Introduction
The effect of anesthetics on cancer has been a topic
of clinical interest. A number of retrospective studies reported
that the use of volatile anesthetic-based general anesthesia was
associated with higher incidence of cancer recurrence and worse
survival compared to the use of intravenous anesthetic-based
anesthesia (1–3). These observations triggered
researchers to investigate the underlying mechanism of how
anesthetics affect cancer. So far clear volatile anesthetic targets
have not been shown in vivo.
Breast cancer is the most frequently diagnosed
cancer and the cause of mortality among all cancers in females
(4). Although spontaneous tumor
growth models or xenogeneic tumor implantation models have been
used to study breast cancer in mice (5), they already have immunologically
altered background. Instead, EO771 tumor cell implantation congenic
model allows us to study tumor growth in fully immunocompetent
mice. EO771 cells are breast adenocarcinoma cells derived from a
spontaneous mammary tumor from a female C57BL/6 mouse (6) and have been used for congenic breast
cancer model (7). Now it is well
recognized that anesthetics can affect leukocyte functions
(8,9). Thus, using congenic model to study the
effect of anesthetics on tumor growth would be logical.
In this study, we examined the impact of commonly
used volatile anesthetic isoflurane on tumor growth and its
underlying mechanism in vivo. We previously reported that
volatile anesthetic isoflurane directly binds to and inhibits
critical adhesion molecules leukocyte function-associated antigen-1
(LFA-1) (10–12) and macrophage-1 antigen (Mac-1)
(13). LFA-1 and Mac-1 are members
of β2 integrins, which belong to a heterodimeric adhesion molecule
family consisting of α- and β2-subunits. LFA-1 is also called αLβ2
or CD11a/CD18 and ubiquitously expressed on leukocytes (14). Mac-1 is also called αMβ2, CD11b/CD18
or complement receptor 3 (CR3) and expressed primarily on myeloid
cells (15). Thus, we also examined
the role of LFA-1 and Mac-1 in tumor growth.
Materials and methods
Mice
Wild type, CD11a (αL) knockout (KO) mice (16) and CD11b (αM) KO mice (17) on the C57BL6 background were obtained
from Jackson Laboratory (Bar Harbor, Maine, USA). They were housed
under specific pathogen-free conditions, with 12-h light and dark
cycles. All animal protocols were approved by the Institutional
Animal Care and Use Committee (IACUC) at Boston Children's Hospital
(Protocols 16-03-3120 and 00001574 ‘Anesthetics and tumor
recurrence or metastasis’).
EO771 tumor implantation model
The experiments were performed between August 2016
and July 2018. EO771 cells were cultured in RPMI1640/10% FBS. On
the day of tumor implantation, mCherry-EO771 cells (7) (mCherry-EO771 cells were kindly given
by Dr. Johnstone at University of Melbourne) were collected and
suspended in Matrigel matrix (Corning, Inc., Corning, NY).
1×105 of EO771 cells per mouse suspended in 50 ul of
Matrigel matrix were implanted at the 4th nipple fat pad in the
morning of the experimental days (7). Given subcutaneous tumor injection is
minimally invasive (18), for the
injection, mice were placed in a quiet room and held in
researcher's hand for injection with a 30G needle. No anesthetics
were used as approved by the IACUC. Then, tumor size was monitored
every other day. Mice behaved actively during our observation.
Tumor volume was calculated ½(length × width2), as
previously described (19). For
IVIS (in vivo imaging system) based tumor imaging, mice were
implanted with cells labeled with firefly luciferase and subjected
to intraperitoneal injection of Luciferin (15–150 mg/kg) 10 min
before the measurement. During the imaging, mice were anesthetized
with isoflurane (4% induction, 2–3% maintenance).
