Introduction
Neuron-restrictive silencer factor (NRSF), also
termed repressor element 1-silencing transcription factor (REST),
is a zinc-finger transcription factor and an important regulator of
neural genes (1). Chong et al
(2) first reported that NRSF/REST is
a silencer protein that reduces the expression of sodium channel
genes in neurons. A previous study demonstrated that NRSF/REST is
an important regulator of neurogenesis in vitro and in
vivo. For example, downregulation of NRSF/REST in embryonic
stem cells induces neuronal lineage differentiation (3), and knockdown of NRSF/REST in cultured
neural stem cells induces the expression of pro-neuronal genes,
including neuronal differentiation 1, neuron-specific class III
β-tubulin and doublecortin (4). In
Xenopus and chicken embryos, NRSF/REST inactivation induces
abnormal neurogenesis and inhibits the repression of neuronal
tubulin and several other neuronal target genes (5,6).
Additionally, NRSF/REST overexpression represses the expression of
neuronal genes, including N-tubulin and neuronal cell adhesion
molecule (7).
Numerous studies have investigated the expression
and, to a lesser extent, the function of NRSF/REST in tumors of the
nervous system (8–15). Certain tumors, including
neuroblastoma, share a number of biological properties with
neuronal progenitor cells and, thus, can acquire neuronal
phenotypes in response to a variety of agents. A study by Nishimura
et al (8) examined the levels
of NRSF/REST mRNA in a human neuroblastoma cell line following
induced differentiation. The study demonstrated that the NRSF/REST
mRNA level was evidently decreased following induction, indicating
that NRSF/REST expression is a biochemical marker of neuronal
differentiation in neuroblastoma cells (8). A similar downregulation of NRSF/REST was
also demonstrated in other studies using different neuroblastoma
cell lines (9,10). In addition, high NRSF/REST levels have
been detected in human medulloblastoma cell lines and tumors
(11–13). Similarly, in human glioblastoma
multiforme (GBM), NRSF/REST is highly expressed (14) and its inhibition suppresses the
proliferation and migration of GBM cells (15).
Certain studies have also investigated the
expression and/or function of NRSF/REST in non-neuroepithelial
tumors in vitro and in vivo. Gurrola-Diaz et
al (16) reported that exogenous
overexpression of NRSF/REST in NRSF/REST-deficient small-cell lung
cancer (SCLC) cell lines induced apoptosis of SCLC cells,
indicating that the inhibition of NRSF/REST activity is a crucial
step in the carcinogenesis of a subgroup of SCLCs (16). Similar results were also observed in
human non-SCLC cell lines (17).
Kreisler et al (18) reported
that loss of NRSF/REST expression was associated with the malignant
progression of SCLC. In human breast cancer cells, NRSF/REST
activity was required for estradiol stimulation of the cell cycle
(19). Immunohistochemistry
previously demonstrated that NRSF/REST expression is significantly
lower in breast cancer samples compared with normal and benign
breast samples, and that knockdown of NRSF/REST expression by short
hairpin RNA in MCF-7 human breast cancer cells resulted in an
increase in cell proliferation, suppression of apoptosis and
reduced sensitivity to anticancer drugs (20). Previous studies have also demonstrated
the important function of NRSF/REST in the pathogenesis of uterine
fibroids (21). However, the
expression of NRSF/REST in liver tumors remains unclear. Thus, the
present study determined the expression profile of NRSF/REST in
liver tumors using tissue microarray (TMA)
immunohistochemistry.
Materials and methods
TMA and pathology
All paraffin-embedded TMAs used in the present study
were purchased from US Biomax, Inc. (Thermo Fisher Scientific,
Inc., Waltham, MA, USA). The hepatic carcinoma and normal hepatic
tissue TMAs (cat. no. BC03118) contained 90 carcinoma samples,
including 15 cholangiocellular carcinoma (CCC), 75 hepatocellular
carcinoma (HCC), and 10 normal hepatic tissue samples. On the basis
of morphology, the liver carcinoma samples were graded 1–3 (or
I–III), according to the Tumor-Node-Metastasis grading system
(22) by the supplier, indicating
well-, moderately- or poorly-differentiated tissue, respectively.
In total, there were 200 tissue samples on the microarray, with two
samples from each patient.
Immunohistochemistry
TMAs were deparaffinized with xylene, rehydrated
with a graded alcohol series and subjected to heat-mediated antigen
retrieval [0.01 M sodium citrate buffer, (pH 6.0)], according to a
previously described protocol (23,24). TMAs
were then rinsed with phosphate-buffered saline [PBS; 0.01 mol/l,
(pH 7.4)] and blocked with 3% H2O2 (v/v in
PBS) for 15 min at room temperature. The sections were then
incubated for 20 min at room temperature with 2% normal goat serum
(v/v; Beijing Zhongshan Golden Bridge Biotechnology Co., Ltd.,
Beijing, China) at room temperature to block the non-specific
binding. Subsequently, the TMAs were incubated overnight at 4°C
with a polyclonal rabbit antibody against NRSF/REST (cat. no.
ab21635; Abcam, Cambridge, UK) that was diluted 1:100 with antibody
diluent (cat. no. S3022; Dako; Agilent Technologies, Inc., Santa
Clara, CA, USA). Following several washes with PBS, the sections
were incubated with biotinylated goat anti-rabbit secondary
antibody (cat. no. ZB2010; 1:200; Beijing Zhongshan, China) for 1 h
at room temperature. The sections were then washed with PBS and
incubated with horseradish peroxidase-labeled streptavidin (cat.
no. ZB2404; Beijing Zhongshan Golden Bridge Biotechnology Co.,
Ltd.) for 1 h at room temperature. Finally, the sections were
incubated with a diaminobenzidine-peroxidase substrate kit (cat.
no. ZLI-9018; Beijing Zhongshan Golden Bridge Biotechnology Co.,
Ltd.) for 5 min at room temperature. The equivalent procedure was
conducted for the blank controls, with the primary antibody
replaced by antibody diluent.
