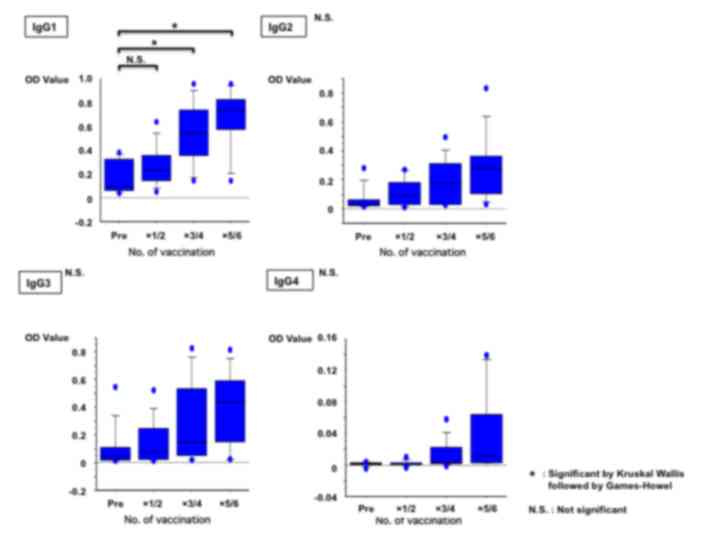
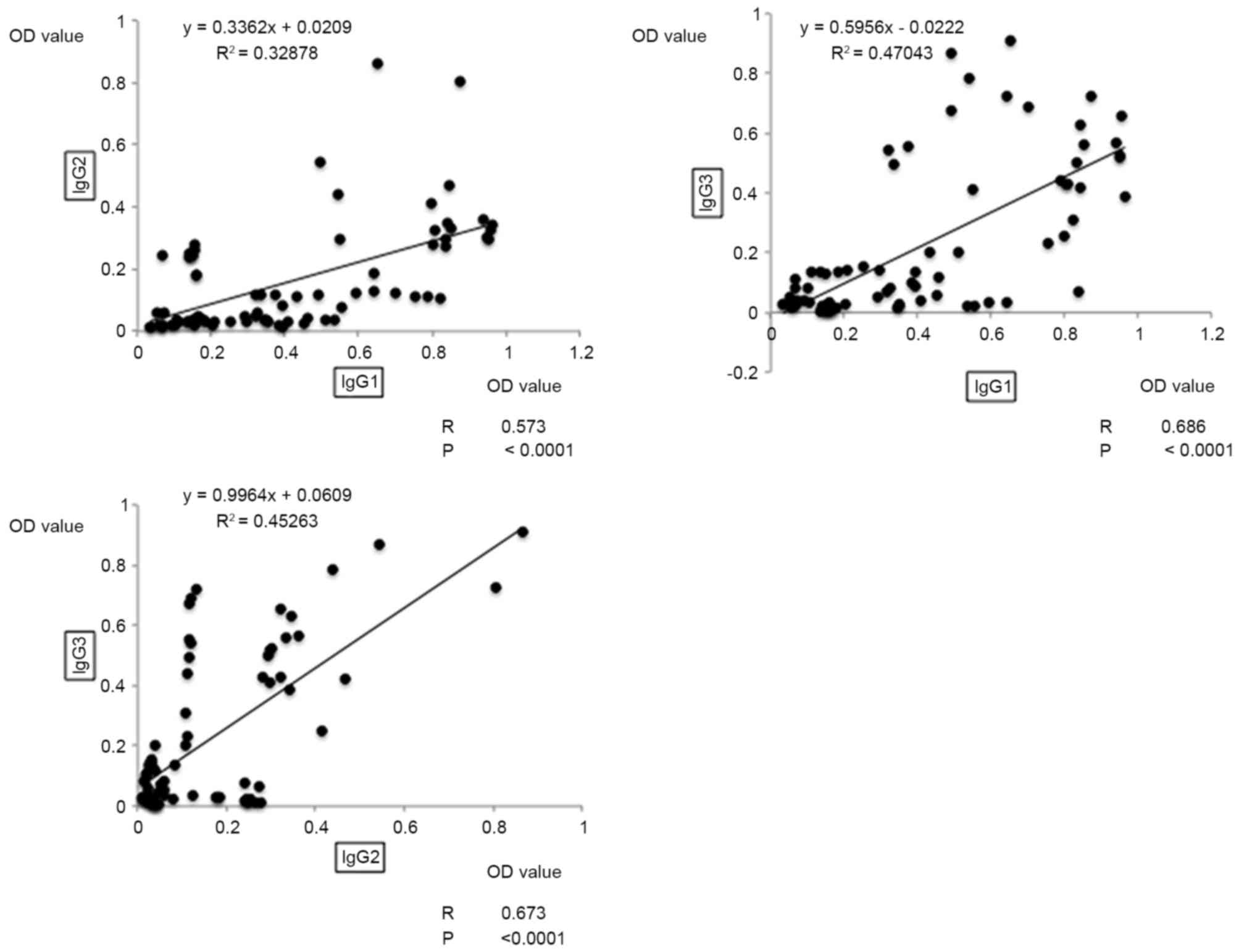
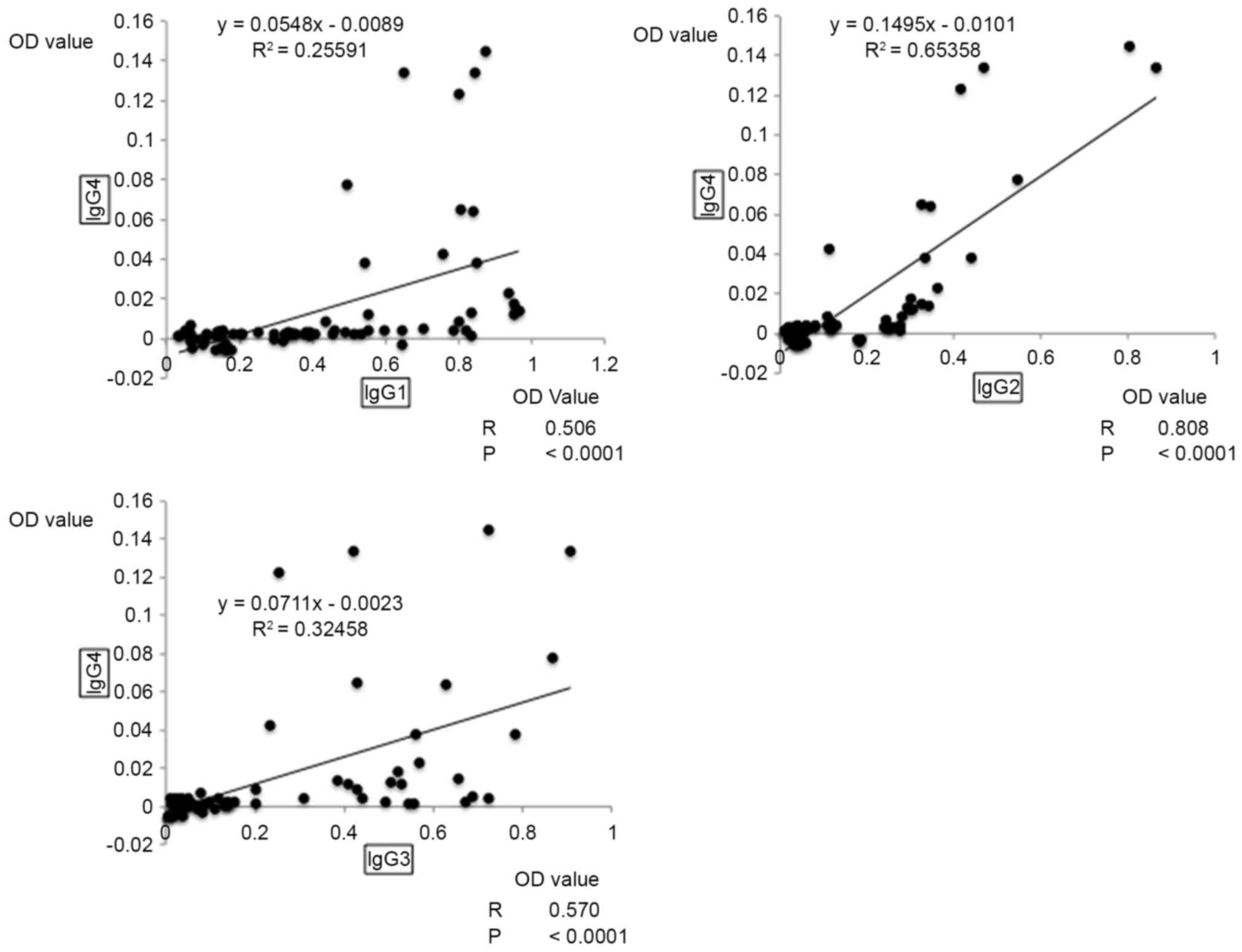
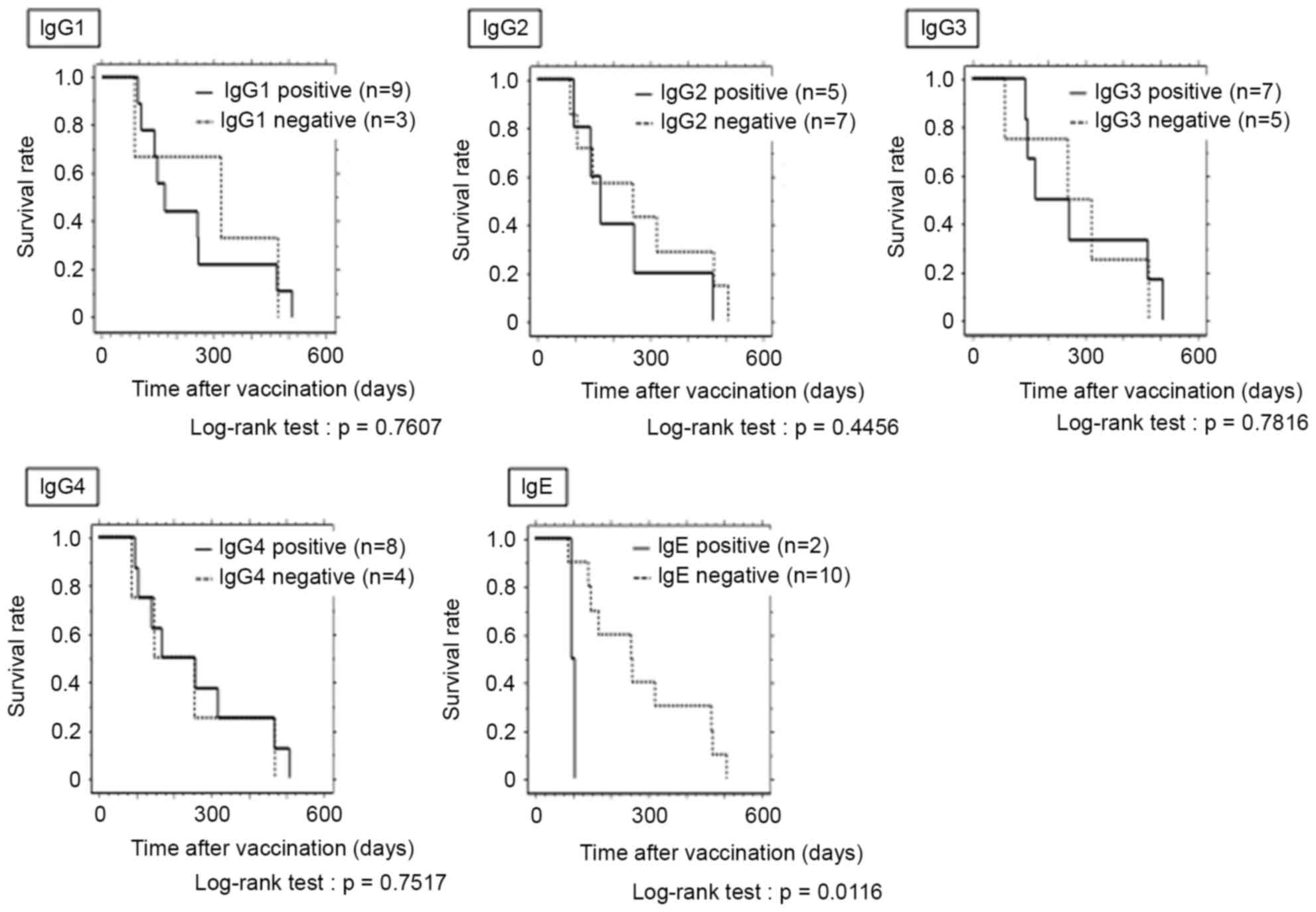
|
1
|
Boon T, Coulie PG, Van den Eynde BJ and
van der Bruggen P: Human T cell responses against melanoma. Annu
Rev Immunol. 24:175–208. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Rosenberg SA: Progress in human tumour
immunology and immunotherapy. Nature. 411:380–384. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Rosenberg SA, Yang JC and Restifo NP:
Cancer immunotherapy: Moving beyond current vaccines. Nat Med.
