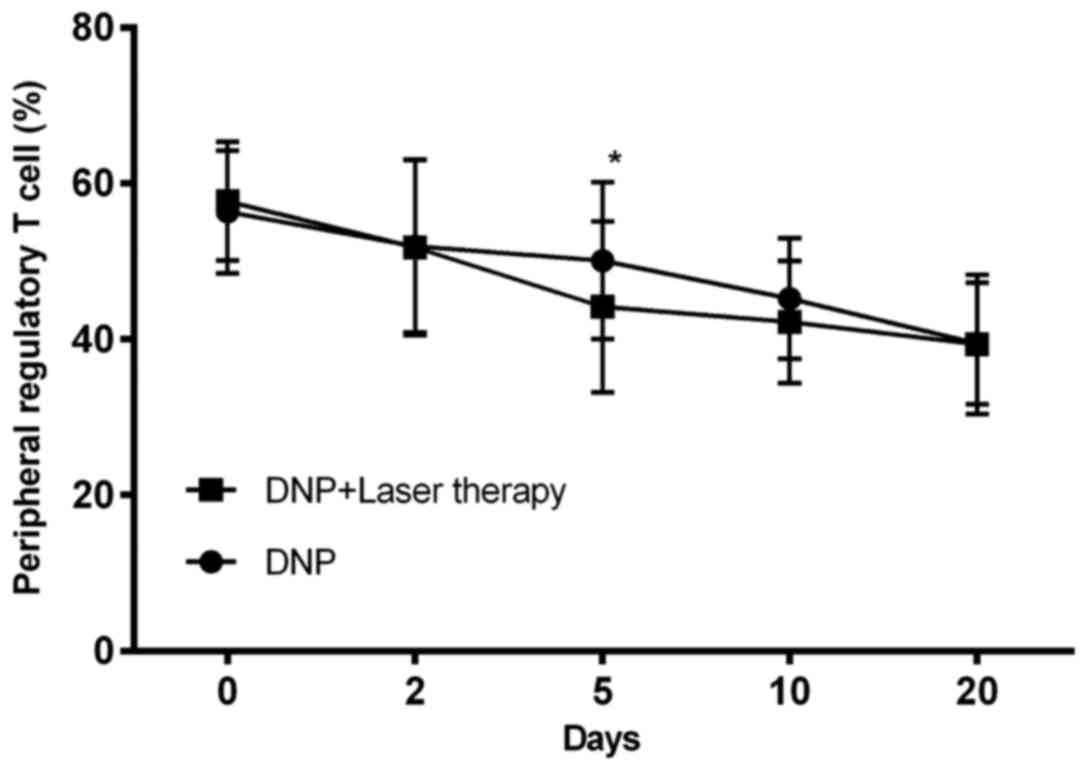
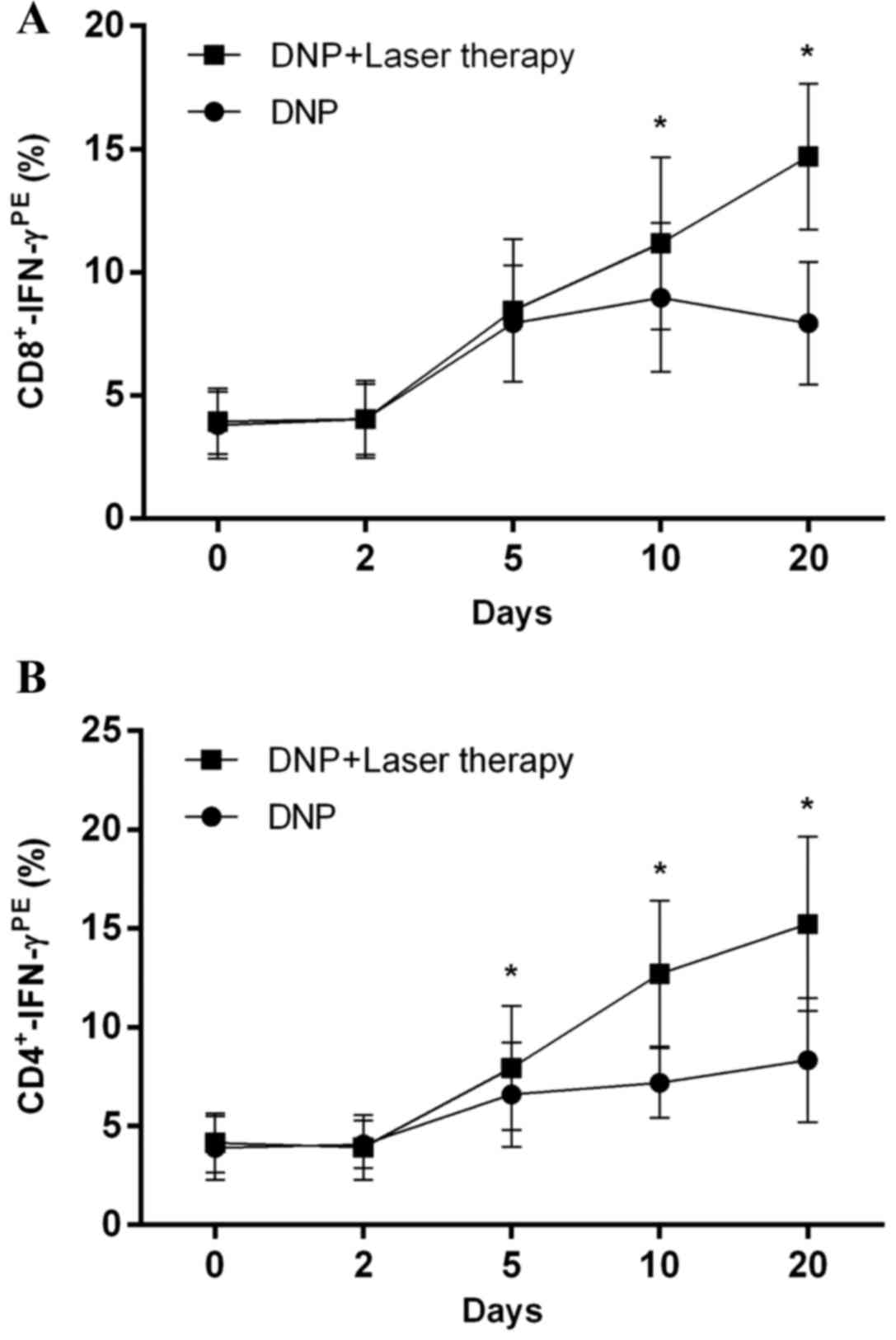
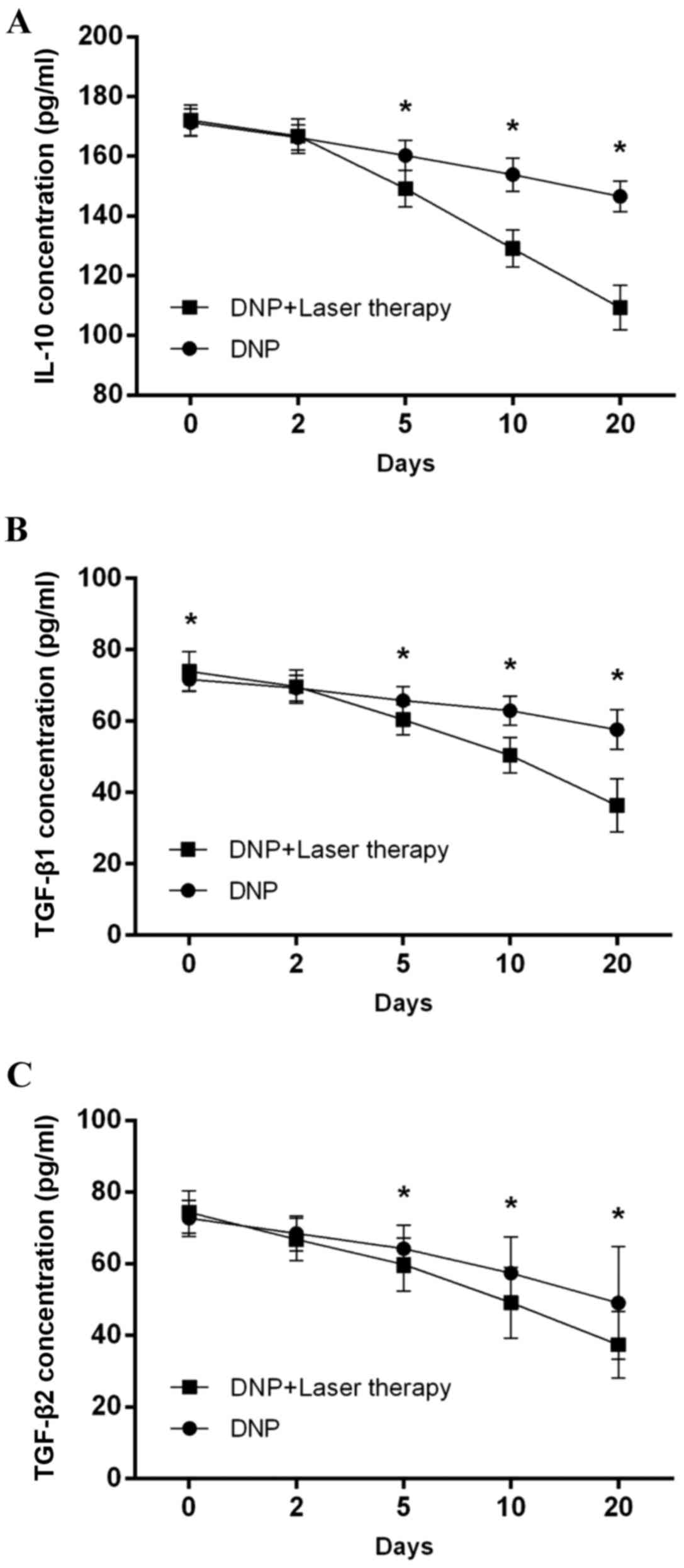
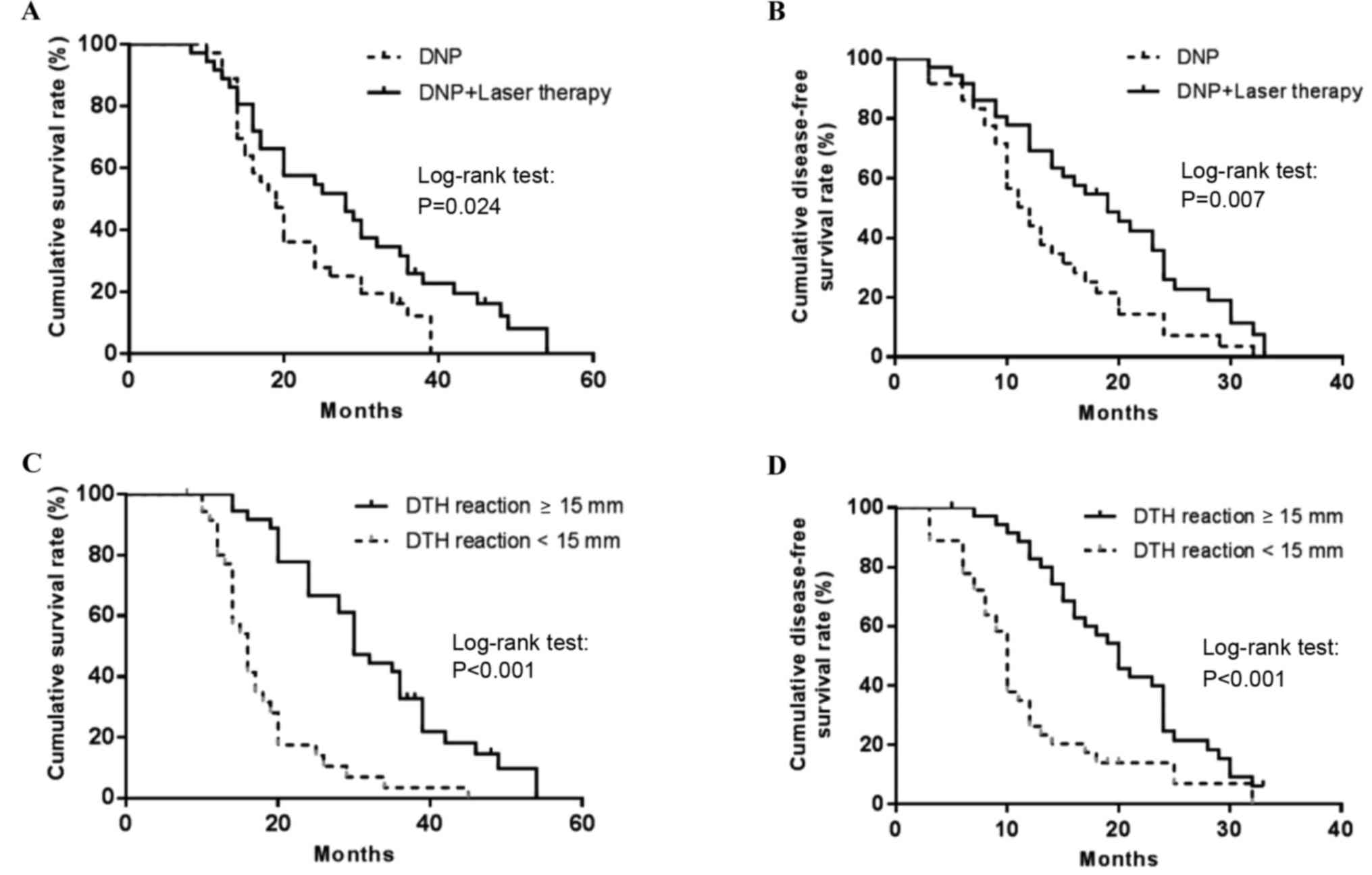
|
1
|
Meier F, Will S, Ellwanger U,
Schlagenhauff B, Schittek B, Rassner G and Garbe C: Metastatic
pathways and time courses in the orderly progression of cutaneous
melanoma. Br J Dermatol. 147:62–70. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Becker JC, Kämpgen E and Bröcker EB:
Classical chemotherapy for metastatic melanoma. Clin Exp Dermatol.
25:503–508. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Garbe C, Peris K, Hauschild A, Saiag P,
Middleton M, Spatz A, Grob JJ, Malvehy J, NewtonBishop J, Stratigos
A, et al: Diagnosis and treatment of melanoma: European consensus
