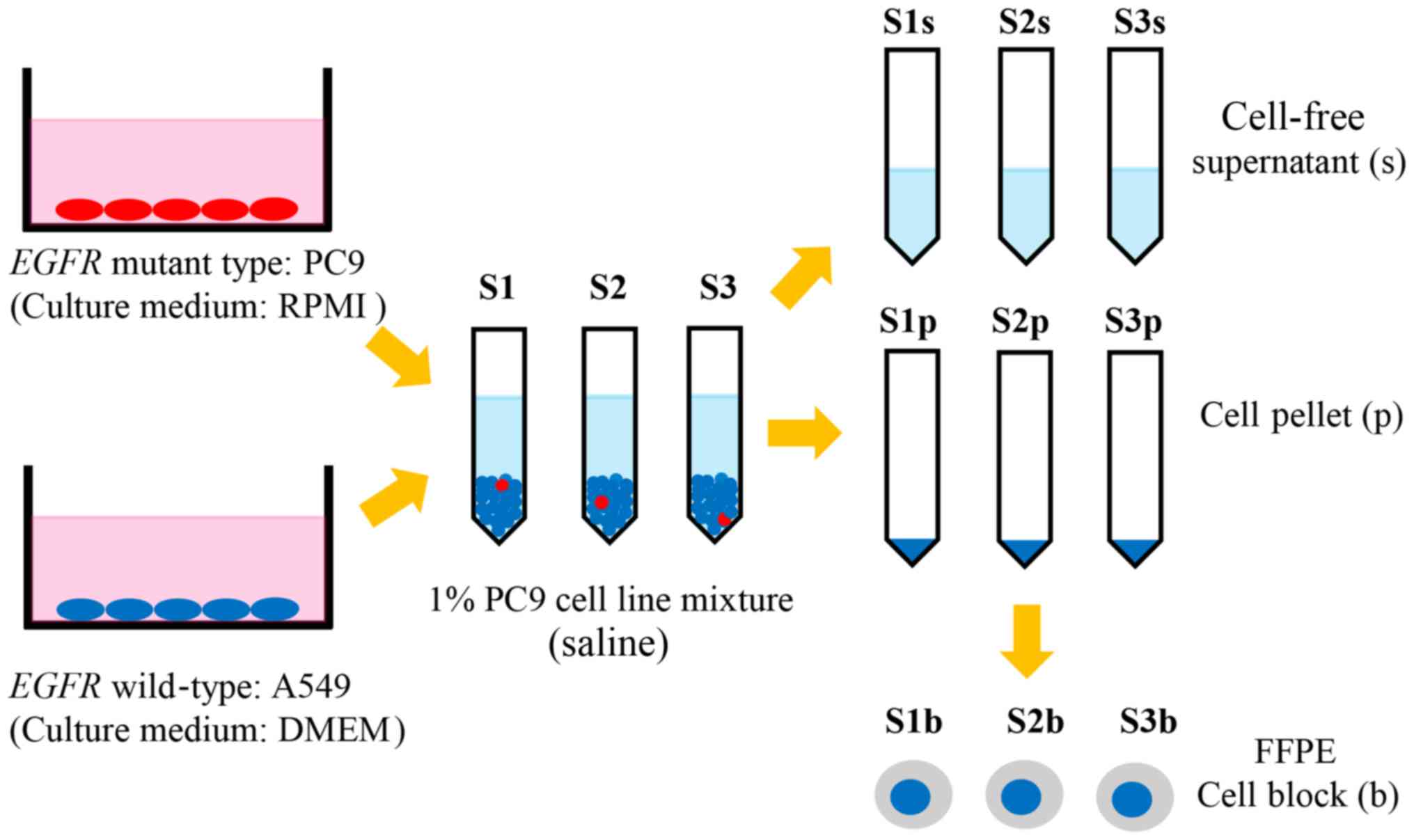
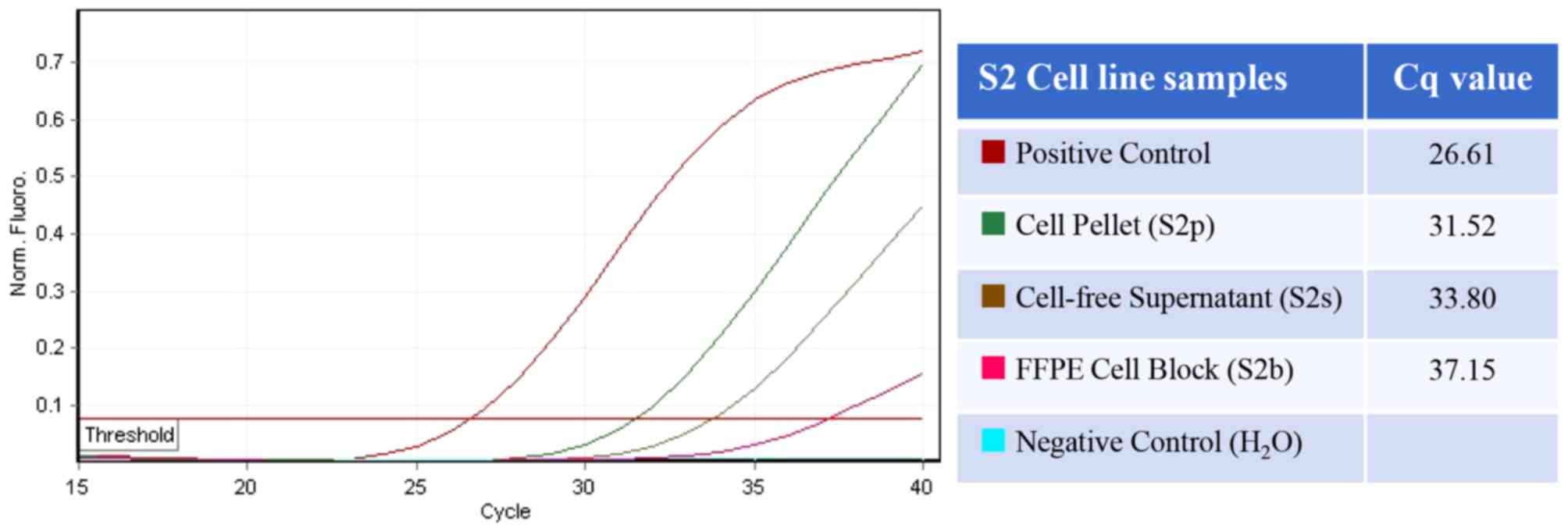
|
1
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global Cancer Statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Maemondo M, Inoue A, Kobayashi K, Sugawara
S, Oizumi S, Isobe H, Gemma A, Harada M, Yoshizawa H, Kinoshita I,
et al: Gefitinib or chemotherapy for non-small-cell lung cancer
with mutated EGFR. N Engl J Med. 362:2380–2388. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Mitsudomi T, Morita S, Yatabe Y, Negoro S,
Okamoto I, Tsurutani J, Seto T, Satouchi M, Tada H, Hirashima T, et
al: Gefitinib versus cisplatin plus docetaxel in patients with
non-small-cell lung cancer harbouring mutations of the epidermal
growth factor receptor (WJTOG3405): An open label, randomised phase
3 trial. Lancet Oncol. 11:121–128. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zhou C, Wu YL, Chen G, Feng J, Liu XQ,
Wang C, Zhang S, Wang J, Zhou S, Ren S, et al: Erlotinib versus
chemotherapy as first-line treatment for patients with advanced
EGFR mutation-positive non-small-cell lung cancer (OPTIMAL,
CTONG-0802): A multicentre, open-label, randomised, phase 3 study.
Lancet Oncol. 12:735–742. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Rosell R, Carcereny E, Gervais R,
Vergnenegre A, Massuti B, Felip E, Palmero R, Garcia-Gomez R,
Pallares C, Sanchez JM, et al: Erlotinib versus standard
chemotherapy as first-line treatment for European patients with
advanced EGFR mutation-positive non-small-cell lung cancer
(EURTAC): A multicentre, open-label, randomised phase 3 trial.