Imaging and data analysis
Images of the immunohistochemical staining were
captured with a DP70 digital camera (Leica Microsystems GmbH,
Wetzlar, Germany) mounted to a BX60 Olympus microscope (Olympus
Corporation, Tokyo, Japan). The staining was scored according to
the previously described four-point system (score 0–3) (24) by a pathologist (double-blinded) as
follows: Score 3, dark staining that is easily visible and present
in >50% of cells; score 2, focal areas of dark staining (<50%
of cells) or moderate staining of >50% of cells; score 1, focal
moderate staining in <50% of cells or pale staining in any
proportion of cells not easily observable at low power; and score
0, none of the above. A high level of expression was defined as a
score of 2–3 and low level of expression was defined as a score of
0–1, as described previously (24).
Considering the comparatively small sample size, an early tumor
stage was defined as stages I and II, and the advanced stage was
defined as stages III and IIIb. Well-differentiated carcinoma (WDC)
was defined as grade 1, moderately-differentiated carcinoma (MDC)
as grade 2 and poorly-differentiated carcinoma (PDC) was defined as
grade 3 (24).
Statistical analysis
All data are expressed as n (%) and were compared
using a χ2 test. A Fisher's exact test was used for
correction when necessary. Statistical analysis was performed using
SPSS software (version 18.0; SPSS, Inc., Chicago, IL, USA). All
P-values were 2-tailed and P<0.05 was considered to indicate a
statistically significant difference.
Results
Subcellular localization of NRSF/REST
immunohistochemical staining
The immunohistochemical analysis demonstrated that,
in normal hepatic tissue, NRSF/REST was present in the nuclei and
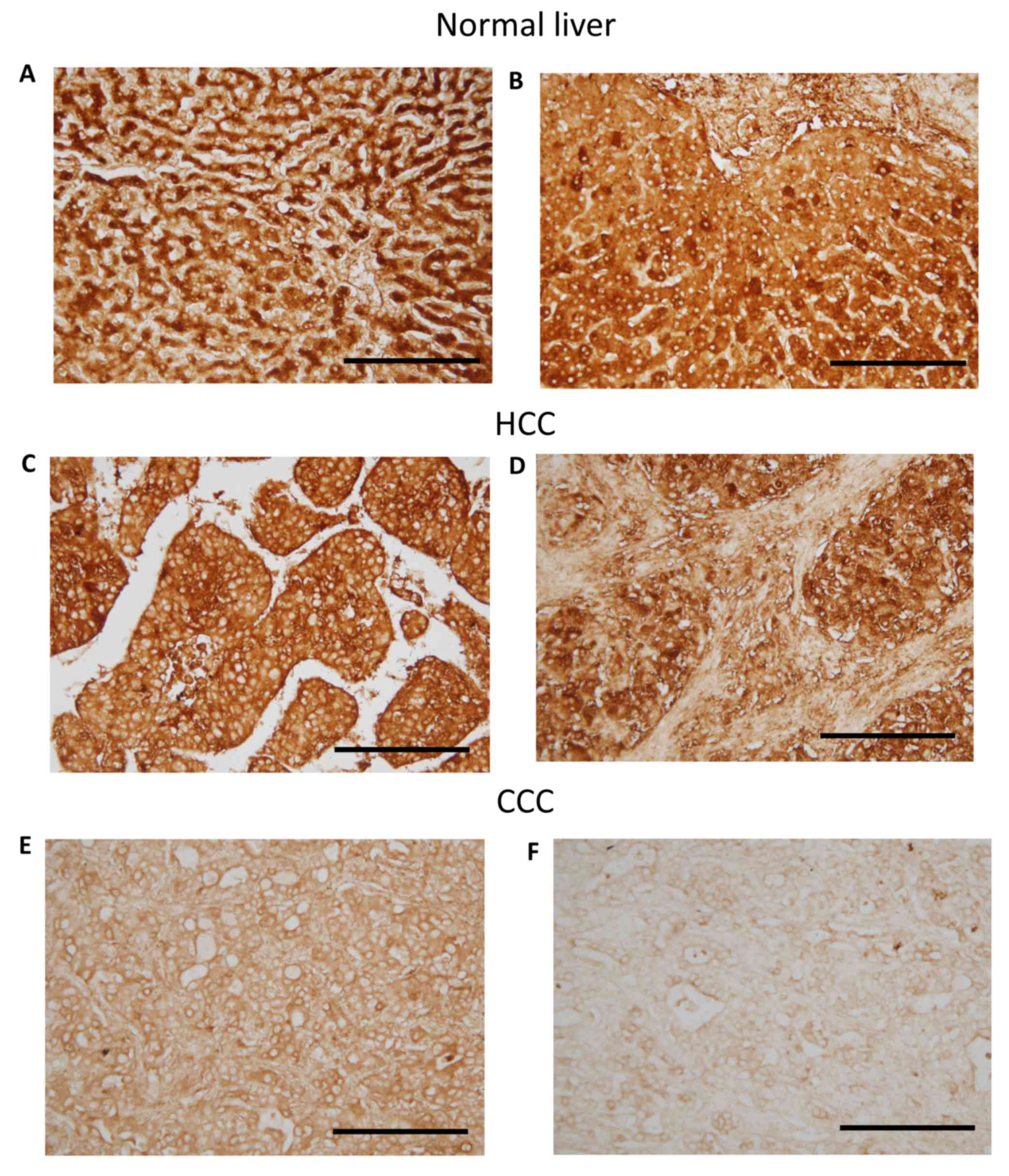
cytoplasm of hepatocytes and cholangiocytes (Fig. 1A and B). In CCC and HCC tissues,
NRSF/REST was predominantly detected in the cytoplasm, with the
nuclei clearly unstained. Additionally, the NRSF/REST
immunohistochemical staining of HCC tissues seemed stronger than
that of CCC tissues (Fig. 1C-F).
