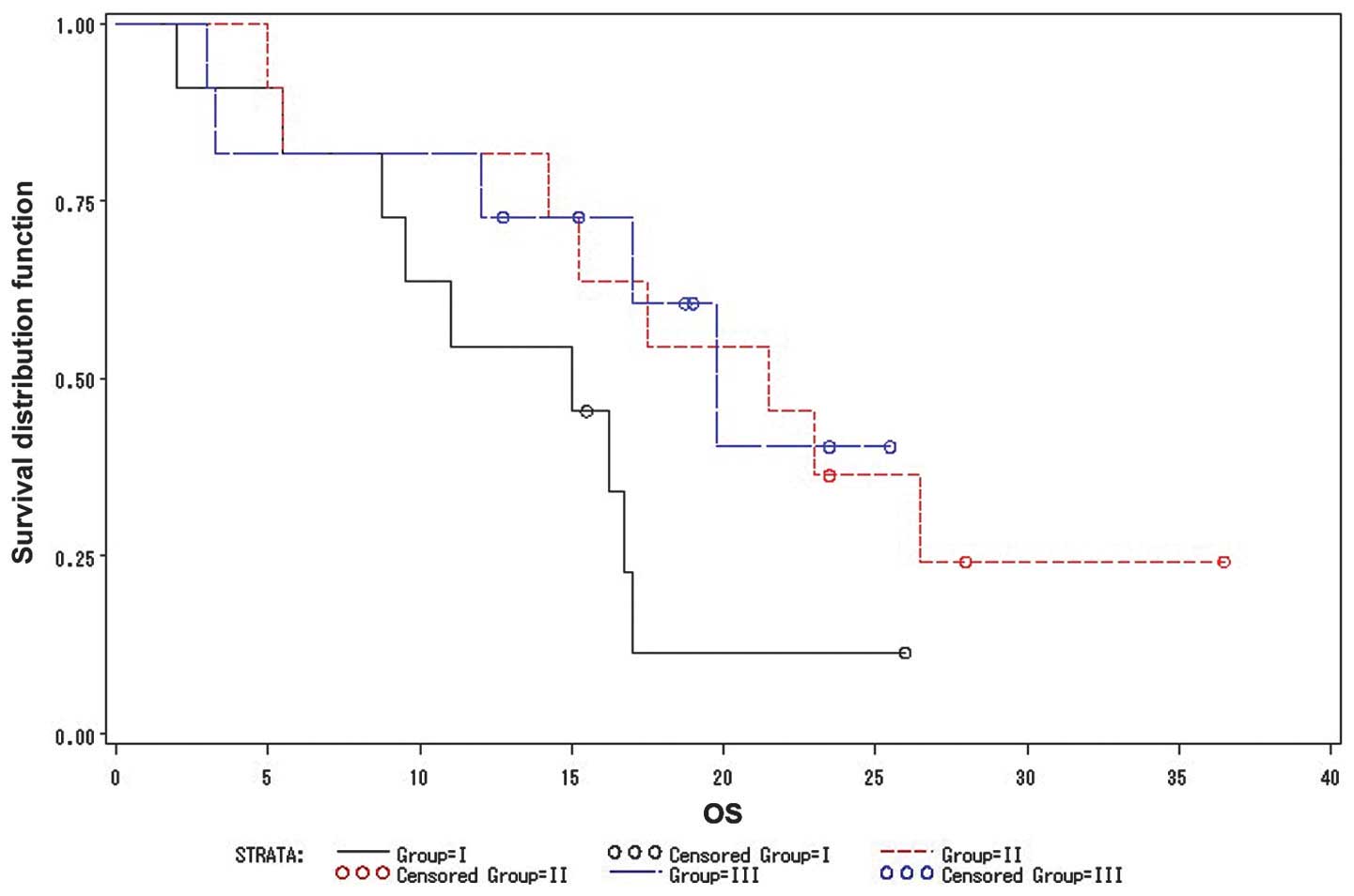
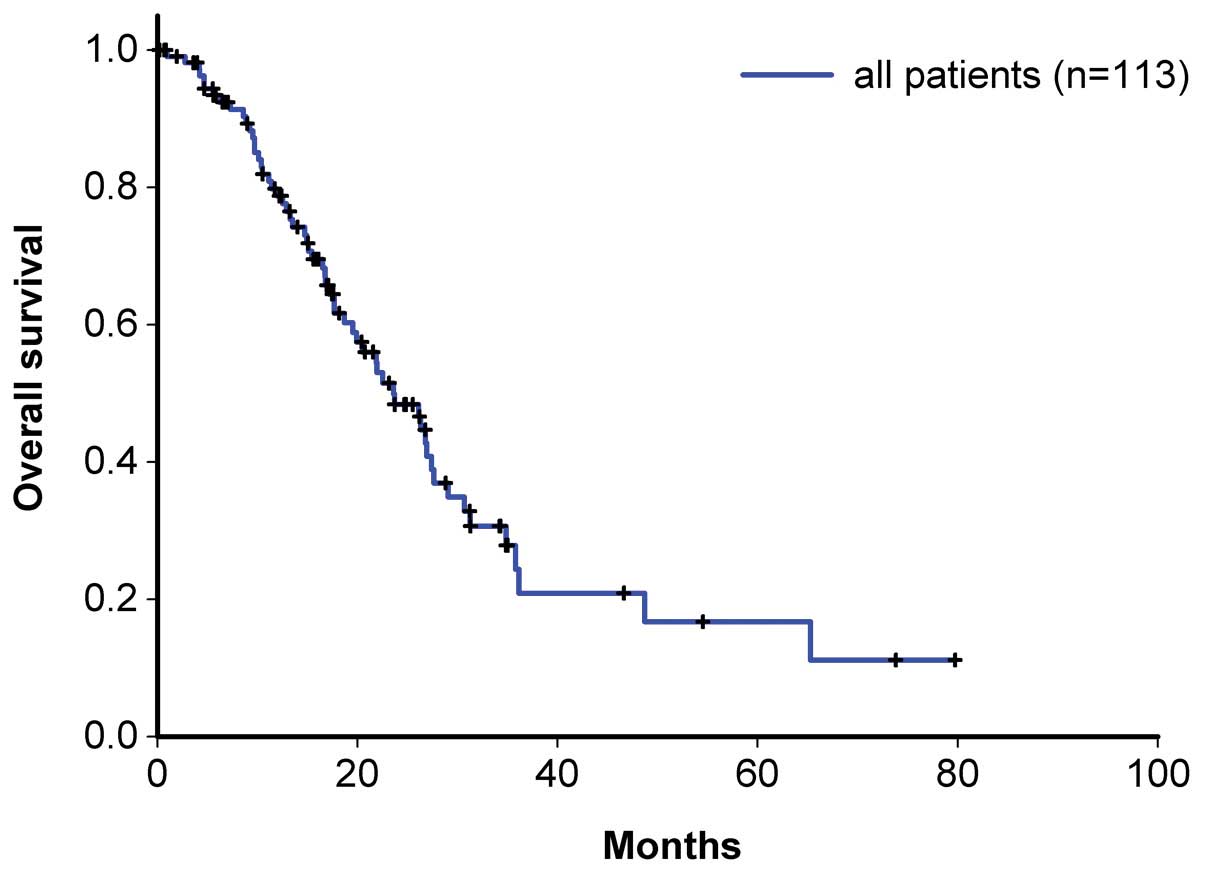
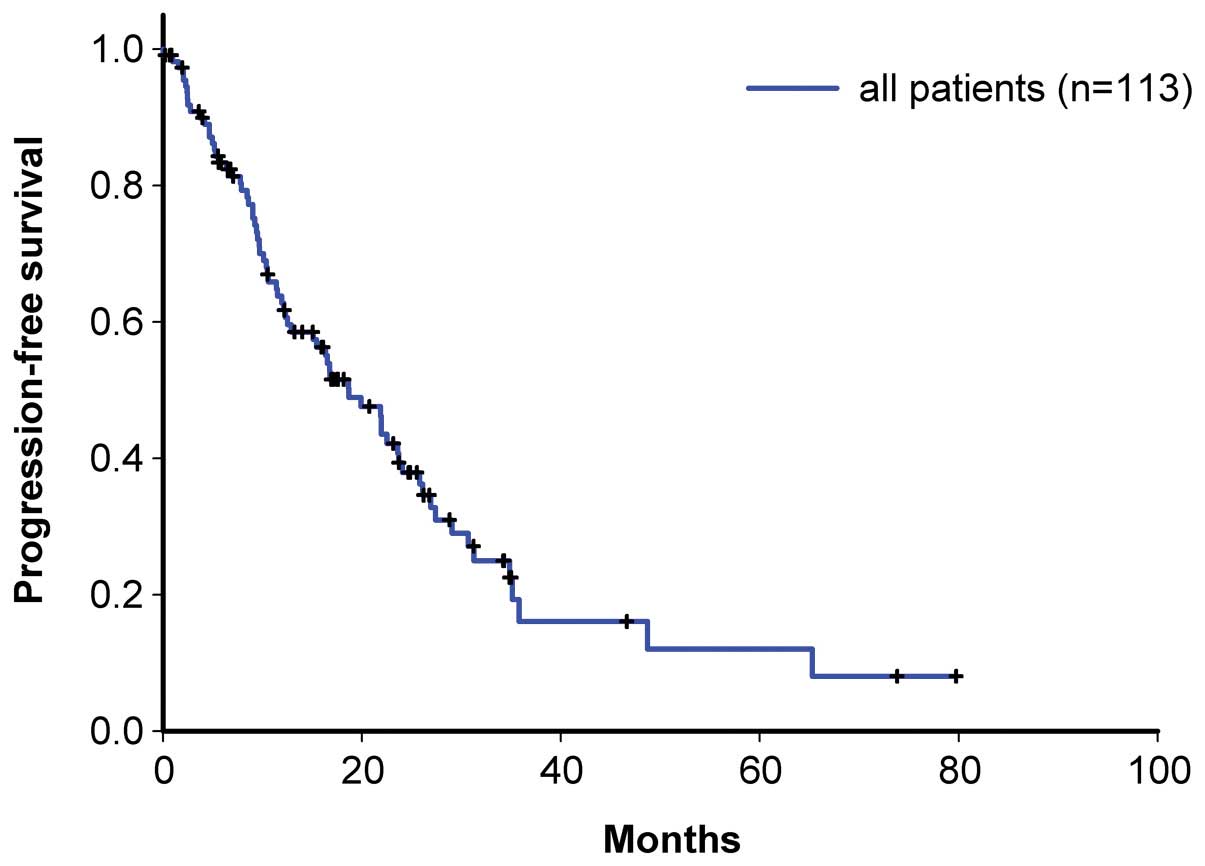