Lancet Oncol. 13:239–246. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wu YL, Zhou C, Hu CP, Feng J, Lu S, Huang
Y, Li W, Hou M, Shi JH, Lee KY, et al: Afatinib versus cisplatin
plus gemcitabine for first-line treatment of Asian patients with
advanced non-small-cell lung cancer harbouring EGFR mutations
(LUX-Lung 6): An open-label, randomised phase 3 trial. Lancet
Oncol. 15:213–222. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Yang JC, Wu YL, Schuler M, Sebastian M,
Popat S, Yamamoto N, Zhou C, Hu CP, O'Byrne K, Feng J, et al:
Afatinib versus cisplatin-based chemotherapy for EGFR
mutation-positive lung adenocarcinoma (LUX-Lung 3 and LUX-Lung 6):
Analysis of overall survival data from two randomised, phase 3
trials. Lancet Oncol. 16:141–151. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Fukuoka M, Wu YL, Thongprasert S,
Sunpaweravong P, Leong SS, Sriuranpong V, Chao TY, Nakagawa K, Chu
DT, Saijo N, et al: Biomarker analyses and final overall survival
results from a phase III, randomized, open-label, first-line study
of gefitinib versus carboplatin/paclitaxel in clinically selected
patients with advanced non-small-cell lung cancer in Asia (IPASS).
J Clin Oncol. 29:2866–2874. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Naidoo J, Sima CS, Rodriguez K, Busby N,
Nafa K, Ladanyi M, Riely GJ, Kris MG, Arcila ME and Yu HA:
Epidermal growth factor receptor exon 20 insertions in advanced
lung adenocarcinomas: Clinical outcomes and response to erlotinib.
Cancer. 121:3212–3220. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lee VH, Tin VP, Choy TS, Lam KO, Choi CW,
Chung LP, Tsang JW, Ho PP, Leung DK, Ma ES, et al: Association of
Exon 19 and 21 EGFR mutation patterns with treatment outcome after
first-line tyrosine kinase inhibitor in metastatic non-small-cell
lung cancer. J Thorac Oncol. 8:1148–1155. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Mitsudomi T, Kosaka T and Yatabe Y:
Biological and clinical implications of EGFR mutations in lung
cancer. Int J Clin Oncol. 11:190–198. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kuiper JL, Heideman DA, Thunnissen E, Paul
MA, van Wijk AW, Postmus PE and Smit EF: Incidence of T790M
mutation in (sequential) rebiopsies in EGFR-mutated NSCLC-patients.
Lung Cancer. 85:19–24. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Wang S, Cang S and Liu D: Third-generation
inhibitors targeting EGFR T790M mutation in advanced non-small cell
lung cancer. J Hematol Oncol. 9:342016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Lindeman NI, Cagle PT, Beasley MB, Chitale
DA, Dacic S, Giaccone G, Jenkins RB, Kwiatkowski DJ, Saldivar JS,
Squire J, et al: Molecular testing guideline for selection of lung
cancer patients for EGFR and ALK tyrosine kinase inhibitors
guideline from the College of American Pathologists, International
Association for the Study of Lung Cancer, and Association for
Molecular Pathology. J Mol Diagn. 15:415–453. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ellison G, Zhu GS, Moulis A, Dearden S,
Speake G and McCormack R: EGFR mutation testing in lung cancer: A
review of available methods and their use for analysis of tumour
tissue and cytology samples. J Clin Pathol. 66:79–89. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Goto K, Satouchi M, Ishii G, Nishio K,
Hagiwara K, Mitsudomi T, Whiteley J, Donald E, McCormack R and Todo
T: An evaluation study of EGFR mutation tests utilized for
non-small-cell lung cancer in the diagnostic setting. Ann Oncol.
23:2914–2919. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kimura H, Fujiwara Y, Sone T, Kunitoh H,
Tamura T, Kasahara K and Nishio K: High sensitivity detection of
epidermal growth factor receptor mutations in the pleural effusion
of non-small cell lung cancer patients. Cancer Sci. 97:642–648.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Lin J, Gu Y, Du R, Deng M, Lu Y and Ding
Y: Detection of EGFR mutation in supernatant, cell pellets of
pleural effusion and tumor tissues from non-small cell lung cancer
patients by high resolution melting analysis and sequencing. Int J
Clin Exp Pathol. 7:8813–8822. 2014.PubMed/NCBI
|
|
19
|
Zhang X, Zhao Y, Wang M, Yap WS and Chang
AY: Detection and comparison of epidermal growth factor receptor
mutations in cells and fluid of malignant pleural effusion in
non-small cell lung cancer. Lung Cancer. 60:175–182. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Yamaguchi F, Kugawa S, Tateno H, Kokubu F
and Fukuchi K: Analysis of EGFR, KRAS and P53 mutations in lung
cancer using cells in the curette lavage fluid obtained by
bronchoscopy. Lung Cancer. 78:201–206. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Sakamoto T, Kodani M, Takata M, Chikumi H,
Nakamoto M, Nishii-Ito S, Ueda Y, Izumi H, Makino H, Touge H, et
al: A novel point-of-care system for high-speed real-time
polymerase chain reaction testing for epidermal growth factor
receptor mutations in bronchial lavage fluids after transbronchial
biopsy in patients with non-small cell lung cancer. Int J Oncol.