Expression of NRSF/REST in liver
carcinoma
The levels of NRSF/REST immunoreactivity were
compared between normal and liver carcinomas. As presented in
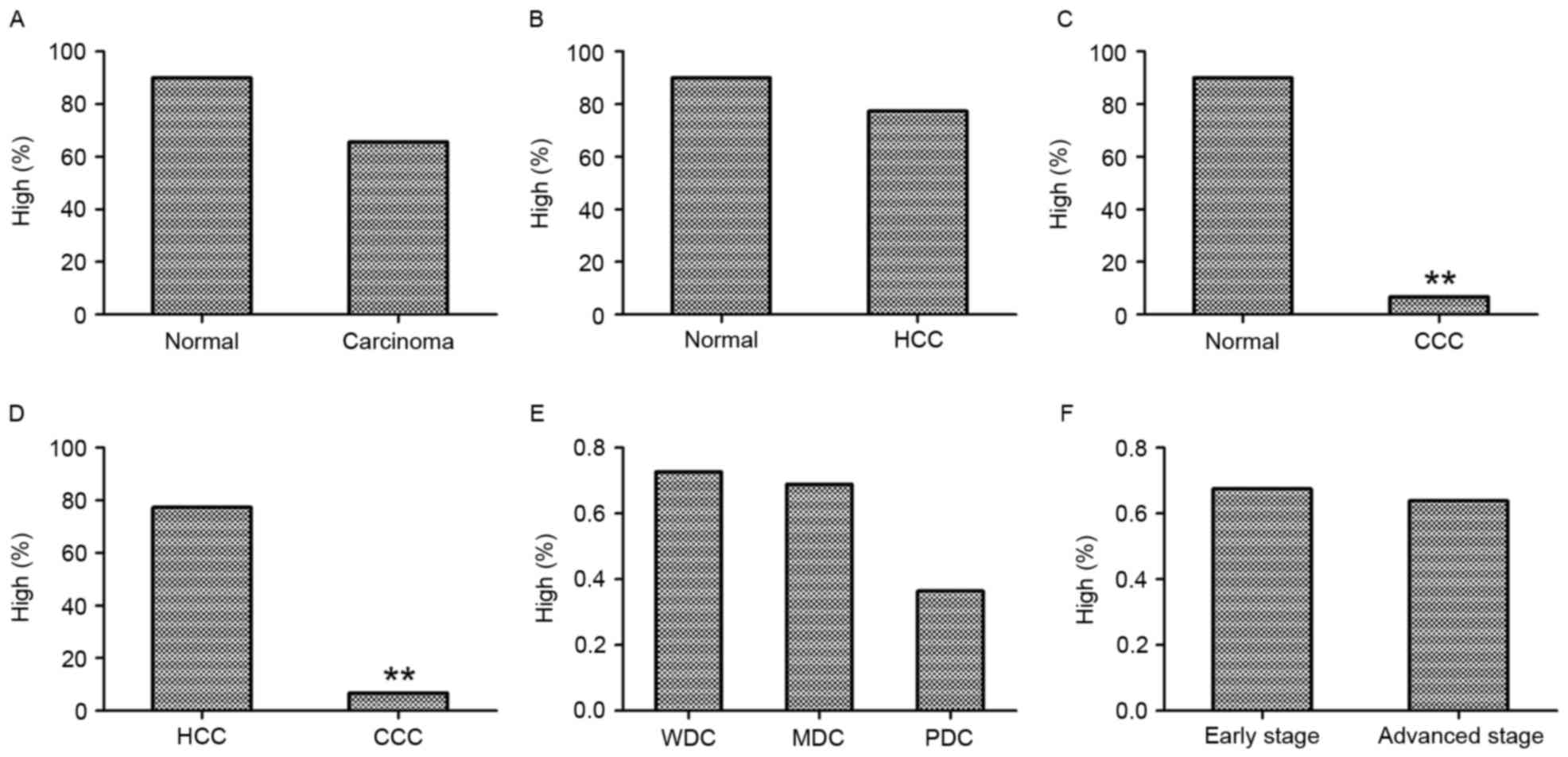
Table I, among the 10 cases of normal
liver tissue on the TMA, 90% exhibited high levels of NRSF/REST
(score 2–3; Fig. 1A and B). Among the
90 cases, 66% (59/90) of liver carcinoma samples and 77% (58/75) of
HCC samples demonstrated high levels of NRSF/REST (Table I; Fig.
