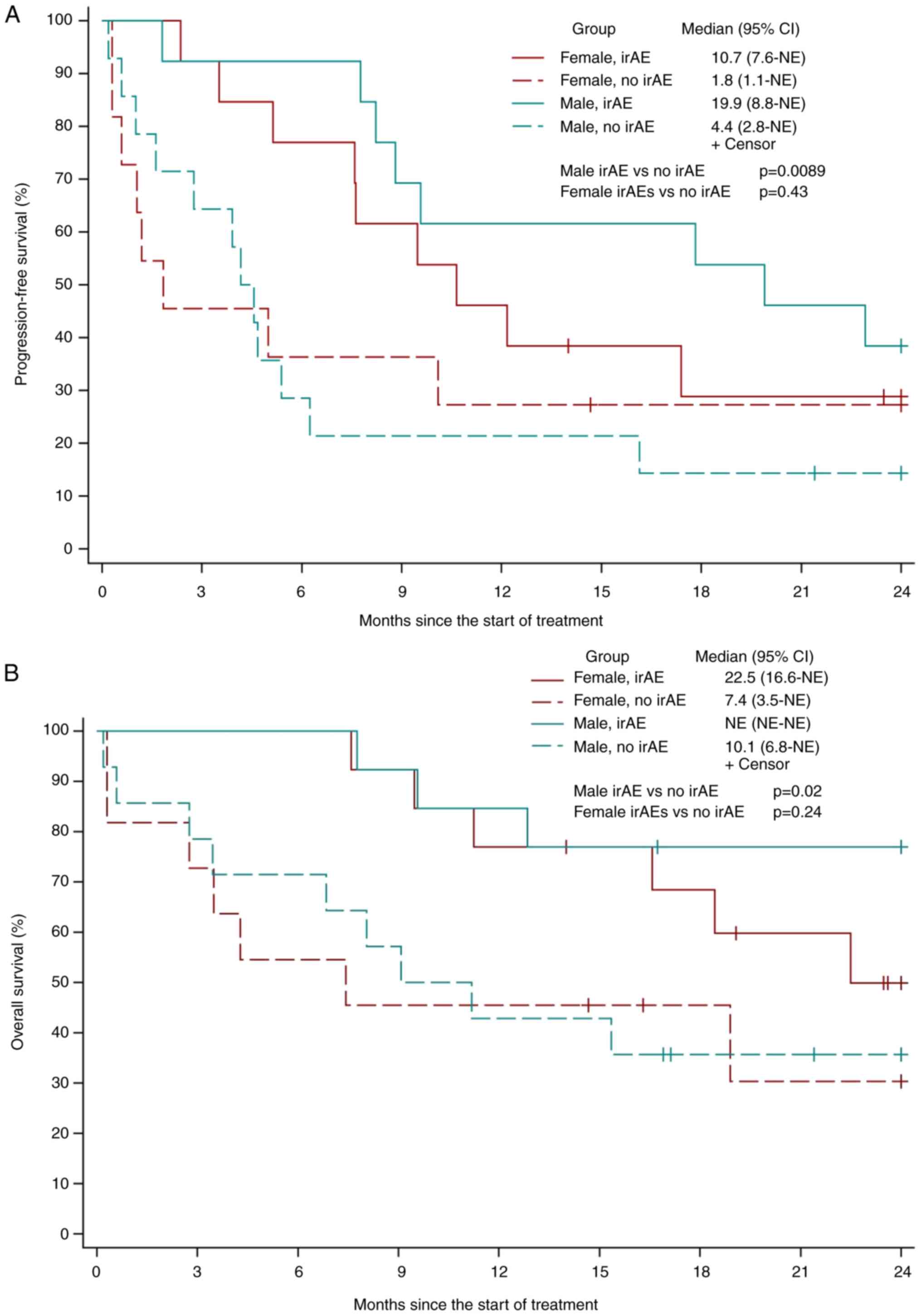
|
1
|
van der Kooij MK, Suijkerbuijk KPM, Aarts
MJB, van den Berkmortel FWPJ, Blank CU, Boers-Sonderen MJ, van
Breeschoten J, van den Eertwegh AJM, de Groot JWB, Haanen JBAG, et
al: Safety and efficacy of checkpoint inhibition in patients with
melanoma and preexisting autoimmune disease: A cohort study. Ann
Intern Med. 174:641–648. 2021. View
Article : Google Scholar : PubMed/NCBI
|
|
2
|
Cortellini A, Buti S, Santini D, Perrone
F, Giusti R, Tiseo M, Bersanelli M, Michiara M, Grassadonia A,
Brocco D, et al: Clinical outcomes of patients with advanced cancer
and pre-existing autoimmune diseases treated with anti-programmed
death-1 immunotherapy: A real-world transverse study. Oncologist.
