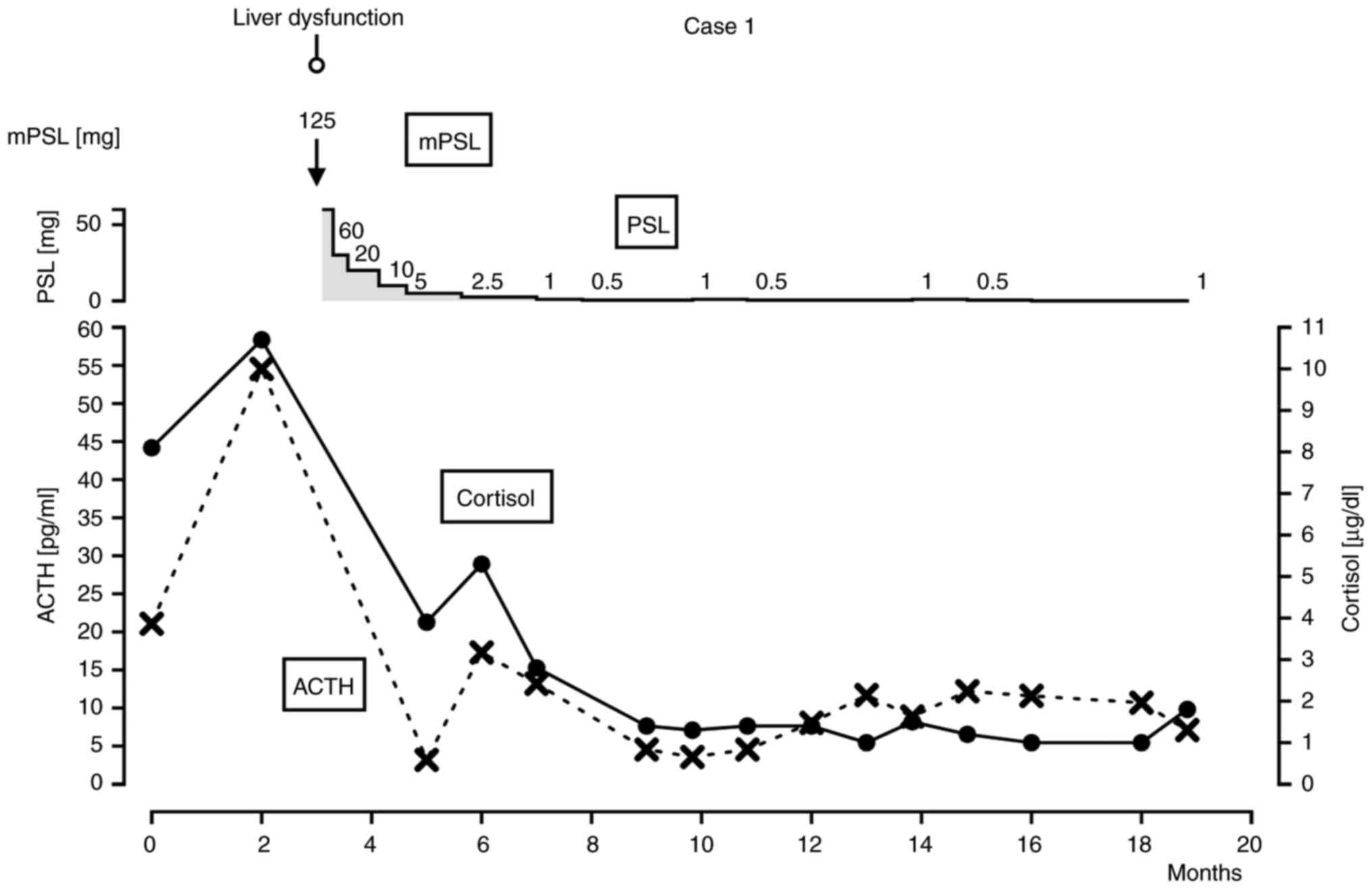
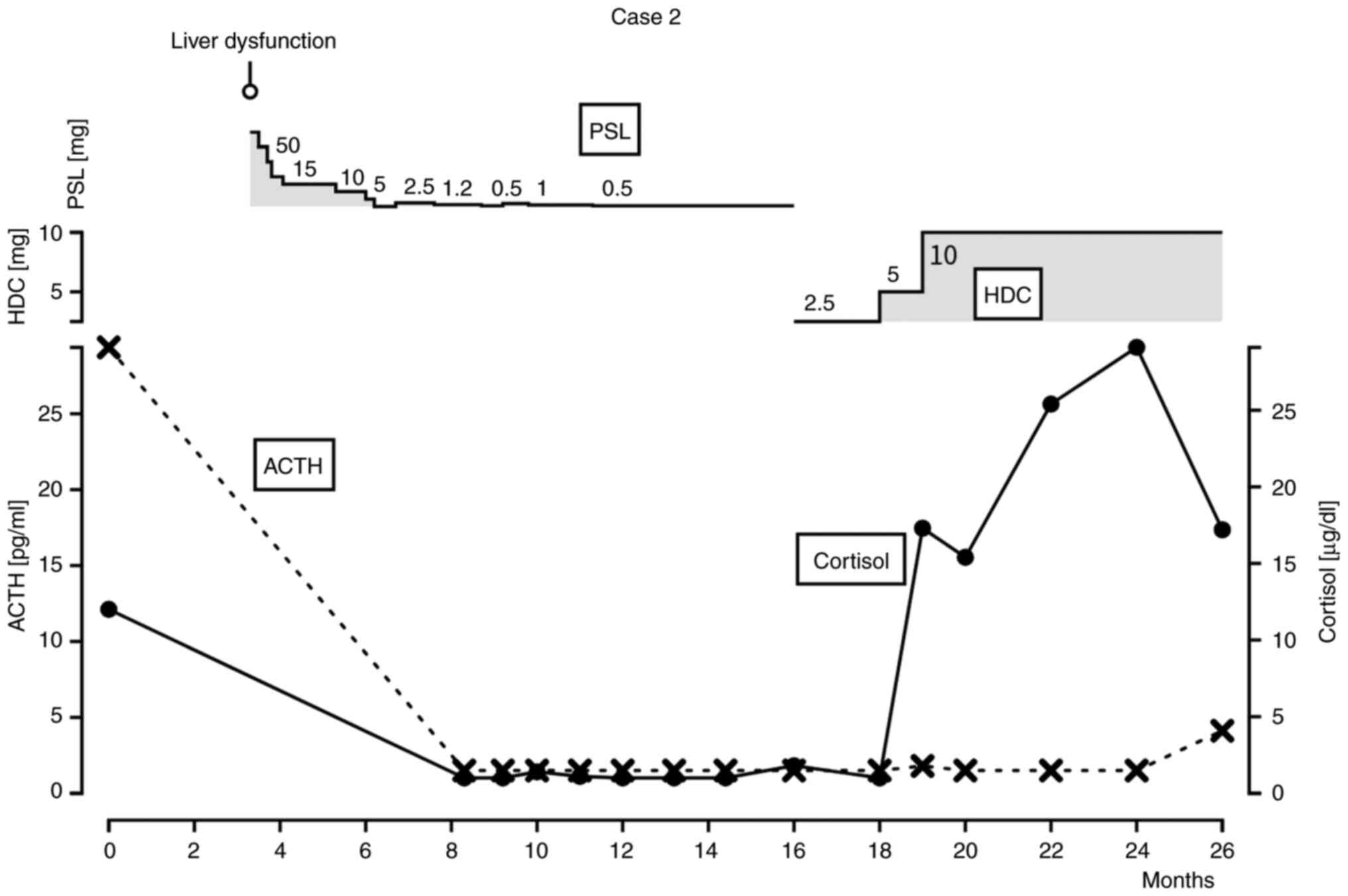
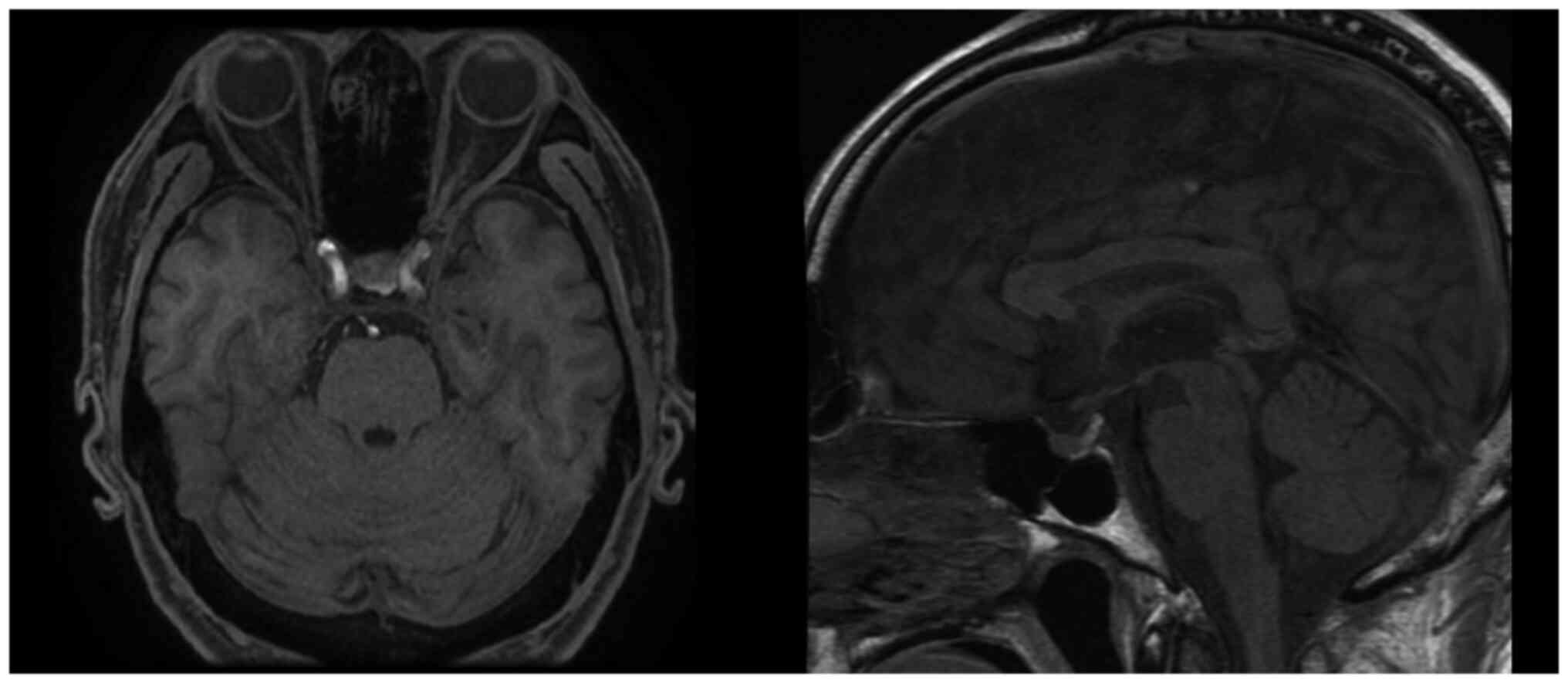
|
1
|
Motzer RJ, Tannir NM, McDermott DF, Arén
Frontera O, Melichar B, Choueiri TK, Plimack ER, Barthélémy P,
Porta C, George S, et al: Nivolumab plus ipilimumab versus
sunitinib in advanced renal-cell carcinoma. N Engl J Med.