2). There was no significant difference in the percentage of
samples with high NRSF/REST levels between the normal liver tissue
and liver carcinoma samples (P>0.05; Fig. 2A; Table
I) or between normal liver tissue and HCC (P>0.05; Fig. 2B; Table
I). However, the percentage of samples with high NRSF/REST
expression was significantly reduced in CCC samples (7%; 1/15)
compared with normal liver tissues (P<0.001; Fig. 2C; Table
I) and with HCC samples (P<0.001; Fig. 2D; Table
I).
 | Table I.Expression of NRSF/REST in normal and
abnormal liver tissues. |
Table I.
Expression of NRSF/REST in normal and
abnormal liver tissues.
| Tissue | High | Low | χ2 | P-value |
|---|
| Carcinoma vs.
normal |
|
| 1.476 | 0.224 |
|
Carcinoma | 59 | 31 |
|
|
|
Normal | 9 | 1 |
|
|
| CCC vs. normal |
|
| 14.063 | <0.001 |
|
CCC | 1 | 14 |
|
|
|
Normal | 9 | 1 |
|
|
| HCC vs. normal |
|
| 0.259 | 0.611 |
|
HCC | 58 | 17 |
|
|
|
Normal | 9 | 1 |
|
|
| CCC vs. HCC |
|
| 27.645 | <0.001 |
|
CCC | 1 | 14 |
|
|
|
HCC | 58 | 17 |
|
|
| Carcinoma
early/advanced stage |
|
| 0.130 | 0.719 |
|
Early | 29 | 14 |
|
|
|
Advanced | 30 | 17 |
|
|
| Carcinoma
differentiation stage |
|
| 4.656 | 0.097 |
|
WDC | 8 | 3 |
|
|
|
MDC | 44 | 20 |
|
|
|
PDC | 4 | 7 |
|
|
Among the 90 cases of liver carcinomas, 11 cases
were graded as WDC, 64 as MDC and 11 as PDC (including grade 2–3).
The grading of the remaining 4 cases was undetermined. The
percentage of high NRSF/REST immunoreactivity was 73% (8/11) in
WDCs, 69% (44/64) in MDCs and 36% (4/11) in PDCs. There was no
statistical difference between the percentages of samples with high
NRSF/REST staining among the three groups (P>0.05; Fig. 2E; Table
I).
Additionally, the association between NRSF/REST
expression and tumor stage was analyzed. Stages I and II were
defined as early stage, and stages III and IIIb as advanced stage.
Thus, 43 cases were early stage and 47 cases were advanced stage.
High levels of NRSF/REST were observed in 67% (29/43) of early
stage and 64% (30/47) of advanced stage samples, with no
statistical difference between the two stages (P>0.05; Fig. 2F; Table
I).
Sex-associated differences in
NRSF/REST in normal and cancerous liver tissue
The occurrence of liver diseases exhibits a certain
degree of sex bias, with a higher percentage detected in males. A
total of 74.11% of new liver cancer cases in 2014 in the USA were
in males (25). Thus, the present
study compared the expression of NRSF/REST in normal and cancerous
