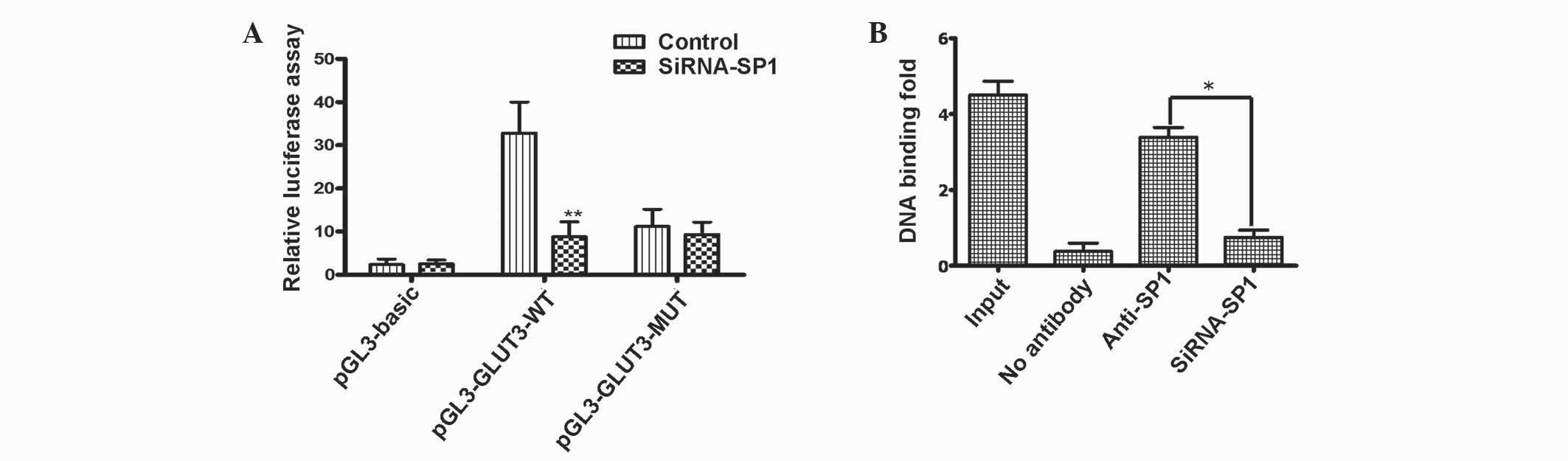
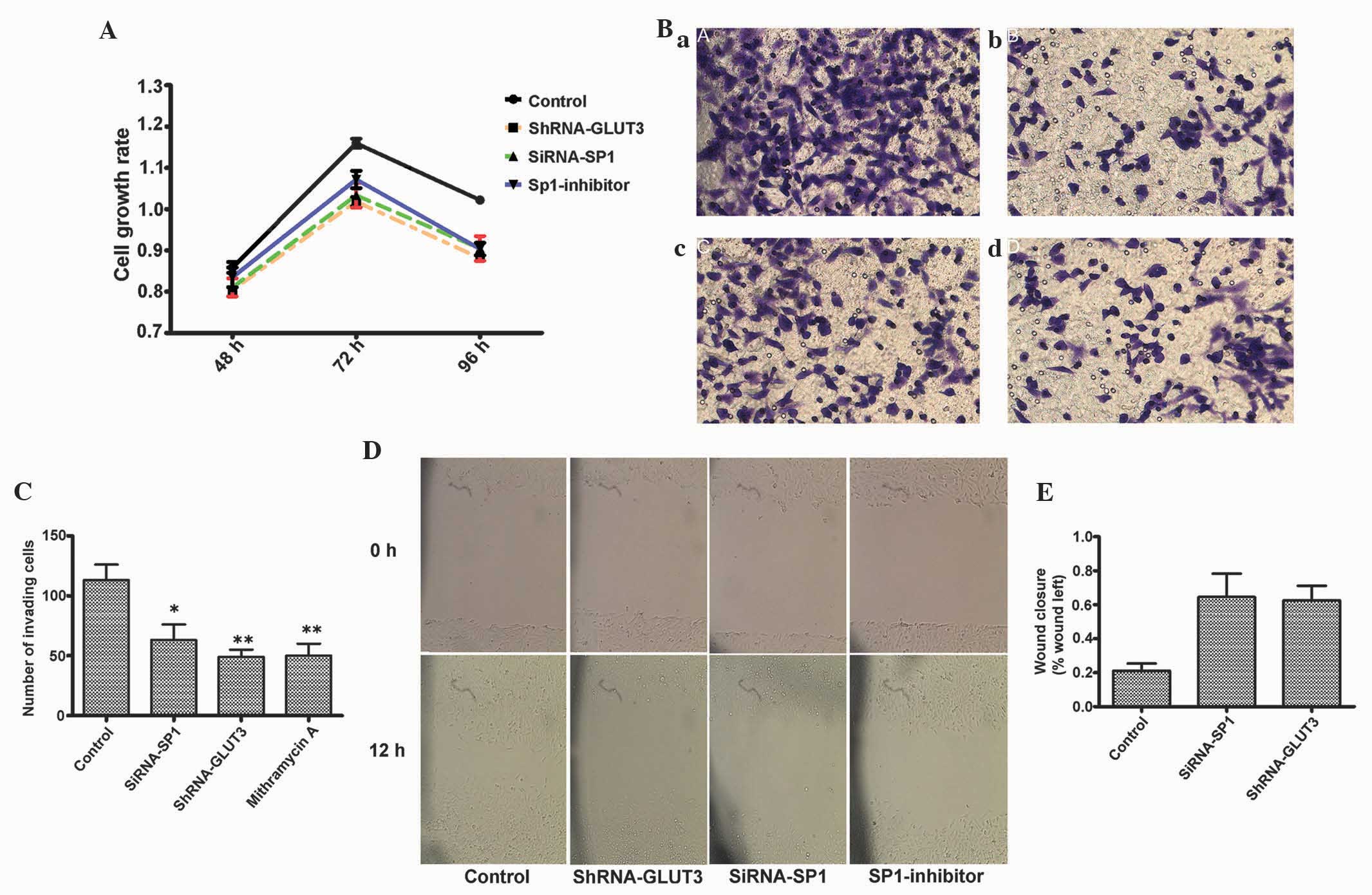
|
1
|
Ahmed R, Oborski MJ, Hwang M, Lieberman FS
and Mountz JM: Malignant gliomas: Current perspectives in
diagnosis, treatment and early response assessment using advanced
quantitative imaging methods. Cancer Manag Res. 6:149–170.
2014.PubMed/NCBI
|
|
2
|
Chung WJ, Lyons SA, Nelson GM, Hamza H,
Gladson CL, Gillespie GY and Sontheimer H: Inhibition of cystine
uptake disrupts the growth of primary brain tumors. J Neurosci.
25:7101–7110. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Flavahan WA, Wu Q, Hitomi M, Rahim N, Kim
Y, Sloan AE, Weil RJ, Nakano I, Sarkaria JN, Stringer BW, et al:
Brain tumor initiating cells adapt to restricted nutrition through
preferential glucose uptake. Nat Neurosci. 16:1373–1382. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zheng J: Energy metabolism of cancer:
Glycolysis versus oxidative phosphorylation (Review). Oncol Lett.
4:1151–1157. 2012.PubMed/NCBI
|
|
5
|
Walker-Samuel S, Ramasawmy R, Torrealdea
F, Rega M, Rajkumar V, Johnson SP, Richardson S, Gonçalves M,
Parkes HG, Arstad E, et al: In vivo imaging of glucose
uptake and metabolism in tumors. Nat Med. 19:1067–1072. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Rodríguez-Enríquez S, Marin-Hernández A,
Gallardo-Pérez JC and Moreno-Sánchez R: Kinetics of transport and
phosphorylation of glucose in cancer cells. J Cell Physiol.
221:552–559. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Boado RJ, Black KL and Pardridge WM: Gene
expression of GLUT3 and GLUT1 glucose transporters in human brain
tumors. Brain Res Mol Brain Res. 27:51–57. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Nagamatsu S, Sawa H, Wakizaka A and
Hoshino T: Expression of facilitative glucose transporter isoforms
in human brain tumors. J Neurochem. 61:2048–2053. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Nishioka T, Oda Y, Seino Y, Yamamoto T,
Inagaki N, Yano H, Imura H, et al: Distribution of the glucose
transporters in human brain tumors. Cancer Res. 52:3972–3979.
