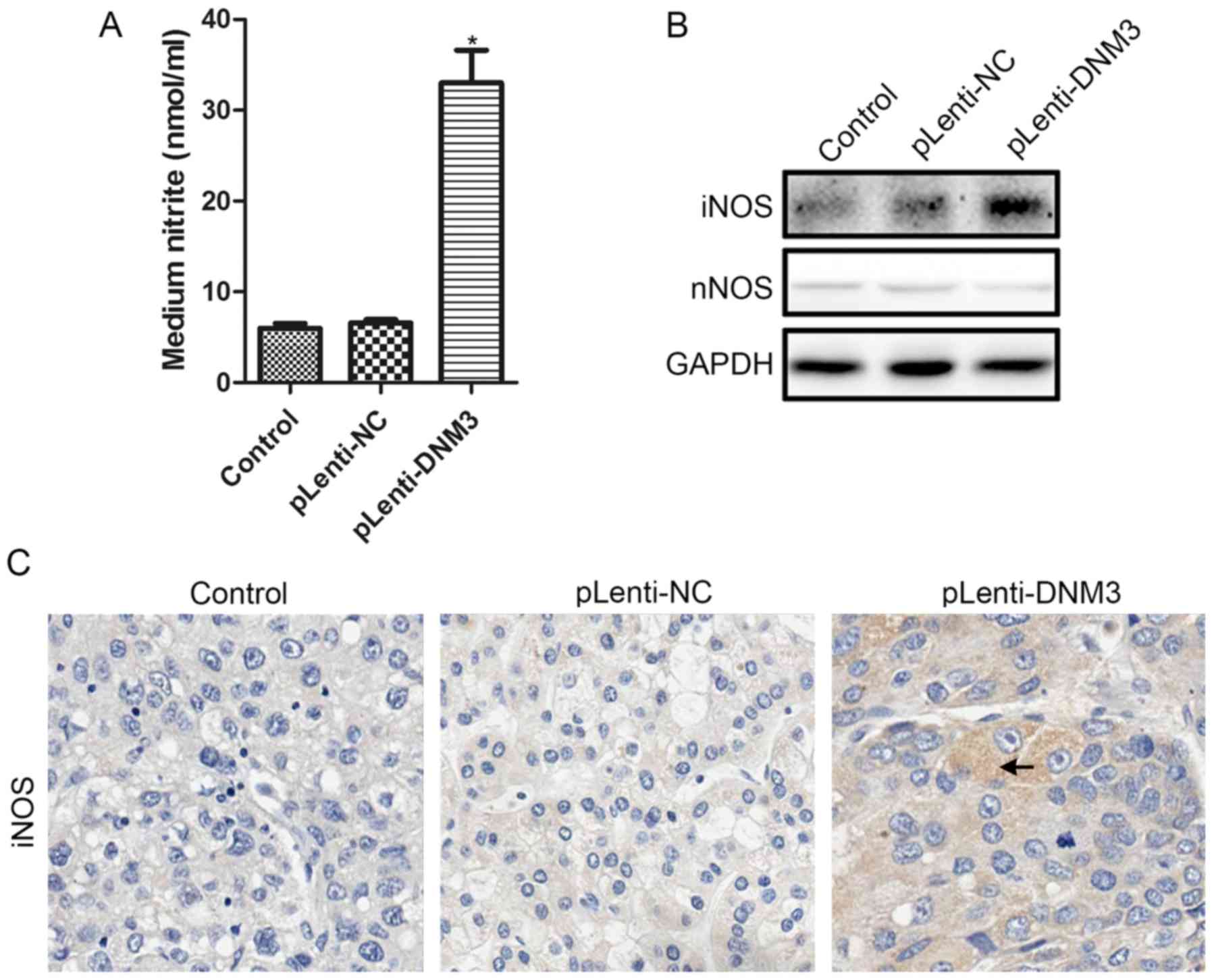
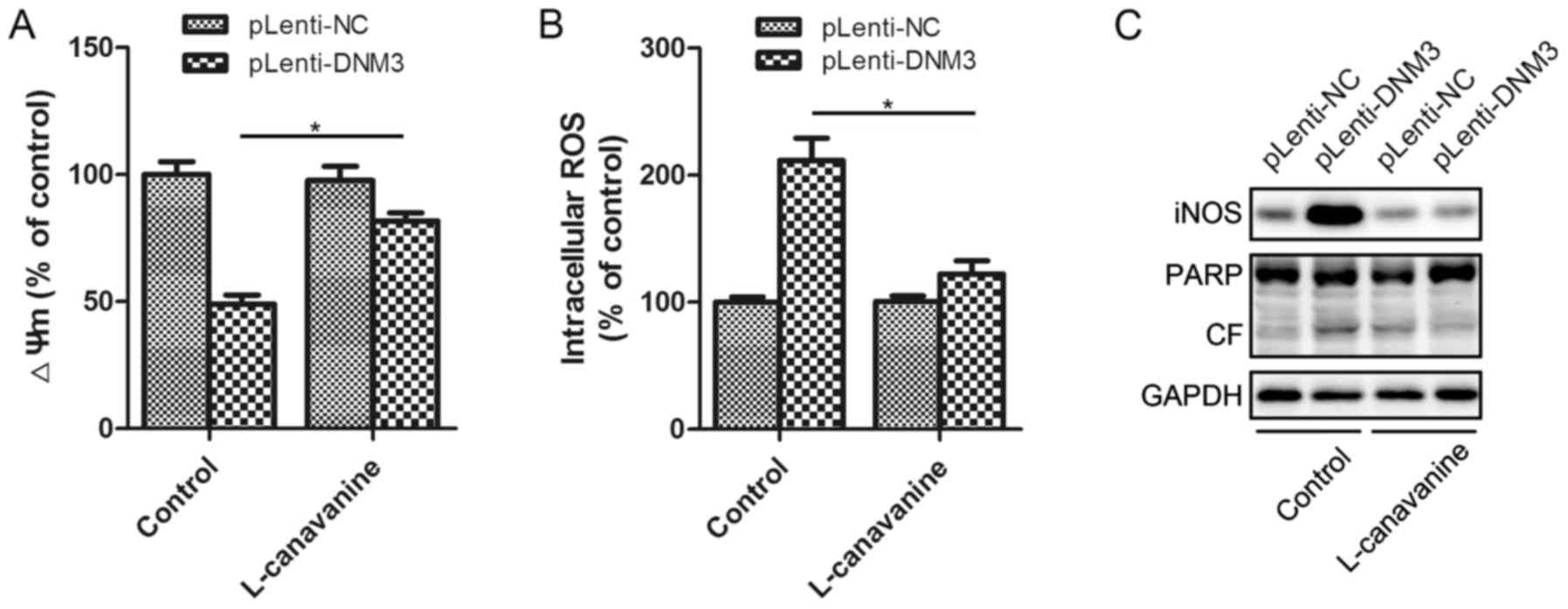
|
1
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Earl TM and Chapman WC: Hepatocellular
carcinoma: Resection versus transplantation. Semin Liver Dis.
33:282–292. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Cusnir M and Patt YZ: Novel systemic
therapy options for hepatocellular carcinoma. Cancer J. 10:97–103.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Yu MC and Yuan JM: Environmental factors
and risk for hepatocellular carcinoma. Gastroenterology. 127 5
Suppl 1:S72–S78. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Shiraha H, Yamamoto K and Namba M: Human
hepatocyte carcinogenesis (Review). Int J Oncol. 42:1133–1138.
2013.PubMed/NCBI
|
|
6
|
El-Serag HB and Rudolph KL: Hepatocellular
carcinoma: Epidemiology and molecular carcinogenesis.
Gastroenterology. 132:2557–2576. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Heymann JA and Hinshaw JE: Dynamins at a
glance. J Cell Sci. 122:3427–3431. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Hinshaw JE: Dynamin and its role in
membrane fission. Annu Rev Cell Dev Biol. 16:483–519. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Schmid SL and Frolov VA: Dynamin:
Functional design of a membrane fission catalyst. Annu Rev Cell Dev
Biol. 27:79–105. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Hyndman KA, Musall JB, Xue J and Pollock
JS: Dynamin activates NO production in rat renal inner medullary
collecting ducts via protein-protein interaction with NOS1. Am J
Physiol Renal Physiol. 301:F118–F124. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Cao S, Yao J and Shah V: The proline-rich
domain of dynamin-2 is responsible for dynamin-dependent in vitro
potentiation of endothelial nitric-oxide synthase activity via
selective effects on reductase domain function. J Biol Chem.
278:5894–5901. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kang-Decker N, Cao S, Chatterjee S, Yao J,
Egan LJ, Semela D, Mukhopadhyay D and Shah V: Nitric oxide promotes
endothelial cell survival signaling through S-nitrosylation and
activation of dynamin-2. J Cell Sci. 120:492–501. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Shen J, Wang S, Zhang YJ, Kappil M, Wu HC,
Kibriya MG, Wang Q, Jasmine F, Ahsan H, Lee PH, et al: Genome-wide
DNA methylation profiles in hepatocellular carcinoma. Hepatology.