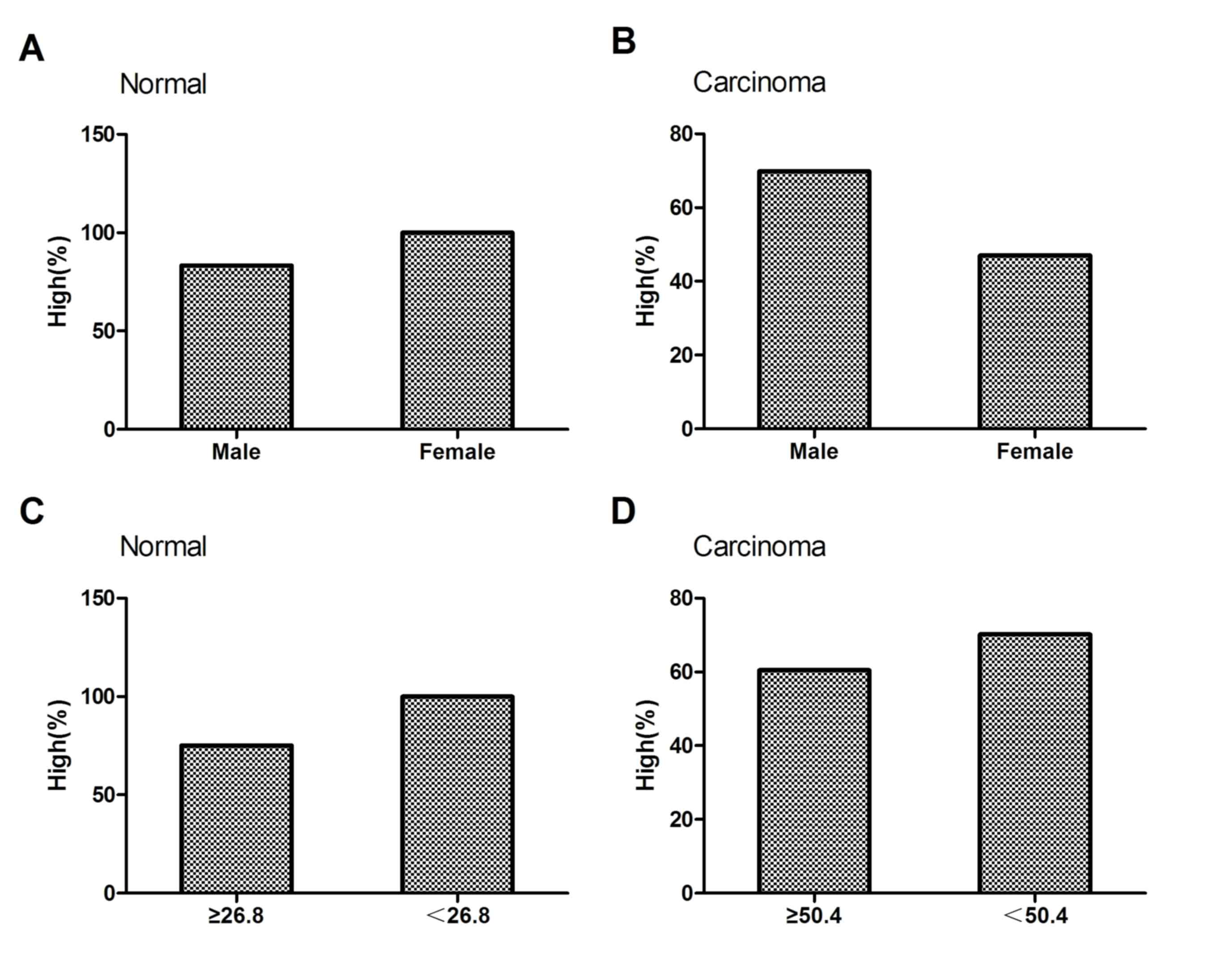
liver tissues from men and women. Among the 10 cases of normal
liver tissue, the percentage of samples with high NRSF/REST levels
was 83% (5/6) in males and 100% (4/4) in females, with no
significant difference between the sexes (P>0.05; Fig. 3A; Table
II). Among the 90 cases of liver carcinoma, the percentage of
samples with high levels of NRSF/REST was 70% (51/73) in males and
47% (8/17) in females, demonstrating no significant difference
between the sexes (P>0.05; Fig.
3B; Table II).
 | Table II.Sex and age-associated differences of
NRSF/REST in normal and cancerous liver tissues. |
Table II.
Sex and age-associated differences of
NRSF/REST in normal and cancerous liver tissues.
| A, Sex-associated
differences |
|---|
|
|---|
| Tissue | High | Low | χ2 | P-value |
|---|
| Normal |
|
| 0.000 | 1.000 |
|
Male | 5 | 1 |
|
|
|
Female | 4 | 0 |
|
|
| Carcinoma |
|
| 3.176 | 0.075 |
|
Male | 51 | 22 |
|
|
|
Female | 8 | 9 |
|
|
| CCC |
|
| 0.000 | 1.000 |
|
Male | 1 | 7 |
|
|
|
Female | 0 | 7 |
|
|
| HCC |
|
| 0.000 | 1.000 |
|
Male | 50 | 15 |
|
|
|
Female | 8 | 2 |
|
|
|
| B,
Age-associated differences |
|
| Tissue | High | Low |
χ2 | P-value |
|
| Normal |
|
| 0.046 | 0.830 |
|
≥26.8 | 3 | 1 |
|
|
|
<26.8 | 6 | 0 |
|
|
| Carcinoma |
|
| 0.945 | 0.331 |
|
≥50.4 | 26 | 17 |
|
|
|
<50.4 | 33 | 14 |
|
|
| CCC |
|
| 0.005 | 0.945 |
|
≥48.5 | 0 | 8 |
|
|
|
<48.5 | 1 | 6 |
|
|
| HCC |
|
| 0.792 | 0.373 |
|
≥50.8 | 27 | 10 |
|
|
|
<50.8 | 31 | 7 |
|
|
Among the 15 cases of CCC, only 1 male demonstrated
high levels of NRSF/REST and the other 14 cases (7 male and 7
female) all demonstrated low levels of NRSF; thus, no significant
sex-associated difference was identified (P>0.05; Table II). Among the 75 cases of HCC, the
percentage of samples with high levels of NRSF/REST was 77% (50/65)
in males and 80% (8/10) in females; thus, there was no significant
sex-associated difference detected (P>0.05; Table II).
Age-associated differences in
NRSF/REST in normal and cancerous liver tissue
The present study also investigated the effect of
age on NRSF/REST immunoreactivity in normal and cancerous liver
tissues. Among the 10 normal liver tissues, the mean age of the
patients was 26.8 years. The percentage of samples with high
expression of NRSF/REST was 75% (3/4) in samples from patients aged
≥26.8 years, and 100% (6/6) in those aged <26.8 years. The
statistical analysis demonstrated no significant difference between
the two age groups (P>0.05; Fig.