46:1473–1480. 2015.PubMed/NCBI
|
|
22
|
Asaka S, Yoshizawa A, Matsuda K, Yamaguchi
A, Yamamoto H, Shiina T, Nakata R, Ogawa K, Zhang M and Honda T: A
novel, rapid point-of-care test for lung cancer patients to detect
epidermal growth factor receptor gene mutations by using real-time
droplet-PCR and fresh liquid cytology specimens. Oncol Rep.
37:1020–1026. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Syed YY: Therascreen® EGFR RGQ
PCR Kit: A companion diagnostic for afatinib and gefitinib in
non-small cell lung cancer. Mol Diagn Ther. 20:191–198. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Vallée A, Le Loupp AG and Denis MG:
Efficiency of the Therascreen® RGQ PCR kit for the
detection of EGFR mutations in non-small cell lung carcinomas. Clin
Chim Acta. 429:8–11. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Lozano MD, Zulueta JJ, Echeveste JI,
Gúrpide A, Seijo LM, Martín-Algarra S, Del Barrio A, Pio R, Idoate
MA, Labiano T and Perez-Gracia JL: Assessment of epidermal growth
factor receptor and K-ras mutation status in cytological stained
smears of non-small cell lung cancer patients: Correlation with
clinical outcomes. Oncologist. 16:877–885. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Reynolds JP, Tubbs RR, Minca EC, MacNamara
S, Almeida FA, Ma PC, Pennell NA and Cicenia JC: EGFR mutational
genotyping of liquid based cytology samples obtained via fine
needle aspiration (FNA) at endobronchial ultrasound of non-small
cell lung cancer (NSCLC). Lung Cancer. 86:158–163. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Dedhia P, Tarale S, Dhongde G, Khadapkar R
and Das B: Evaluation of DNA extraction methods and real time PCR
optimization on formalin-fixed paraffin-embedded tissues. Asian Pac
J Cancer Prev. 8:55–59. 2007.PubMed/NCBI
|
|
28
|
Gailey MP, Stence AA, Jensen CS and Ma DQ:
Multiplatform comparison of molecular oncology tests performed on
cytology specimens and formalin-fixed, paraffin-embedded tissue.
Cancer Cytopathol. 123:30–39. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Harada S, Agosto-Arroyo E, Levesque JA,
Alston E, Janowski KM, Coshatt GM and Eltoum IA: Poor cell block
adequacy rate for molecular testing improved with the addition of
Diff-Quik-stained smears: Need for better cell block processing.
Cancer Cytopathol. 123:480–487. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Xu H, Sun W, Zhang G and Cheng Y:
Detection of epidermal growth factor receptor mutation in
non-small-cell lung carcinoma using cytological and histological
specimens. J BUON. 20:142–145. 2015.PubMed/NCBI
|
|
31
|
Kimura H, Fujiwara Y, Sone T, Kunitoh H,
Tamura T, Kasahara K and Nishio K: EGFR mutation status in
tumour-derived DNA from pleural effusion fluid is a practical basis
for predicting the response to gefitinib. Br J Cancer.
95:1390–1395. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Khode R, Larsen DA, Culbreath BC, Parrish
S, Walker KL, Sayage-Rabie L, Beissner RS and Rao A: Comparative
study of epidermal growth factor receptor mutation analysis on
cytology smears and surgical pathology specimens from primary and
metastatic lung carcinomas. Cancer Cytopathol. 121:361–369. 2013.
View Article : Google Scholar : PubMed/NCBI
|