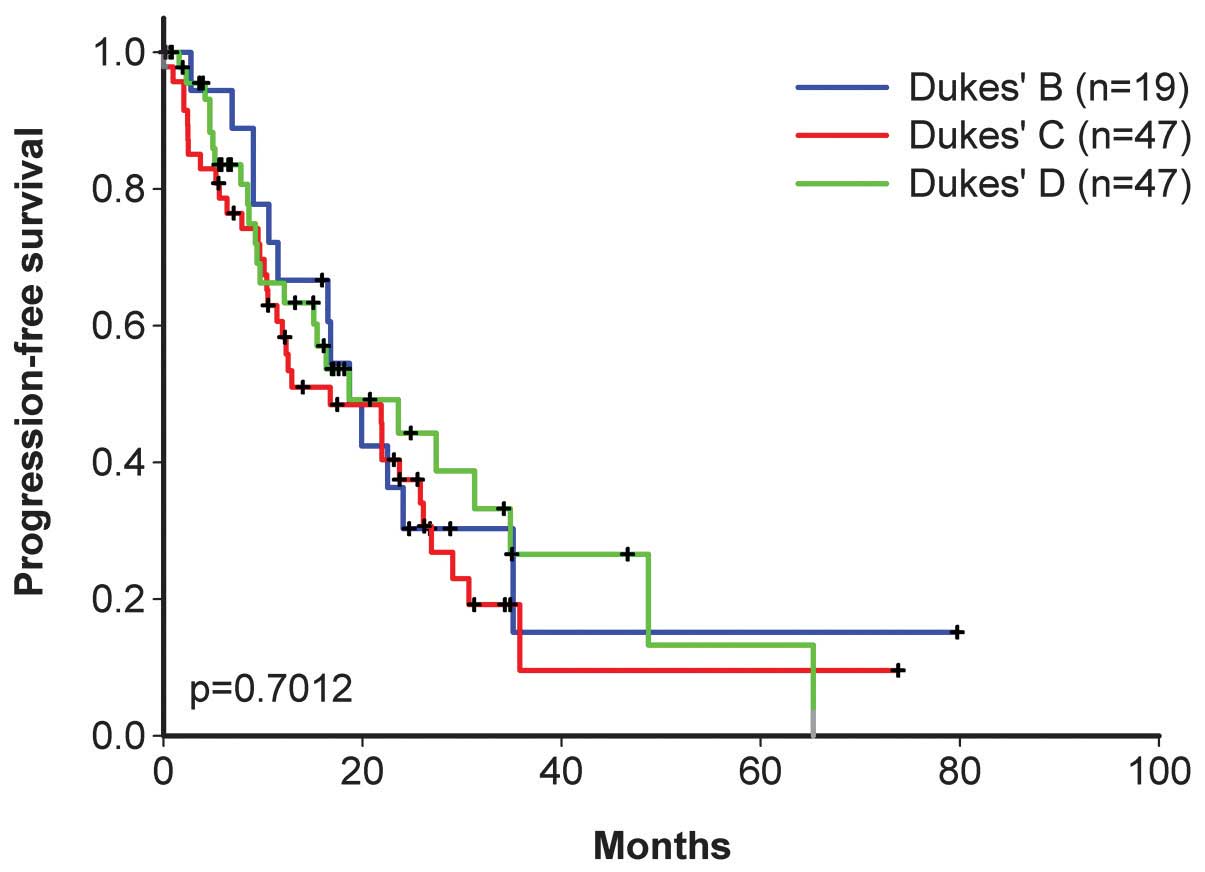
|
1
|
Yeh KH, Cheng AL, Lin MT, et al: A phase
II study of weekly 24-hour infusion of high-dose 5-fluorouracil and
leucovorin (HDFL) in the treatment of recurrent or metastatic
colorectal cancers. Anticancer Res. 17:3867–3871. 1997.