3C; Table II). Among the 90
cases of liver carcinoma, the mean age was 50.4 years. The
percentage of samples with high NRSF/REST expression was 60%
(26/43) in patients aged ≥50.4 years and 70% (33/47) in those aged
<50.4 years. Statistical analysis demonstrated no significant
difference between the two age groups (P>0.05; Fig. 3D; Table
II).
The age-associated difference of NRSF/REST
immunoreactivity in individual liver carcinoma tissues was also
analyzed. Among the 15 cases of CCC, the mean age of the patients
was 48.5 years. Only 1 patient aged <48.5 years demonstrated
high levels of NRSF/REST and statistical analysis revealed no
significant difference between the two age groups (P>0.05;
Table II). Among the 75 cases of
HCC, the mean age of the patients was 50.8 years. The percentage
with high expression of NRSF/REST was 73% (27/37) in patients ≥50.8
years old and 82% (31/38) in those <50.8 years old. Statistical
analysis demonstrated no significant difference between the two age
groups (P>0.05; Table II).
Discussion
The present study examined the expression of
NRSF/REST in normal liver tissue and liver carcinomas using TMA
immunohistochemistry. The results demonstrated that, in the normal
liver tissue, NRSF/REST expression was present in the nuclei and
cytoplasm, whereas, in liver tumor tissue, nuclear NRSF/REST
staining was clearly reduced and expression was detected in the
cytoplasm. Furthermore, the highest levels of NRSF/REST were
detected in normal liver tissue (90% of cases); however, the
expression of NRSF/REST among all liver tumors, including HCC and
CCC, did not demonstrate any significant difference compared with
the normal liver tissue. However, in CCC tissues, the number of
samples with high NRSF/REST expression was significantly decreased
compared with normal or HCC tissues, with only 7% of CCCs
exhibiting high levels of NRSF/REST. The expression of NRSF/REST
was not statistically different among the grades of tumor
differentiation (WDC, MDC and PDC) or between pathological stages.
Finally, no age- or sex-associated differences were identified in
the number of samples with high NRSF/REST immunoreactivity among
all the tissues examined, including normal and cancerous liver
tissues.
As a transcription factor, NRSF/REST expression has
typically been detected in cell nuclei in previous studies. For
example, in human medulloblastoma tumors (13) and normal human brain tissue (26), NRSF/REST staining was observed to be
localized to the nuclei. Conti et al (14) also reported that in gliomas, NRSF/REST
exhibited a nuclear staining pattern (14). In the present study, positive
NRSF/REST staining was detected in the nuclei and cytoplasm in
normal liver tissue; whereas, in liver carcinoma, NRSF/REST was
predominantly detected in the cytoplasm. Similar results have been
previously observed in other tissues. For example, Conti et
al (14) also reported that in
normal human tissue, NRSF/REST immunoreactivity was detected in the
cytoplasm of selected neurons. Furthermore, Orta-Salazar et
al (27) reported that in the
hippocampus of a 3×Tg-AD mouse (a mouse model of Alzheimer's
disease), the nuclei and cytoplasm were NRSF/REST-positive.
Additionally, these mice exhibited decreased cytoplasmic and
increased nuclear NRSF/REST staining compared with control mice,
which was indicated to be associated with the degeneration observed
in Alzheimer's disease. Lu et al (26) reported that in normal human brain
tissue, NRSF/REST was predominantly localized in the cell nuclei;
however, in several brain tissue samples from patients with
dementia, NRSF/REST was predominantly absent from the cell nuclei,
but present in the cytoplasm. This nuclear loss and cytoplasmic
translocation was indicated to be one of the causes of the reduced
repression of certain dementia/stress-associated genes that are
highly expressed in dementia (26).
In the present study, nuclear/cytoplasmic translocation of
NRSF/REST was clearly observed; this shift indicated that loss of
nuclear NRSF/REST may contribute to hepatic carcinogenesis.
It has been previously reported that NRSF/REST may
be tumor-suppressive or exert an oncogenic effect (28). Using array-comparative genomic
hybridization analysis, Westbrook et al (29) identified that NRSF/REST is frequently
deleted in colorectal cancer, and proposed that it functions as a
tumor suppressor. Additionally, Blom et al (30) used gene copy number analyses to
demonstrate that the majority of brain tumors exhibited low-level
amplification of NRSF/REST. Wagoner et al (31) reported that expression of NRSF/REST
was lost in aggressive breast cancer, and that this loss was
positively correlated with poor prognosis and higher recurrence
rates. Similarly, Lv et al (20) reported that NRSF/REST expression was
decreased in breast cancer samples. Furthermore, Varghese et
al (21) demonstrated that
expression of NRSF/REST was reduced in uterine fibroids
(leiomyomas). A decreased NRSF/REST expression profile was also
detected in human brain tissue from patients with dementia and in
the brain tissue from an Alzheimer's disease mouse model (26,27).
However, certain studies have also reported that
NRSF/REST expression is increased in individual tumors or other
tissues. For example, Lawinger et al (11) reported that NRSF/REST levels were high
in three types of human medulloblastoma cells. Furthermore, Fuller
et al (13) reported that
expression of NRSF/REST was increased in human medulloblastoma
tumors compared with normal brain tissue samples (13) and Conti et al (14) observed that NRSF/REST expression was
increased in human GBM. The present study demonstrated that the
number of samples with high NRSF/REST expression was reduced in CCC
compared with normal tissues, indicating that decreased NRSF/REST
levels may be associated with the occurrence and/or poor prognosis
of CCC.