In some experiment, either 100 µg of isotype control
or CD11a monoclonal antibody (mAb) (clone M17/4) was given on day 7
and day 10 after tumor implantation. Some mice were also exposed to
1% isoflurane (induction and maintenance) or 2.1% sevoflurane
(induction and maintenance) for 4 h on day 7 after tumor
implantation. Because the minimum alveolar concentration (MAC; the
concentration at which 50% of mice do not respond to tail clamping)
is 1.3% for isoflurane (20) and 1
MAC is 2.8% for sevoflurane (21),
2.1% sevoflurane matches the potency of 1% isoflurane. We intended
to provide them to mice at clinically relevant doses, not for full
general anesthesia. The total number of mice used was described in
each Figure legend. We observed tumor growth for 2 weeks expect the
experiment using CD11b KO mice. For CD11b KO mice experiment, we
observed up to 3 weeks due to slower tumor growth. If tumors
exhibited abrasion and fluid leakage, we euthanized and excluded
mice from the study. In this study, we euthanized one mouse due to
the leakage from the tumor bed. We observed redness (abrasion) at
the leakage site but did not measure the size of the abrasion. At
the end of observation, all mice were euthanized with
CO2 (30–70% of the chamber volume per minute,
approximately 4–5 min). Euthanasia was confirmed by the lack of
movement including respiration and heartbeat.
Tumor bed histology analysis
Tumor tissue beds were fixed using 4%
paraformaldehyde. Hematoxylin & Eosin (H&E) staining was
done using the Leica ST5020 Multi-staining machine in Boston
Children's Hospital pathology core.
Eicosanoid measurements of mass at
tumor bed
Tumor beds were removed and kept in −80°C freezer
until use. Then, tumor mass was subjected to mechanical disruption
for lipid extraction. The lipids were extracted with methanol and
diluted with water containing 0.1% formic acid to yield a final
methanol concentration of 20%. Reverse-phase mass spectrometry
(MS)-based quantitation technique for eicosanoids was previously
described (22). After addition of
deuterium-labeled internal standards, the samples were loaded on
Oasis HLB cartridge (Waters, Milford, MA). The column was washed
with 1 ml of water, 1 ml of 15% methanol, and 1 ml of petroleum
ether and then eluted with 0.2 ml of methanol containing 0.1%
formic acid. Eicosanoids were quantified by reverse-phase
HPLC-electrospray ionization-tandem MS method.
Reverse transcription-quantitative PCR
(RT-qPCR)
Tumor bed tissues were collected and kept in −80°C
until use. Tissues were suspended in Trizol (Thermofischer,
Waltham, MA) and homogenized. Then, samples were subjected to RNA
purification per the company's protocol. A total of 1 µg RNA was
then converted to first-strand cDNA. RT-qPCR was performed using
SYBR Green PCR Master Mix (Thermo Fisher Scientific) on StepOnePlus
System (Applied Biosystems, Waltham, MA). For data normalization,
GAPDH was used as an internal reference, and the fold change in
gene expression was calculated using the comparative Ct method
(2-ddCt) (23). Primers used for
RT-qPCR were TNF-α Forward
CCCTCACACTCAGATCATCTTCT,ReverseGCTACGACGTGGGCTACAG; IL-1β forward
GCAACTGTTCCTGAACTCAACT, reverse ATCTTTTGGGGTCCGTCAACT; IL-6 forward
GCTACCAAACTGGATATAATCAGG A reverse CCAGGTAGCTATGGTACTCCAGAA; CXCR1
forward TCTGGACTAATCCTGAGGGTG, reverse GCCTGTTGGTTATTGGAACTCTC;
G-CSF forward ATGGCTCAACTTTCTGCCCAG, reverse CTGACAGTGACCAGGGGAAC;
GAPDH forward GCACAGTCAAGGCCGAGAAT, GAPDH reverse
GCCTTCTCCATGGTGGTGAA.
In vitro EO771 cell growth
assessment
We examined the growth of EO771 cells with or
without isoflurane exposure. Isoflurane exposure was done in an
airtight chamber as we previously performed (24,25).