10:909–915. 2004. View
Article : Google Scholar : PubMed/NCBI
|
|
4
|
Oba K, Teramukai S, Kobayashi M, Matsui T,
Kodera Y and Sakamoto J: Efficacy of adjuvant immunochemotherapy
with polysaccharide K for patients with curative resections of
gastric cancer. Cancer Immunol Immunother. 56:905–911. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Takeoka T, Nagase H, Kurose K, Ohue Y,
Yamasaki M, Takiguchi S, Sato E, Isobe M, Kanazawa T, Matsumoto M,
et al: NY-ESO-1 protein cancer vaccine with poly-ICLC and OK-432:
Rapid and strong induction of NY-ESO-1-specific immune responses by
poly-ICLC. J Immunother. 40:140–147. 2017. View Article : Google Scholar
|
|
6
|
Hoon DS, Yuzuki D, Hayashida M and Morton
DL: Melanoma patients immunized with melanoma cell vaccine induce
antibody responses to recombinant MAGE-1 antigen. J immunol.
154:730–737. 1995.PubMed/NCBI
|
|
7
|
Noguchi M, Mine T, Komatsu N, Suekane S,
Moriya F, Matsuoka K, Yutani S, Shichijo S, Yamada A, Toh U, et al:
Assessment of immunological biomarkers in patients with advanced
cancer treated by personalized peptide vaccination. Cancer Biol
Ther. 10:1266–1279. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Yoshida K, Noguchi M, Mine T, Komatsu N,
Yutani S, Ueno T, Yanagimoto H, Kawano K, Itoh K and Yamada A:
Characteristics of severe adverse events after peptide vaccination
for advanced cancer patients: Analysis of 500 cases. Oncol Rep.
25:57–62. 2011.PubMed/NCBI
|
|
9
|
Romagnani S: TH1 and TH2 in human
diseases. Clin Immunol Immunopathol. 80:225–235. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Shaw DR, Khazaeli MB and LoBuglio AF:
Mouse/human chimeric antibodies to a tumor-associated antigen:
Biologic activity of the four human IgG subclasses. J Natl Cancer
Inst. 80:1553–1559. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Oken MM, Creech RH, Tormey DC, Horton J,
Davis TE, McFadden ET and Carbone PP: Toxicity and response
criteria of the Eastern Cooperative Oncology Group. Am J Clin
Oncol. 5:649–655. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kyogoku N, Ikeda H, Tsuchikawa T, Abiko T,
Fujiwara A, Maki T, Yamamura Y, Ichinokawa M, Tanaka K, Imai N, et
al: Time-dependent transition of the immunoglobulin G subclass and
immunoglobulin E response in cancer patients vaccinated with
cholesteryl pullulan-melanoma antigen gene-A4 nanogel. Oncol Lett.
12:4493–4504. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hoos A, Parmiani G, Hege K, Szno M,
Loibner H, Eggermont A, Urba W, Blumenstein B, Sacks N, Keilholz U,
et al: A clinical development paradigm for cancer vaccine and
related biologics. J Immunother. 30:1–15. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Aoki M, Ueda S, Nishikawa H, Kitano S,
Hirayama M, Ikeda H, Toyoda H, Tanaka K, Kanai M, Takabayashi A, et
al: Antibody responses against NY-ESO-1 and HER2 antigens in
patients vaccinated with combinations of cholesteryl pullulan
(CHP)-NY-ESO-1 and CHP-HER2 with OK-432. Vaccine. 27:6854–6861.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kageyama S, Kitano S, Hirayama M, Nagata
Y, Imai H, Shiraishi T, Akiyoshi K, Scott AM, Murphy R, Hoffman EW,
et al: HUmoral immune responses in patients vaccinated with 1–146
HER2 protein complexed with cholesteryl pullulan nanogel. Cancer
Sci. 99:601–607. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kageyama S, Wada H, Muro K, NIwa Y, Ueda
S, Miyata H, Takiguchi S, Sugino SH, Miyahara Y, Ikeda H, et al:
Dose-dependent effects of NY-ESO-1 protein vaccine complexed with
cholesteryl pullulan (CHP-NY-ESO-1) on immune responses and
survival benefits of esophageal cancer patients. J Transl Med.