55:1799–1808. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Inokawa Y, Nomoto S, Hishida M, Hayashi M,
Kanda M, Nishikawa Y, Takeda S, Fujiwara M, Koike M, Sugimoto H, et
al: Dynamin 3: A new candidate tumor suppressor gene in
hepatocellular carcinoma detected by triple combination array
analysis. Onco Targets Ther. 6:1417–1424. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Nie X, Song S, Zhang L, Qiu Z, Shi S, Liu
Y, Yao L and Zhu D: 15-Hydroxyeicosatetraenoic acid (15-HETE)
protects pulmonary artery smooth muscle cells from apoptosis via
inducible nitric oxide synthase (iNOS) pathway. Prostaglandins
Other Lipid Mediat. 97:50–59. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yin XY, Jiang JM, Liu JY and Zhu JR:
Effects of endogenous nitric oxide induced by 5-fluorouracil and
L-Arg on liver carcinoma in nude mice. World J Gastroenterol.
13:6249–6253. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Lemasters JJ: V. Necrapoptosis and the
mitochondrial permeability transition: Shared pathways to necrosis
and apoptosis. Am J Physiol. 276:G1–G6. 1999.PubMed/NCBI
|
|
19
|
Pastorino JG and Hoek JB: Ethanol
potentiates tumor necrosis factor-alpha cytotoxicity in hepatoma
cells and primary rat hepatocytes by promoting induction of the
mitochondrial permeability transition. Hepatology. 31:1141–1152.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Circu ML and Aw TY: Reactive oxygen
species, cellular redox systems, and apoptosis. Free Radic Biol
Med. 48:749–762. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lala PK and Chakraborty C: Role of nitric
oxide in carcinogenesis and tumour progression. Lancet Oncol.
2:149–156. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Soliman MK, Mazzio E and Soliman KF:
Levodopa modulating effects of inducible nitric oxide synthase and
reactive oxygen species in glioma cells. Life Sci. 72:185–198.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Vannini F, Kashfi K and Nath N: The dual
role of iNOS in cancer. Redox Biol. 6:334–343. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Bonavida B and Baritaki S: Dual role of NO
donors in the reversal of tumor cell resistance and EMT:
Downregulation of the NF-κB/Snail/YY1/RKIP circuitry. Nitric Oxide.
24:1–7. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kim SF, Huri DA and Snyder SH: Inducible
nitric oxide synthase binds, S-nitrosylates, and activates
cyclooxygenase-2. Science. 310:1966–1970. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lepore DA: Nitric oxide
synthase-independent generation of nitric oxide in muscle
ischemia-reperfusion injury. Nitric Oxide. 4:541–545. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Moncada S, Palmer RM and Higgs EA: Nitric
oxide: Physiology, pathophysiology, and pharmacology. Pharmacol
Rev. 43:109–142. 1991.PubMed/NCBI
|
|
28
|
Cohen RA and Vanhoutte PM:
Endothelium-dependent hyperpolarization. Beyond nitric oxide and
cyclic GMP. Circulation. 92:3337–3349. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Panda SP, Gao YT, Roman LJ, Martásek P,
Salerno JC and Masters BS: The role of a conserved serine residue
within hydrogen bonding distance of FAD in redox properties and the
modulation of catalysis by Ca2+/calmodulin of constitutive
nitric-oxide synthases. J Biol Chem. 281:34246–34257. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Bredt DS, Hwang PM, Glatt CE, Lowenstein
C, Reed RR and Snyder SH: Cloned and expressed nitric oxide
synthase structurally resembles cytochrome P-450 reductase. Nature.
351:714–718. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Dominiczak AF and Bohr DF: Nitric oxide
and its putative role in hypertension. Hypertension. 25:1202–1211.
1995. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Crane BR, Arvai AS, Gachhui R, Wu C, Ghosh
DK, Getzoff ED, Stuehr DJ and Tainer JA: The structure of nitric
oxide synthase oxygenase domain and inhibitor complexes. Science.
278:425–431. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Schulz R and Triggle CR: Role of NO in
vascular smooth muscle and cardiac muscle function. Trends
Pharmacol Sci. 15:255–259. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Crompton M: The mitochondrial permeability
transition pore and its role in cell death. Biochem J. 341:233–249.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Higuchi Y: Chromosomal DNA fragmentation
in apoptosis and necrosis induced by oxidative stress. Biochem
Pharmacol. 66:1527–1535. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Bartosz G: Reactive oxygen species:
Destroyers or messengers? Biochem Pharmacol. 77:1303–1315. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Buttke TM and Sandstrom PA: Oxidative
stress as a mediator of apoptosis. Immunol Today. 15:7–10. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Sabharwal SS and Schumacker PT:
Mitochondrial ROS in cancer: Initiators, amplifiers or an Achilles'
heel? Nat Rev Cancer. 14:709–721. 2014. View Article : Google Scholar : PubMed/NCBI
|