Isoflurane concentration was measured by infrared spectroscopy
(Ultima, Datex Instrument Corp., Helsinki, Finland). Cells were
detached by trypsin and the number of live cells was counted
following trypan blue staining using a hemocytometer.
Statistical analysis
Data are presented as the mean ± SD. Unpaired
Student's t-test and two-way mixed ANOVA with Bonferroni post hoc
analysis were used. Statistical significance was defined as P<
0.05. All the statistical calculations were performed using PRISM5
software (GraphPad Software, La Jolla, CA).
Results
Isoflurane exposure facilitated breast
cancer growth
We examined the effect of commonly used volatile
anesthetic isoflurane on breast cancer growth (Fig. 1A). We administered 1% isoflurane for
4 h to mice at 7 days after EO771 implantation, mimicking the
duration for patients receiving breast cancer resection. As
expected, isoflurane significantly facilitated breast cancer growth
(tumor size at day 13, 343.3 +/- 132.9 mm3, maximum 683
mm3 for no exposure and 686.7 +/- 265.8 mm3,
maximum 1,366 mm3 for isoflurane exposure) (Fig. 1B). We also tested another volatile
anesthetic sevoflurane. Sevoflurane also significantly facilitated
breast cancer growth (tumor size at day 13, 331.0 +/- 122.0
mm3, maximum 614 mm3 for no exposure and
731.4 +/- 292.6 mm3, maximum 1,503 mm3 for
sevoflurane exposure) (Fig.
1C).
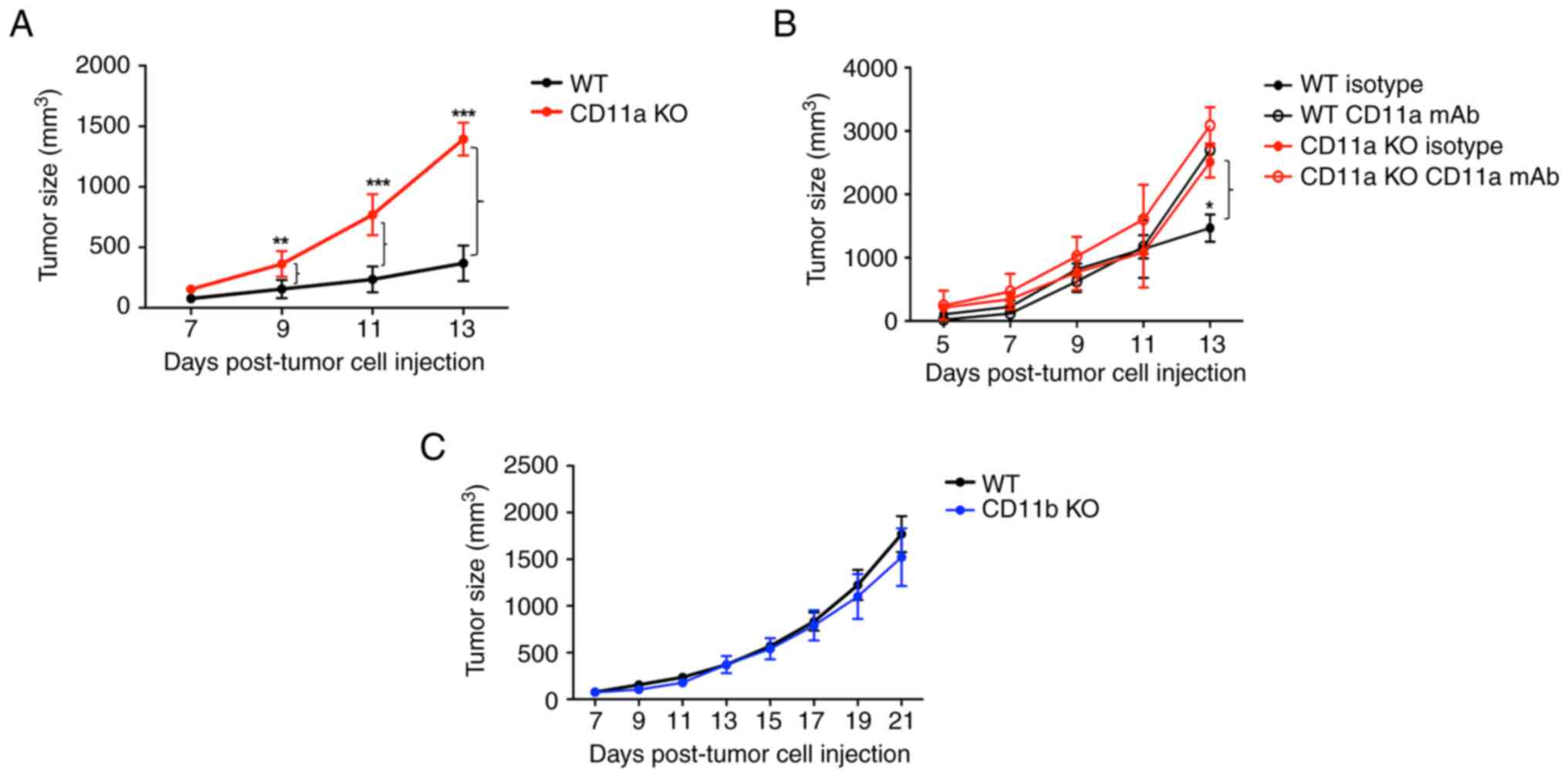
LFA-1 deficiency was associated with
faster tumor growth, but Mac-1 deficiency was not
We previously showed that isoflurane directly bound
to and inhibited adhesion molecules LFA-1 and Mac-1. Thus, we first
examined the role of LFA-1 and Mac-1 in breast cancer growth. The
deficiency of LFA-1 significantly facilitated the growth of EO771
cells as the tumor size at day 13 was 369.4 +/- 146.4