378:1277–1290. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Makker V, Colombo N, Casado Herráez A,
Santin AD, Colomba E, Miller DS, Fujiwara K, Pignata S, Baron-Hay
S, Ray-Coquard I, et al: Lenvatinib plus pembrolizumab for advanced
endometrial cancer. N Engl J Med. 386:437–448. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Rini BI, Plimack ER, Stus V, Gafanov R,
Hawkins R, Nosov D, Pouliot F, Alekseev B, Soulières D, Melichar B,
et al: Pembrolizumab plus axitinib versus sunitinib for advanced
renal-cell carcinoma. N Engl J Med. 380:1116–1127. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Li X, Shao C, Shi Y and Han W: Lessons
learned from the blockade of immune checkpoints in cancer
immunotherapy. J Hematol Oncol. 11:312018. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Li Y, Li F, Jiang F, Lv X, Zhang R, Lu A
and Zhang G: A mini-review for cancer immunotherapy: Molecular
understanding of PD-1/PD-L1 pathway & translational
blockade of immune checkpoints. Int J Mol Sci. 17:11512016.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Cukier P, Santini FC, Scaranti M and Hoff
AO: Endocrine side effects of cancer immunotherapy. Endocr Relat
Cancer. 24:T331–T347. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Arima H, Iwama S, Inaba H, Ariyasu H,
Makita N, Otsuki M, Kageyama K, Imagawa A and Akamizu T: Management
of immune-related adverse events in endocrine organs induced by
immune checkpoint inhibitors: clinical guidelines of the Japan
endocrine society. Endocr J. 66:581–586. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bertrand A, Kostine M, Barnetche T,
Truchetet ME and Schaeverbeke T: Immune related adverse events
associated with anti-CTLA-4 antibodies: Systematic review and
meta-analysis. BMC Med. 13:2112015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Barroso-Sousa R, Barry WT, Garrido-Castro
AC, Hodi FS, Min L, Krop IE and Tolaney SM: Incidence of endocrine
dysfunction following the use of different immune checkpoint
inhibitor regimens: A systematic review and meta-analysis. JAMA
Oncol. 4:173–182. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Powles T, Plimack ER, Soulières D, Waddell
T, Stus V, Gafanov R, Nosov D, Pouliot F, Melichar B, Vynnychenko
I, et al: Pembrolizumab plus axitinib versus sunitinib monotherapy
as first-line treatment of advanced renal cell carcinoma
(KEYNOTE-426): Extended follow-up from a randomised, open-label,
phase 3 trial. Lancet Oncol. 21:1563–1573. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Iwama S, De Remigis A, Callahan MK, Slovin
SF, Wolchok JD and Caturegli P: Pituitary expression of CTLA-4
mediates hypophysitis secondary to administration of CTLA-4
blocking antibody. Sci Transl Med. 6:230ra452014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Caturegli P, Di Dalmazi G, Lombardi M,
Grosso F, Larman HB, Larman T, Taverna G, Cosottini M and Lupi I:
Hypophysitis secondary to cytotoxic T-lymphocyte-associated protein
4 blockade: Insights into pathogenesis from an autopsy series. Am J
Pathol. 186:3225–3235. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kanie K, Iguchi G, Bando H, Urai S, Shichi
H, Fujita Y, Matsumoto R, Suda K, Yamamoto M, Fukuoka H, et al:
Mechanistic insights into immune checkpoint inhibitor-related
hypophysitis: A form of paraneoplastic syndrome. Cancer Immunol
Immunother. 70:3669–3677. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ohara N, Ohashi K, Fujisaki T, Oda C,
Ikeda Y, Yoneoka Y, Hashimoto T, Hasegawa G, Suzuki K and Takada T:
Isolated adrenocorticotropin deficiency due to nivolumab-induced
hypophysitis in a patient with advanced lung adenocarcinoma: A case
report and literature review. Intern Med. 57:527–535. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Postow MA, Sidlow R and Hellmann MD:
Immune-related adverse events associated with immune checkpoint
blockade. N Engl J Med. 378:158–168. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Brahmer JR, Lacchetti C, Schneider BJ,
Atkins MB, Brassil KJ, Caterino JM, Chau I, Ernstoff MS, Gardner
JM, Ginex P, et al: Management of immune-related adverse events in
patients treated with immune checkpoint inhibitor therapy: American
society of clinical oncology clinical practice guideline. J Clin
Oncol. 36:1714–1768. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Takagi T, Yoshida K, Kondo T, Fukuda H,
Ishihara H, Kobayashi H, Iizuka J, Ishida H and Tanabe K:
Hypopituitarism in patients with metastatic renal cell carcinoma
treated with ipilimumab and nivolumab combination therapy. Jpn J
Clin Oncol. 51:1744–1750. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Motzer RJ, Rini BI, McDermott DF, Arén
Frontera O, Hammers HJ, Carducci MA, Salman P, Escudier B,
Beuselinck B, Amin A, et al: Nivolumab plus ipilimumab versus
sunitinib in first-line treatment for advanced renal cell
carcinoma: Extended follow-up of efficacy and safety results from a
randomised, controlled, phase 3 trial. Lancet Oncol. 20:1370–1385.
2019. View Article : Google Scholar : PubMed/NCBI
|