|
|
2
|
Mermershtain W, Lavrenkov K and Cohen Y:
Phase II study of weekly high-dose fluorouracil in previously
treated patients with metastatic colorectal cancer. J Chemother.
12:183–185. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Cunningham D, Pyrhönen S, James RD, et al:
Randomized trial of irinotecan plus supportive care versus
supportive care alone after fluorouracil failure for patient with
metastatic colorectal cancer. Lancet. 352:1413–1418. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Douillard JY, Cunningham D, Roth AD, et
al: Irinotecan combined with fluorouracil compared with
fluorouracil alone as first-line treatment for metastatic
colorectal cancer: a multicenter randomized trial. Lancet.
355:1041–1047. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Rothenberg ML, Cox JV, Devore RF, et al: A
multicenter phase II trial of weekly irinotecan (CPT-11) in
patients with previously treated colorectal carcinoma. Cancer.
85:786–795. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Van Cutsem E, Pozzo C, Starkhammar H, et
al: A phase II study of irinotecan alternated with five days bolus
of 5-fluorouracil and leucovorin in first-line chemotherapy of
metastatic colorectal cancer. Ann Oncol. 9:1199–1204. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ota K, Taguchi T and Kimura K: Report on
nationwide-pooled data and cohort investigation in UFT phase II
study. Cancer Chemother Pharmacol. 22:333–338. 1988. View Article : Google Scholar
|
|
8
|
Malet-Martino M and Martino R: Clinical
studies of three oral prodrugs of 5-fluorouracil (capecitabine,
UFT, S-1): a review. Oncologist. 7:288–323. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kusunoki M, Yanagi H, Noda M, et al:
Results of pharmacokinetic modulating chemotherapy in combination
with hepatic arterial 5-fluorouracil infusion and oral UFT after
resection of hepatic colorectal metastases. Cancer. 89:1228–1235.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Mackay HJ, Hill M, Twelves C, et al: A
phase I/II study of oral uracil/tegafur (UFT), leucovorin, and
irinotecan in patients with advanced colorectal cancer. Ann Oncol.
14:1264–1269. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Mibu R, Tanaka S, Futami K, et al: Phase
I/II study of irinotecan and UFT for advanced or metastatic
colorectal cancer. Anticancer Res. 27:2673–2678. 2007.PubMed/NCBI
|
|
12
|
Bajetta E, Bartolomeo MD, Buzzoni R, et
al: Uracil/ftorafur/leucovorin combined with irinotecan (TEGAFIRI)
or oxaliplatin (TEGAFOX) as first-line treatment for metastatic
colorectal cancer patient: results of randomized phase II study. Br
J Cancer. 96:439–444. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Delord JP, Bennouna J, Artru P, et al:
Phase II study of UFT with leucovorin and irinotecan (TEGAFIRI):
first-line therapy for metastatic colorectal cancer. Br J Cancer.