24:e327–e337. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Beaver JA and Pazdur R: The wild west of
checkpoint inhibitor development. N Engl J Med. 386:1297–1301.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Granier C, De Guillebon E, Blanc C,
Roussel H, Badoual C, Colin E, Saldmann A, Gey A, Oudard S and
Tartour E: Mechanisms of action and rationale for the use of
checkpoint inhibitors in cancer. ESMO Open. 2:e0002132017.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Cancer Institute N, . Common Terminology
Criteria for Adverse Events (CTCAE) Common Terminology Criteria for
Adverse Events (CTCAE) v5.0. 2017.
|
|
6
|
Naidoo J, Murphy C, Atkins MB, Brahmer JR,
Champiat S, Feltquate D, Krug LM, Moslehi J, Pietanza MC, Riemer J,
et al: Society for immunotherapy of cancer (SITC) consensus
definitions for immune checkpoint inhibitor-associated
immune-related adverse events (irAEs) terminology. J Immunother
Cancer. 11:e0063982023. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Tarhini AA, Lee SJ, Hodi FS, Rao UNM,
Cohen GI, Hamid O, Hutchins LF, Sosman JA, Kluger HM, Eroglu Z, et
al: Phase III study of adjuvant ipilimumab (3 or 10 mg/kg) versus
high-dose interferon Alfa-2b for resected high-risk melanoma: North
American Intergroup E1609. J Clin Oncol. 38:567–575. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wolchok JD, Chiarion-Sileni V, Gonzalez R,
Rutkowski P, Grob JJ, Cowey CL, Lao CD, Wagstaff J, Schadendorf D,
Ferrucci PF, et al: Overall survival with combined nivolumab and
ipilimumab in advanced melanoma. N Engl J Med. 377:1345–1356. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Eggermont AMM, Blank CU, Mandalà M, Long
GV, Atkinson VG, Dalle S, Haydon AM, Meshcheryakov A, Khattak A,
Carlino MS, et al: Adjuvant pembrolizumab versus placebo in
resected stage III melanoma (EORTC 1325-MG/KEYNOTE-054): Distant
metastasis-free survival results from a double-blind, randomised,
controlled, phase 3 trial. Lancet Oncol. 22:643–654. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Weber JS, Hodi FS, Wolchok JD, Topalian
SL, Schadendorf D, Larkin J, Sznol M, Long GV, Li H, Waxman IM, et
al: Safety profile of nivolumab monotherapy: A pooled analysis of
patients with advanced melanoma. J Clin Oncol. 35:785–792. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Johnson DB, Nebhan CA, Moslehi JJ and
Balko JM: Immune-checkpoint inhibitors: Long-term implications of
toxicity. Nat Rev Clin Oncol. 19:254–267. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Das S and Johnson DB: Immune-related
adverse events and anti-tumor efficacy of immune checkpoint
inhibitors. J Immunother Cancer. 7:3062019. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Postow MA, Sidlow R and Hellmann MD:
Immune-related adverse events associated with immune checkpoint
blockade. N Engl J Med. 378:158–168. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Mangan BL, McAlister RK, Balko JM, Johnson
DB, Moslehi JJ, Gibson A and Phillips EJ: Evolving insights into
the mechanisms of toxicity associated with immune checkpoint
inhibitor therapy. Br J Clin Pharmacol. 86:1778–1789. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Pantuck M, McDermott D and Drakaki A: To
treat or not to treat: Patient exclusion in immune oncology
clinical trials due to preexisting autoimmune disease. Cancer.
125:3506–3513. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Khan SA, Pruitt SL, Xuan L and Gerber DE:
Prevalence of autoimmune disease among patients with lung cancer:
Implications for immunotherapy treatment options. JAMA Oncol.
2:1507–1508. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ortona E, Pierdominici M, Maselli A,
Veroni C, Aloisi F and Shoenfeld Y: Sex-based differences in
autoimmune diseases. Ann Ist Super Sanita. 52:205–212.