11:2462013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Gu XG, Schmitt M, Hiasa A, Nagata Y, Ikeda
H, Sasaki Y, Akiyoshi K, Sunamoto J, Nakamura H, Kuribayashi K and
Shiku H: A novel hydrophobized polysaccharide/oncoprotein complex
vaccine induces in vitro and in vivo cellular and humoral immune
responses against HER2-expressing murine sarcoma. Cancer Res.
58:3385–3390. 1998.PubMed/NCBI
|
|
18
|
Hsu SM, Raine L and Fanger H: The use of
antiavidin antibody and avidin-biotin-peroxidase complex in
immunoperoxidase technics. Am J Clin Pathol. 75:816–821. 1981.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Goydos JS, Elder E, Whiteside TL, Finn OJ
and Lotze MT: A phase I trial of a synthetic mucin peptide vaccine.
Induction of specific immune reactivity in patients with
adenocarcinoma. J syrg Res. 63:298–304. 1996. View Article : Google Scholar
|
|
20
|
Miyagi Y, Imai N, Sasatomi T, Yamada A,
Mine T, Katagiri K, Nakagawa M, Muto A, Okouchi S, Isomoto H, et
al: Induction of cellular immune responses to tumor cells and
peptides in colorectal cancer patients by vaccination with SART3
peptides. Clin Cancer Res. 7:3950–3962. 2001.PubMed/NCBI
|
|
21
|
Tsuruma T, Hata F, Torigoe T, Furuhata T,
Idenoue S, Kurotaki T, Yamamoto M, Yagihashi A, Ohmura T, Yamaguchi
K, et al: Phase I clinical study of anti-apoptosis protein,
survivin-derived peptide vaccine therapy for patients with advanced
or recurrent colorectal cancer. J Transl Med. 2:192004. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wolchok JD, Hoos A, O'Day S, Weber JS,
Hamid O, Lebbé C, Maio M, Binder M, Bohnsack O, Nichol G, et al:
Guidelines for the evaluation of immune therapy activity in solid
tumors: Immune-related response criteria. Clin Cancer Res.
15:7412–7420. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Janetzki S, Britten CM, Kalos M, Levitsky
HI, Maecker HT, Melief CJ, Old LJ, Romero P, Hoos A and Davis MM:
‘MIATA’-minimal information about T cell assays. Immunity.
31:527–528. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kidd P: Th1/Th2 balance: The hypothesis,
its limitations, and implications for health and disease. Altern
Med Rev. 8:223–246. 2003.PubMed/NCBI
|
|
25
|
Turner MC, Chen Y, Krewski D, Ghadirian P,
Thun MJ and Calle EE: Cancer mortality among US men and women with
asthma and hay fever. Am J Epidemiol. 162:212–221. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wang H and Diepgen TL: Is atopy a
protective or a risk factor for cancer? A review of epidemiological
studies. Allergy. 60:1098–1111. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Turner MC, Chen Y, Krewski D and Ghadirian
P: An overview of the association between allergy and cancer. Int J
Cancer. 118:3124–3132. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Nagy E, Berczi I and Sehon AH: Growth
inhibition of murine mammary carcinoma by monoclonal IgE antibodies
specific for the mammary tumor virus. Cancer Immunol Immunother.
34:63–69. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Kershaw MH, Darcy PK, Trapani JA,
MacGregor D and Smyth MJ: Tumor-specific IgE-mediated inhibition of
human colorectal carcinoma xenograft growth. Oncol Res. 10:133–142.