Liver disease is a major global health concern
(32), with liver and intrahepatic
bile duct cancer among the top 10 causes of cancer-associated
mortality in the United States (25).
In China, liver carcinomas (including HCC and CCC) are also
commonly diagnosed and have been identified as one of the leading
causes of cancer-associated mortality (33). Therefore, it is important to
investigate the mechanisms of liver disease and to identify
potential novel therapeutic targets. A study by Sedaghat et
al (34) reported that in mouse
liver, NRSF/REST regulates the expression of neuronal markers,
including brain-derived neurotrophic factor and various other genes
(a total of 433 genes, of which 25% were downregulated and 75%
upregulated), particularly those associated with the cell cycle,
cell growth, proliferation and cancer.
The present study observed cytoplasmic translocation
of NRSF/REST in liver carcinomas compared with NRSF/REST detected
in normal liver tissues and the levels of NRSF/REST expression were
reduced in CCC. In conclusion, the results of the current and
previous studies indicate that NRSF/REST has an important function
in liver carcinomas. Furthermore, the observed nuclear/cytoplasmic
translocation may contribute to tumor formation and the reduced
levels of NRSF/REST may potentially be used as a biomarker of CCC.
Notably, as NRSF/REST may paradoxically exert tumor suppressive or
oncogenic effects (28), the
expression and importance of NRSF/REST in normal and abnormal liver
tissues requires further investigation.
Acknowledgements
The present study was supported by the National
Science Foundation of China (grant nos. 81571059 and 81270525).
References
|
1
|
Zhao Y, Zhu M, Yu Y, Qiu L, Zhang Y, He L
and Zhang J: Brain REST/NRSF is not only a silent repressor but
also an active protector. Mol Neurobiol. 54:541–550. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Chong JA, Tapia-Ramírez J, Kim S,
Toledo-Aral JJ, Zheng Y, Boutros MC, Altshuller YM, Frohman MA,
Kraner SD and Mandel G: REST: A mammalian silencer protein that
restricts sodium channel gene expression to neurons. Cell.
80:949–957. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Gupta SK, Gressens P and Mani S: NRSF
downregulation induces neuronal differentiation in mouse embryonic
stem cells. Differentiation. 77:19–28. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Gao Z, Ure K, Ding P, Nashaat M, Yuan L,
Ma J, Hammer RE and Hsieh J: The master negative regulator
REST/NRSF controls adult neurogenesis by restraining the neurogenic
program in quiescent stem cells. J Neurosci. 31:9772–9786. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Chen ZF, Paquette AJ and Anderson DJ:
NRSF/REST is required in vivo for repression of multiple neuronal
target genes during embryogenesis. Nat Genet. 20:136–142. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Olguín P, Oteíza P, Gamboa E,
Gómez-Skármeta JL and Kukuljan M: RE-1 silencer of
transcription/neural restrictive silencer factor modulates
ectodermal patterning during Xenopus development. J
Neurosci. 26:2820–2829. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Paquette AJ, Perez SE and Anderson DJ:
Constitutive expression of the neuron-restrictive silencer factor
(NRSF)/REST in differentiating neurons disrupts neuronal gene
expression and causes axon pathfinding errors in vivo. Proc Natl
Acad Sci USA. 97:12318–12323. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Nishimura E, Sasaki K, Maruyama K, Tsukada
T and Yamaguchi K: Decrease in neuron-restrictive silencer factor
(NRSF) mRNA levels during differentiation of cultured neuroblastoma
cells. Neurosci Lett. 211:101–104. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lee JH, Chai YG and Hersh LB: Expression
patterns of mouse repressor element-1 silencing transcription
factor 4 (REST4) and its possible function in neuroblastoma. J Mol
Neurosci. 15:205–214. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lepagnol-Bestel AM, Maussion G, Ramoz N,
Moalic JM, Gorwood P and Simonneau M: Nrsf silencing induces
molecular and subcellular changes linked to neuronal plasticity.
Neuroreport. 18:441–446. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lawinger P, Venugopal R, Guo ZS, Immaneni
A, Sengupta D, Lu W, Rastelli L, Carneiro Marin Dias A, Levin V,
Fuller GN, et al: The neuronal repressor REST/NRSF is an essential
regulator in medulloblastoma cells. Nat Med. 6:826–831. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Su X, Gopalakrishnan V, Stearns D, Aldape
K, Lang FF, Fuller G, Snyder E, Eberhart CG and Majumder S:
Abnormal expression of REST/NRSF and Myc in neural stem/progenitor
cells causes cerebellar tumors by blocking neuronal
differentiation. Mol Cell Biol. 26:1666–1678. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Fuller GN, Su X, Price RE, Cohen ZR, Lang
FF, Sawaya R and Majumder S: Many human medulloblastoma tumors
overexpress repressor element-1 silencing transcription
(REST)/neuron-restrictive silencer factor, which can be
functionally countered by REST-VP16. Mol Cancer Ther. 4:343–349.