1992.PubMed/NCBI
|
|
10
|
Li L and Davie JR: The role of Sp1 and Sp3
in normal and cancer cell biology. Ann Anat. 192:275–283. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Copland JA, Pardini AW, Wood TG, Yin D,
Green A, Bodenburg YH, Urban RJ and Stuart CA: IGF-1 controls GLUT3
expression in muscle via the transcriptional factor Sp1. Biochim
Biophys Acta. 1769:631–640. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Hsu TI, Wang MC, Chen SY, Yeh YM, Su WC,
Chang WC and Hung JJ: Sp1 expression regulates lung tumor
progression. Oncogene. 31:3973–3988. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kou XX, Hao T, Meng Z, Zhou YH and Gan YH:
Acetylated Sp1 inhibits PTEN expression through binding to PTEN
core promoter and recruitment of HDAC1 and promotes cancer cell
migration and invasion. Carcinogenesis. 34:58–67. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Safe S, Imanirad P, Sreevalsan S, Nair V
and Jutooru I: Transcription factor Sp1, also known as specificity
protein 1 as a therapeutic target. Expert Opin Ther Targets.
18:759–769. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Sleiman SF, Langley BC, Basso M, Berlin J,
Xia L, Payappilly JB, Kharel MK, et al: Mithramycin is a
gene-selective Sp1 inhibitor that identifies a biological
intersection between cancer and neurodegeneration. J Neurosci.
31:6858–6870. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Malek A, Núñez LE, Magistri M, Brambilla
L, Jovic S, Carbone GM, Morís F and Catapano CV: Modulation of the
activity of Sp transcription factors by mithramycin analogues as a
new strategy for treatment of metastatic prostate cancer. PLoS One.
7:e351302012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Copland JA, Pardini AW, Wood TG, Yin D,
Green A, Bodenburg YH, Urban RJ and Stuart CA: IGF-1 controls GLUT3
expression in muscle via the transcriptional factor Sp1. Biochim
Biophys Acta. 1769:631–640. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Guan H, Cai J, Zhang N, Wu J, Yuan J, Li J
and Li M: Sp1 is upregulated in human glioma, promotes
MMP-2-mediated cell invasion and predicts poor clinical outcome.
Int J Cancer. 130:593–601. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Garcia-Huerta P, Díaz-Hernandez M,
Delicado EG, Pimentel-Santillana M, Miras-Portugal MT and
Gómez-Villafuertes R: The specificity protein factor Sp1 mediates
transcriptional regulation of P2X7 receptors in the nervous system.
J Biol Chem. 287:44628–44644. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Maher F, Davies-Hill TM, Lysko PG,
Henneberry RC and Simpson IA: Expression of two glucose
transporters, GLUT1 and GLUT3, in cultured cerebellar neurons:
Evidence for neuron-specific expression of GLUT3. Mol Cell
Neurosci. 2:351–360. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Macheda ML, Rogers S and Best JD:
Molecular and cellular regulation of glucose transporter (GLUT)
proteins in cancer. J Cell Physiol. 202:654–662. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Seznec J, Silkenstedt B and Naumann U:
Therapeutic effects of the Sp1 inhibitor mithramycin A in
glioblastoma. J Neurooncol. 101:365–377. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Amann T and Hellerbrand C: GLUT1 as a
therapeutic target in hepatocellular carcinoma. Expert Opin Ther
Targets. 13:1411–1427l. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Blume SW, Snyder RC, Ray R, Thomas S,
Koller CA and Miller DM: Mithramycin inhibits Sp1 binding and
selectively inhibits transcriptional activity of the dihydrofolate
reductase gene in vitro and in vivo. J Clin Invest.
88:1613–1621. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Van Dyke MW and Dervan PB: Chromomycin,
mithramycin and olivomycin binding sites on heterogeneous
deoxyribonucleic acid. Footprinting with (methidiumpropyl-EDTA)
iron (II). Biochemistry. 22:2373–2377. 1983. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Remsing LL, Bahadori HR, Carbone GM,
McGuffie EM, Catapano CV and Rohr J: Inhibition of c-src
transcription by mithramycin: Structure-activity relationships of
biosynthetically produced mithramycin analogues using the c-src
promoter as target. Biochemistry. 42:8313–8324. 2003. View Article : Google Scholar : PubMed/NCBI
|