1998.PubMed/NCBI
|
|
30
|
Gould HJ, Mackay GA, Karagiannis SN,
O'Toole CM, Marsh PJ, Daniel BE, Coney LR, Zurawski VR Jr, Joseph
M, Capron M, et al: Comparison of IgE and IgG antibody-dependent
cytotoxicity in vitro and in a SCID mouse xenograft model of
ovarian carcinoma. Eur J Immunol. 29:3527–3537. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Reali E, Greiner JW, Corti A, Gould HJ,
Bottazzoli F, Paganelli G, Schlom J and Siccardi AG: IgEs targeted
on tumor cells: Therapeutic activity and potential in the design of
tumor vaccines. Cancer Res. 61:5517–5522. 2001.PubMed/NCBI
|
|
32
|
Riemer AB, Untersmayr E, Knittelfelder R,
Duschl A, Pehamberger H, Zielinski CC, Scheiner O and
Jensen-Jarolim E: Active induction of tumor-specific IgE antibodies
by oral mimotope vaccination. Cancer Res. 67:3406–3411. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Jensen-Jarolim E, Achatz G, Turner MC,
Karagiannis S, Legrand F, Capron M, Penichet ML, Rodríguez JA,
Siccardi AG, Vangelista L, et al: AllergoOncology: The role of
IgE-mediated allergy in cancer. Allergy. 63:1255–1266. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Singer J and Jensen-Jarolim E: IgE-based
immunotherapy of cancer: Challenges and chances. Allergy.
69:137–149. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Josephs DH, Spicer JF, Karagiannis P,
Gould HJ and Karagiannis SN: IgE immunotherapy: A novel concept
with promise for the treatment of cancer. MAbs. 6:54–72. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Staff C, Magnusson CG, Hojjat-Farsangi M,
Mosolits S, Liljefors M, Frodin JE, Wahrén B, Mellstedt H and
Ullenhag GJ: Induction of IgM, IgA and IgE antibodies in colorectal
cancer patients vaccinated with a recombinant CEA protein. J Clin
Lmmunol. 32:855–865. 2012. View Article : Google Scholar
|
|
37
|
Schur PH: IgG subclasses. A historical
perspective. Monogr Allergy. 23:1–11. 1988.PubMed/NCBI
|
|
38
|
Vidarsson G, Dekkers G and Rispens T: IgG
subclasses and allotypes: From structure to effector function.
Front Immunol. 5:5202014. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Shamji MH, Ljørring C, Francis JN,
Calderon MA, Larché M, Kimber I, Frew AJ, Ipsen H, Lund K, Würtzen
PA and Durham SR: Functional rather than immunoreactive levels of
IgG4 correlate closely with clinical response to grass pollen
immunotherapy. Allergy. 67:217–226. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Crescioli S, Correa I, Karagiannis P,
Davies AM, Sutton BJ, Nestle FO and Karagiannis SN: IgG4
characteristics nad functions in cancer immunity. Curr Allergy
Asthma Rep. 16:72016. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Karagiannis P, Gilbert AE, Josephs DH, Ali
N, Dodev T, Saul L, Correa I, Roberts L, Beddowes E, Koers A, et
al: IgG4 subclass antibodies impair antitumor immunity in melanoma.
J Clin Invest. 123:1457–1474. 2013. View
Article : Google Scholar : PubMed/NCBI
|
|
42
|
Karagiannis P, Villanova F, Josephs DH,
Correa I, Van Hemelrijck M, Hobbs C, Saul L, Egbuniwe IU, Tosi I,
Ilieva KM, et al: Elevated IgG4 in patient circulation is
associated with the risk of disease progression in melanoma.
Oncoimmunology. 4:e10324922015. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Harada K, Shimoda S, Kimura Y, Sato Y,
Ikeda H, Igarashi S, Ren XS, Sato H and Nakanuma Y: Significance of
immunoglobulin G4 (IgG4)-positive cells in extrahepatic
cholangiocarcinoma: Molecular mechanism of IgG4 reaction in cancer
tissue. Hepatology. 56:157–164. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Jäger E, Gnjatic S, Nagata Y, Stockert E,
Jäger D, Karbach J, Neumann A, Rieckenberg J, Chen YT, Ritter G, et
al: Induction of primary NY-ESO-1 immunity: CD8+ T lymphocyte and
antibody responses in peptide-vaccinated patients with NY-ESO-1+
cancers. Proc Natl Acad Sci USA. 97:pp. 12198–12203. 2000;
View Article : Google Scholar : PubMed/NCBI
|