2016.PubMed/NCBI
|
|
18
|
Wahli MN, Hayoz S, Hoch D, Ryser CO,
Hoffmann M, Scherz A, Schwacha-Eipper B, Häfliger S, Wampfler J,
Berger MD, et al: The role of immune checkpoint inhibitors in
clinical practice: An analysis of the treatment patterns, survival
and toxicity rates by sex. J Cancer Res Clin Oncol. Aug
23–2022.(Epub ahead of print). PubMed/NCBI
|
|
19
|
Amin M, Edge S and Greene F: AJCC cancer
staging manual. 8th edition. Springer; 2017, View Article : Google Scholar
|
|
20
|
Eisenhauer EA, Therasse P, Bogaerts J,
Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S,
Mooney M, et al: New response evaluation criteria in solid tumours:
Revised RECIST guideline (version 1.1). Eur J Cancer. 45:228–247.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Duma N, Abdel-Ghani A, Yadav S, Hoversten
KP, Reed CT, Sitek AN, Enninga EAL, Paludo J, Aguilera JV,
Leventakos K, et al: Sex differences in tolerability to
anti-programmed cell death protein 1 therapy in patients with
metastatic melanoma and non-small cell lung cancer: Are we all
equal? Oncologist. 24:e1148–e1155. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Alexander S, Swami U, Kaur A, Gao Y,
Fatima M, Ginn MM, Stein JE, Grivas P, Zakharia Y and Singh N:
Safety of immune checkpoint inhibitors in patients with cancer and
pre-existing autoimmune disease. Ann Transl Med. 9:10332021.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Boland P, Pavlick AC, Weber J and
Sandigursky S: Immunotherapy to treat malignancy in patients with
pre-existing autoimmunity. J Immunother Cancer. 8:e0003562020.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Johnson DB, Sullivan RJ, Ott PA, Carlino
MS, Khushalani NI, Ye F, Guminski A, Puzanov I, Lawrence DP,
Buchbinder EI, et al: Ipilimumab therapy in patients with advanced
melanoma and preexisting autoimmune disorders. JAMA Oncol.
2:234–240. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Tison A, Quéré G, Misery L, Funck-Brentano
E, Danlos FX, Routier E, Robert C, Loriot Y, Lambotte O, Bonniaud
B, et al: Safety and efficacy of immune checkpoint inhibitors in
patients with cancer and preexisting autoimmune disease: A
nationwide, multicenter cohort study. Arthritis Rheumatol.
71:2100–2111. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Tang H, Zhou J and Bai C: The efficacy and
safety of immune checkpoint inhibitors in patients with cancer and
preexisting autoimmune disease. Front Oncol. 11:6258722021.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Fan Y, Xie W, Huang H, Wang Y, Li G, Geng
Y, Hao Y and Zhang Z: Association of immune related adverse events
with efficacy of immune checkpoint inhibitors and overall survival
in cancers: A systemic review and meta-analysis. Front Oncol.
11:6330322021. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Unger JM, Vaidya R, Albain KS, LeBlanc M,
Minasian LM, Gotay CC, Henry NL, Fisch MJ, Lee SM, Blanke CD and
Hershman DL: Sex differences in risk of severe adverse events in
patients receiving immunotherapy, targeted therapy, or chemotherapy
in cancer clinical trials. J Clin Oncol. 40:1474–1486. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Ileana Dumbrava EE, Suarez-Almazor ME,
Painter J, Johanns T, Dougan ML, Cappelli L, Bingham CO, Wang Y,
Gupta S, Warner BM, et al: A phase Ib study of nivolumab in
patients with autoimmune disorders and advanced malignancies
(AIM-NIVO). J Clin Oncol. 38 (Suppl):TPS31582020. View Article : Google Scholar
|
|
30
|
Özdemir BC, Coukos G and Wagner AD:
Immune-related adverse events of immune checkpoint inhibitors and
the impact of sex-what we know and what we need to learn. Ann
Oncol. 29:10672018. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Triggianese P, Novelli L, Galdiero MR,
Chimenti MS, Conigliaro P, Perricone R, Perricone C and Gerli R:
Immune checkpoint inhibitors-induced autoimmunity: The impact of
gender. Autoimmun Rev. 19:1025902020. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Polanczyk MJ, Hopke C, Vandenbark AA and
Offner H: Estrogen-mediated immunomodulation involves reduced
activation of effector T cells, potentiation of Treg cells, and
enhanced expression of the PD-1 costimulatory pathway. J Neurosci
Res. 84:370–378. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Ozdemir BC and Dotto GP: Sex hormones and
anticancer immunity. Clin Cancer Res. 25:4603–4610. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Centanni M, Moes DJAR, Trocóniz IF,
Ciccolini J and van Hasselt JGC: Clinical pharmacokinetics and
pharmacodynamics of immune checkpoint inhibitors. Clin
Pharmacokinet. 58:835–857. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Hurkmans DP, Sassen SDT, de Joode K,
Putter L, Basak EA, Wijkhuijs AJM, Joerger M, Debets R, Koch BCP,
Van der Leest CH, et al: Prospective real-world study on the
pharmacokinetics of pembrolizumab in patients with solid tumors. J
Immunother Cancer. 9:e0023442021. View Article : Google Scholar : PubMed/NCBI
|