based interdisciplinary guideline. Eur J Cancer. 46:270–283. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Legha SS, Ring S, Eton O, Bedikian A,
Buzaid AC, Plager C and Papadopoulos N: Development of a
biochemotherapy regimen with concurrent administration of
cisplatin, vinblastine, dacarbazine, interferon alfa, and
interleukin-2 for patients with metastatic melanoma. J Clin Oncol.
16:1752–1759. 1998.PubMed/NCBI
|
|
5
|
O'Day SJ, Gammon G, Boasberg PD, Martin
MA, Kristedja TS, Guo M, Stern S, Edwards S, Fournier P, Weisberg
M, et al: Advantages of concurrent biochemotherapy modified by
decrescendo interleukin-2, granulocyte colony-stimulating factor
and tamoxifen for patients with metastatic melanoma. J Clin Oncol.
17:2752–2761. 1999.PubMed/NCBI
|
|
6
|
Middleton MR, Grob JJ, Aaronson N,
Fierlbeck G, Tilgen W, Seiter S, Gore M, Aamdal S, Cebon J, Coates
A, et al: Randomized phase III study of temozolomide versus
dacarbazine in the treatment of patients with advanced metastatic
malignant melanoma. J Clin Oncol. 18:158–166. 2000.PubMed/NCBI
|
|
7
|
Atkins M: The treatment of metastatic
melanoma with chemotherapy and biologics. Curr Opin Oncol.
9:205–213. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Legha S, Ring S, Papadopoulos N, Raber M
and Benjamin RS: A phase II trial of taxol in metastatic melanoma.
Cancer. 65:2478–2481. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Bedikian A, Weiss G, Legha S, Burris HA
III, Eckardt JR, Jenkins J, Eton O, Buzaid AC, Smetzer L, Von Hoff
DD, et al: Phase II trial of docetaxel in patients with advanced
cutaneous malignant melanoma previously untreated with
chemotherapy. J Clin Oncol. 13:2895–2899. 1995.PubMed/NCBI
|
|
10
|
Avril M, Aamdal S, Grob J, Hauschild A,
Mohr P, Bonerandi JJ, Weichenthal M, Neuber K, Bieber T, Gilde K,
et al: Fotemustine compared with dacarbazine in patients with
disseminated malignant melanoma: A phase III study. J Clin Oncol.
22:1118–1125. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Berd D: A tale of two pities: Autologous
melanoma vaccines on the brink. Hum Vaccin Immunother. 8:1146–1151.
2012. View
Article : Google Scholar : PubMed/NCBI
|
|
12
|
Calalona WJ, Taylor PT, Rabson AS and
Chretien PB: A method for dinifrochlorobengene contact
sensitigafron: A clinico pathological study. N Engl J Med.