97:297–301. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ogata Y, Mori S, Ishibashi Y, et al:
Metronomic chemotherapy using weekly low-dosage CPT-11 and UFT as
postoperative adjuvant therapy in colorectal cancer at high risk to
recurrence. J Exp Clin Cancer Res. 26:475–482. 2007.
|
|
15
|
Hsu TC: Combination chemotherapy with
irinotecan and oral uracil-tegafur for recurrent or metastatic
colorectal cancer - results of different regimens. J Chinese Oncol
Soc. 25:254–261. 2009.(In Chinese).
|
|
16
|
Oken MM, Creech RH, Tormey DC, et al:
Toxicity and response criteria of the Eastern Cooperative Oncology
Group. Am J Clin Oncol. 5:649–655. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Schag CC, Heinrich RL and Ganz PA:
Karnofsky performance status revisited: reliability, validity, and
guidelines. J Clin Oncology. 2:187–193. 1984.
|
|
18
|
Dukes CE: The surgical pathology of rectal
cancer. J Clin Pathol. 2:95–98. 1949. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Eisenhauer EA, Therasse P, Bogaerts J, et
al: New response evaluation criteria in solid tumours: revised
RECIST guideline (version 1.1). Eur J Cancer. 45:228–247. 2009.
View Article : Google Scholar
|
|
20
|
Cancer Therapy Evaluation Program. Common
Toxicity Criteria, Version 2.0. April 30–1999, http://prevention.cancer.gov/files/clinical-trials/common-toxicity-criteria.pdf.
Accessed November 22, 2014
|
|
21
|
Jemal A, Tiwari RC, Murray T, et al:
American Cancer Society: Cancer statistics, 2004. CA Cancer J Clin.
54:8–29. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Parkin DM, Pisani P and Ferlay J: Global
cancer statistics. CA Cancer J Clin. 49:33–64. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Colorectal Cancer. Estimated Incidence,
Mortality and Prevalence Worldwide in 2012. GLOBOCAN. 2012,
http://globocan.iarc.fr/old/FactSheets/cancers/colorectal-new.asp.
Accessed November 22, 2014
|
|
24
|
Colorectal Cancer Facts & Figures
2014–2016. American Cancer Society; 2014, http://www.cancer.org/acs/groups/content/documents/document/acspc-042280.pdf.
Accessed November 22, 2014
|
|
25
|
DeVita VT Jr, Hellman S and Rosenberg SA:
Cancer Principles and Practice of Oncology. 5th edition.
Lippincott-Raven; Philadelphia, PA: pp. 437–452. 1997
|
|
26
|
Fujii S, Kitano S, Ikenaka K and Shirasaka
T: Studies on coadministration of uracil or cytosine on antitumor
activity of FT-207 or 5-FU derivatives. Jpn J Cancer Chemother.
6:377–384. 1979.
|
|
27
|
Douillard JY, Hoff P, Skillings JR, et al:
Multicenter phase III study of uracil/tegafur and oral leucovorin
versus fluorouracil and leucovorin in patients with previously
untreated metastatic colorectal cancer. J Clin Oncol. 20:3605–3616.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Hoff PM, Lassere Y and Pazdur R:
Tegafur/uracil + calcium folinate in colorectal cancer: double
modulation of fluorouracil. Drugs. 58:77–83. 1999. View Article : Google Scholar
|
|
29
|
Köhne CH and Peters GJ: UFT: mechanism of
drug action. Oncology (Williston Park). 14(10 Suppl 9): 13–18.
2000.
|
|
30
|
Pazdur R, Hoff PM, Medgyesy D, et al: The
oral fluorouracil prodrugs. Oncology (Williston Park). 12(10 Suppl
7): 48–51. 1998.
|
|
31
|
Carmichael J, Popiela T, Radstone D, et
al: Randomized comparative study of tegafur/uracil and oral
leucovorin versus parenteral fluorouracil and leucovorin in
patients with previously untreated metastatic colorectal cancer. J
Clin Oncol. 20:3617–3627. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kusunoki M, Yanagi H, Noda M and Yamamura
T: The usefulness of pharmacokinetic modulating chemotherapy (UFT
plus 5FU) in the treatment of unresectable colorectal carcinomas.
Oncol Rep. 6:547–552. 1999.PubMed/NCBI
|
|
33
|
Lin JK, Wang WS, Hsieh RK, et al: Phase II
study of oral tegafur-uracil and folinic acid as first-line therapy
for metastatic colorectal cancer: Taiwan experience. Jpn J Clin
Oncol. 30:510–514. 2000. View Article : Google Scholar
|
|
34
|
Hsiao SC, Lin JF, Chuang MT, Lee YA and Wu
DL: Retrospectively comparative evaluation of the first- and
second-line chemotherapy with campto and oxaliplatin combined with
oral tegafur/uracil (UFT)/leucovorin (LV) in patients with
metastatic colorectal cancer. Int Surg. 94:298–303. 2009.
|