2005.PubMed/NCBI
|
|
14
|
Conti L, Crisafulli L, Caldera V,
Tortoreto M, Brilli E, Conforti P, Zunino F, Magrassi L, Schiffer D
and Cattaneo E: REST controls self-renewal and tumorigenic
competence of human glioblastoma cells. PLoS One. 7:e384862012.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zhang D, Li Y, Wang R, Li Y, Shi P, Kan Z
and Pang X: Inhibition of rest suppresses proliferation and
migration in glioblastoma cells. Int J Mol Sci. 17:pii: E664.
2016.
|
|
16
|
Gurrola-Diaz C, Lacroix J, Dihlmann S,
Becker CM and von Knebel Doeberitz M: Reduced expression of the
neuron restrictive silencer factor permits transcription of glycine
receptor alpha1 subunit in small-cell lung cancer cells. Oncogene.
22:5636–5645. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Watanabe H, Mizutani T, Haraguchi T,
Yamamichi N, Minoguchi S, Yamamichi-Nishina M, Mori N, Kameda T,
Sugiyama T and Iba H: SWI/SNF complex is essential for
NRSF-mediated suppression of neuronal genes in human nonsmall cell
lung carcinoma cell lines. Oncogene. 25:470–479. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Kreisler A, Strissel PL, Strick R, Neumann
SB, Schumacher U and Becker CM: Regulation of the NRSF/REST gene by
methylation and CREB affects the cellular phenotype of small-cell
lung cancer. Oncogene. 29:5828–5838. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Bronson MW, Hillenmeyer S, Park RW and
Brodsky AS: Estrogen coordinates translation and transcription,
revealing a role for NRSF in human breast cancer cells. Mol
Endocrinol. 24:1120–1135. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Lv H, Pan G, Zheng G, Wu X, Ren H, Liu Y
and Wen J: Expression and functions of the repressor element 1
(RE-1)-silencing transcription factor (REST) in breast cancer. J
Cell Biochem. 110:968–974. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Varghese BV, Koohestani F, McWilliams M,
Colvin A, Gunewardena S, Kinsey WH, Nowak RA, Nothnick WB and
Chennathukuzhi VM: Loss of the repressor REST in uterine fibroids
promotes aberrant G protein-coupled receptor 10 expression and
activates mammalian target of rapamycin pathway. Proc Natl Acad Sci
USA. 110:2187–2192. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Edge SB and Compton CC: The American Joint
Committee on Cancer: The VIIth edition of the AJCC cancer staging
manual and the future of TNM. Ann Surg Oncol. 17:1471–1474. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Liu M, Zhang K, Zhao Y, Guo Q, Guo D and
Zhang J: Evidence for involvement of steroid receptors and
coactivators in neuroepithelial and meningothelial tumors. Tumour
Biol. 36:3251–3261. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Liu C, Zhang Y, Zhang K, Bian C, Zhao Y
and Zhang J: Expression of estrogen receptors, androgen receptor
and steroid receptor coactivator-3 is negatively correlated to the
differentiation of astrocytic tumors. Cancer Epidemiol. 38:291–297.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Siegel R, Ma J, Zou Z and Jemal A: Cancer
statistics, 2014. CA Cancer J Clin. 64:9–29. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lu T, Aron L, Zullo J, Pan Y, Kim H, Chen
Y, Yang TH, Kim HM, Drake D, Liu XS, et al: REST and stress
resistance in ageing and Alzheimer's disease. Nature. 507:448–454.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Orta-Salazar E, Aguilar-Vázquez A,
Martínez-Coria H, Luquín-De Anda S, Rivera-Cervantes M, Beas-Zarate
C, Feria-Velasco A and Díaz-Cintra S: REST/NRSF-induced changes of
ChAT protein expression in the neocortex and hippocampus of the
3xTg-AD mouse model for Alzheimer's disease. Life Sci. 116:83–89.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Majumder S: REST in good times and bad:
Roles in tumor suppressor and oncogenic activities. Cell Cycle.
5:1929–1935. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Westbrook TF, Martin ES, Schlabach MR,
Leng Y, Liang AC, Feng B, Zhao JJ, Roberts TM, Mandel G, Hannon GJ,
et al: A genetic screen for candidate tumor suppressors identifies
REST. Cell. 121:837–848. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Blom T, Tynninen O, Puputti M, Halonen M,
Paetau A, Haapasalo H, Tanner M and Nupponen NN: Molecular genetic
analysis of the REST/NRSF gene in nervous system tumors. Acta
Neuropathol. 112:483–490. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Wagoner MP, Gunsalus KT, Schoenike B,
Richardson AL, Friedl A and Roopra A: The transcription factor REST
is lost in aggressive breast cancer. PLoS Genet. 6:e10009792010.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Hansel MC, Davila JC, Vosough M,
Gramignoli R, Skvorak KJ, Dorko K, Marongiu F, Blake W and Strom
SC: The use of induced pluripotent stem cells for the study and
treatment of liver diseases. Curr Protoc Toxicol.
67:14.13.1–14.13.27. 2016. View Article : Google Scholar
|
|
33
|
Chen W, Zheng R, Baade PD, Zhang S, Zeng
H, Bray F, Jemal A, Yu XQ and He J: Cancer statistics in China,
2015. CA Cancer J Clin. 66:115–132. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Sedaghat Y, Bui HH, Mazur C and Monia BP:
Identification of REST-regulated genes and pathways using a
REST-targeted antisense approach. Nucleic Acid Ther. 23:389–400.
2013. View Article : Google Scholar : PubMed/NCBI
|