286:399–402. 1972. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Fujisawa Y, Nabekura T, Nakao T, Nakamura
Y, Takahashi T, Kawachi Y, Otsuka F and Onodera M: The induction of
tumor-specific CD4+ T cells via major histocompatibility
complex class II is required to gain optimal anti-tumor immunity
against B16 melanoma cell line in tumor immunotherapy using
dendritic cells. Exp Dermatol. 18:396–403. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Terabe M and Berzofsky JA:
Immunoregulatory T cells in tumor immunity. Curr Opin Immunol.
16:157–162. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Somasundaram R, Jacob L, Swoboda R, Caputo
L, Song H, Basak S, Monos D, Peritt D, Marincola F, Cai D, et al:
Inhibition of cytolytic T lymphocyte proliferation by autologous
CD4+/CD25+ regulatory T cells in a colorectal
carcinoma patient is mediated by transforming growth factor-beta.
Cancer Res. 62:5267–5272. 2002.PubMed/NCBI
|
|
16
|
Jacobs JF, Nierkens S, Figdor CG, de Vries
IJ and Adema GJ: Regulatory T cells in melanoma: The final hurdle
towards effective immunotherapy? Lancet Oncol. 13:e32–e42. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wolf AM, Wolf D, M Gastl G Steurer,
Gunsilius E and Grubeck-Loebenstein B: Increase of regulatory T
cell in the peripheral blood of cancer patients. Clin Cancer Res.
9:606–612. 2003.PubMed/NCBI
|
|
18
|
Read S, Malmström V and Powrie F:
Cytotoxic T lymphocyte-associated antigen 4 plays an essential role
in the function of CD25(+)CD4(+) regulatory cells that control
intestinal inflammation. J Exp Med. 192:295–302. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Correll A, Tuettenberg A, Becker C and
Jonuleit H: Increased regulatory T-cell frequencies in patients
with advanced melanoma correlate with a generally impaired T-cell
responsiveness and are restored after dendritic cell-based
vaccination. Exp Dermatol. 19:e213–e221. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ladányi A, Mohos A, Somlai B, Liszkay G,
Gilde K, Fejos Z, Gaudi I and Tímár J: FOXP3+ cell
density in primary tumor has no prognostic impact in patients with
cutaneous malignant melanoma. Pathol Oncol Res. 16:303–309. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lagouros E, Salomao D, Thorland E, Hodge
DO, Vile R and Pulido JS: Infiltrative T regulatory cells in
enucleated uveal melanomas. Trans Am Ophthalmol Soc. 107:223–228.
2009.PubMed/NCBI
|
|
22
|
Mougiakakos D, Johansson CC, Trocme E,
AllEricsson C, Economou MA, Larsson O, Seregard S and Kiessling R:
Intratumoral forkhead boxP3-positive regulatory T cells predict
poor survival in cyclooxygenase-2-positive uveal melanoma. Cancer.
116:2224–2233. 2010.PubMed/NCBI
|
|
23
|
Mourmouras V, Fimiani M, Rubegni P,
Epistolato MC, Malagnino V, Cardone C, Cosci E, Nisi MC and Miracco
C: Evaluation of tumour-infiltrating
CD4+CD25+FOXP3+ regulatory T cells
in human cutaneous benign and atypical naevi, melanomas and
melanoma metastases. Br J Dermatol. 157:531–539. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Edge SB and Cancer A.J.C.O.: AJCC cancer
staging handbook: From the AJCC cancer staging manual.
Springer-Verlag; New York: 2010
|
|
25
|
Karnofsky DA, Abelmann WH, Craver LF and
Burchenal JH: The use of the nitrogen mustards in the palliative
treatment of carcinoma. With particular reference to bronchogenic
carcinoma. Cancer. 1:634–656. 1948. View Article : Google Scholar
|
|
26
|
Eisenhauer EA, Therasse P, Bogaerts J,
Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S,
Mooney M, et al: New response evaluation criteria in solid tumours:
Revised RECIST guideline (version 1.1). Eur J Cancer. 45:228–247.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
U.S. Department of Health and Human
Services, National Institutes of Health, National Cancer Institute,
. Common Terminology Criteria for Adverse Events (CTCAE) Version
4.0. Published 28 May 2009 (v4.03: June 14, 2010); cited 14 January
2014. Available from. http://evs.nci.nih.gov/ftp1/CTCAE/CTCAE_4.03_2010-2006-14_QuickReference_5x7.pdf
|
|
28
|
Trcka J, Kämpgen E, Becker JC, Schwaaf A
and Bröcker EB: Immune chemotherapy of malignant melanoma. Epifocal
administration of dinitrochlorobenzene (DNCB) combined with
systemic chemotherapy with dacarbazine (DTIC). Hautarzt. 49:17–22.
1998.(In German). View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Calalona WJ, Taylor PT, Rabson AS and
Chretien PB: A method for dinitrochlorobenzene contact
sensitization. A clinicopathological study. N Engl J Med.
286:399–402. 1972. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Barth A, Wanek LA and Morton DL:
Prognostic factors in 1,521 melanoma patients with distant
metastasis. J Am Coll Surg. 181:193–201. 1995.PubMed/NCBI
|
|
31
|
Li X, Min M, Gu Y and Du N: Laser
immunotherapy: Concept, possible mechanism, clinical applications
and recent experimental results. IEEE J Selected Topics Quantum
Electronics. 18:1434–1438. 2012. View Article : Google Scholar
|
|
32
|
Li X, Naylor MF, Le H, Nordquist RE,
Teague TK, Howard CA, Murray C and Chen WR: Clinical effects of in
situ photoimmunotherapy on late-stage melanoma patients: A
preliminary study. Cancer Biol Ther. 10:1081–1087. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Li X, Le H, Wolf RF, Chen VA, Sarkar A,
Nordquist RE, Ferguson H, Liu H and Chen WR: Long-term effect on
EMT6 tumors in mice induced by combination of laser immunotherapy
and surgery. Integrative Cancer Ther. 10:368–373. 2011. View Article : Google Scholar
|
|
34
|
Terheyden P, Kortüm AK, Schulze HJ, Durani
B, Remling R, Mauch C, Junghans V, Schadendorf D, Beiteke U, Jünger
M, Becker JC and Bröcker EB: Chemoimmunotherapy for cutaneous
melanoma with dacarbazine and epifocal contact sensitizers: Results
of a nationwide survey of the German Dermatologic Co-operative
Oncology Group. J Cancer Res Clin Oncol. 133:437–444. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Quillien V, Lesimple T and Toujas L:
Vaccinal cell therapy in melanoma. Bull Cancer. 90:722–733.
2003.(In French). PubMed/NCBI
|
|
36
|
Berd D, Sato T and Mastrangelo MJ: Effect
of the dose and composition of an autologous hapten-modified
melanoma vaccine on the development of delayed-type
hypersensitivity responses. Cancer Immunol Immunother. 51:320–326.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Ridolfi L, Petrini M, Fiammenqhi L,
Granato AM, Ancarani V, Pancisi E, Brolli C, Selva M, Scarpi E,
Valmorri L, et al: Dendritic cell-based vaccine in advanced
melanoma: Update of clinical outcome. Melanoma Res. 21:524–529.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Stamell EF, Wolchok JD, Gnjatic S, Lee NY
and Brownell I: The abscopal effect associated with a systemic
anti-melanoma immune response. Int J Radiat Oncol Biol Phys.
85:293–295. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Wack C, Kirst A, Becker JC, Lutz WK,
Bröcker EB and Fischer WH: Chemoimmunotherapy for melanoma with
dacarbazine and 2,4-dinitrochlorobenzene elicits a specific T
cell-dependent immune response. Cancer Immunol Immunother.
51:431–439. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Kumamoto T, Huang EK, Paek HJ, Morita A,
Matsue H, Valentini RF and Takashima A: Induction of tumor-specific
protective immunity by in situ Langerhans cell vaccine. Nat
Biotechnol. 20:64–69. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Mahipal A, Terai M, Berd D, Chervoneva I,
Patel K, Mastrangelo MJ and Sato T: Tumor-derived interleukin-10 as
a prognostic factor in stage III patients undergoing adjuvant
treatment with an autologous melanoma cell vaccine. Cancer Immunol
Immunother. 60:1039–1045. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Postow MA, Callahan MK, Barker CA, Yamada
Y, Yuan J, Kitano S, Mu Z, Rasalan T, Adamow M, Ritter E, et al:
Immunologic correlates of the abscopal effect in a patient with
melanoma. N Engl J Med. 366:925–931. 2012. View Article : Google Scholar : PubMed/NCBI
|