mm3 (maximum 685 mm3) for WT mice and 1,393.8
+/- 134.6 mm3 (maximum 1,639 mm3) for CD11a
KO mice (Fig. 2A). Because KO mice
could have compensatory changes, we also examined the effect of
LFA-1 using CD11a monoclonal blocking antibody in both WT and CD11a
KO mice. In line with the finding in Fig. 2A, CD11a mAb administration
facilitated the growth of EO771 cells in WT mice (tumor size at day
13 1,467.2 +/- 372.6 mm3, maximum 1,725 mm3
for isotype antibody group and 2,697.0 +/- 109.2 mm3,
maximum 2,725 mm3 for CD11a mAb group) (Fig. 2B). No difference was observed in
CD11a KO mice (CD11a KO with isotype group, 2,510.9+/-350.0
mm3, maximum 2,758 mm3, and CD11a KO with
CD11a mAb group, 3,088.0 +/- 405.4 mm3, maximum 3,374
mm3). In contrast, Mac-1 deficiency did not affect tumor
growth (tumor size at day 21 1,769.3 +/- 545.4 mm3,
maximum 2,798 mm3 for WT and 1,521.9 +/- 689.6
mm3, maximum 2,343 mm3 for CD11b KO mice)
(Fig. 2C). Taken together, we found
that both LFA-1 deficiency and inhibition significantly enhanced
the growth of EO771 cells.
Breast cancer bed showed higher
pro-tumor cytokine levels and PGE2/LTD2
levels
Because proinflammatory cytokines and a subset of
lipid mediators have been shown associated with the growth of
tumor, we examined their levels in CD11a KO mice. Pro-tumor
cytokines IL-6, CXCR1 and G-CSF levels were significantly elevated
in the tumor bed of CD11a KO mice (Fig.
3A). We also examined prostaglandin (PG) and leukotriene (LT)
levels. We found that PGE2 and LTD4 levels
were significantly elevated in CD11a KO mice (Fig. 3B). These data are in line with
clinical data as PGE2 mediated signal is important in
propagating breast cancer (26) and
LTD4 level was elevated in patients with breast cancer
(27).
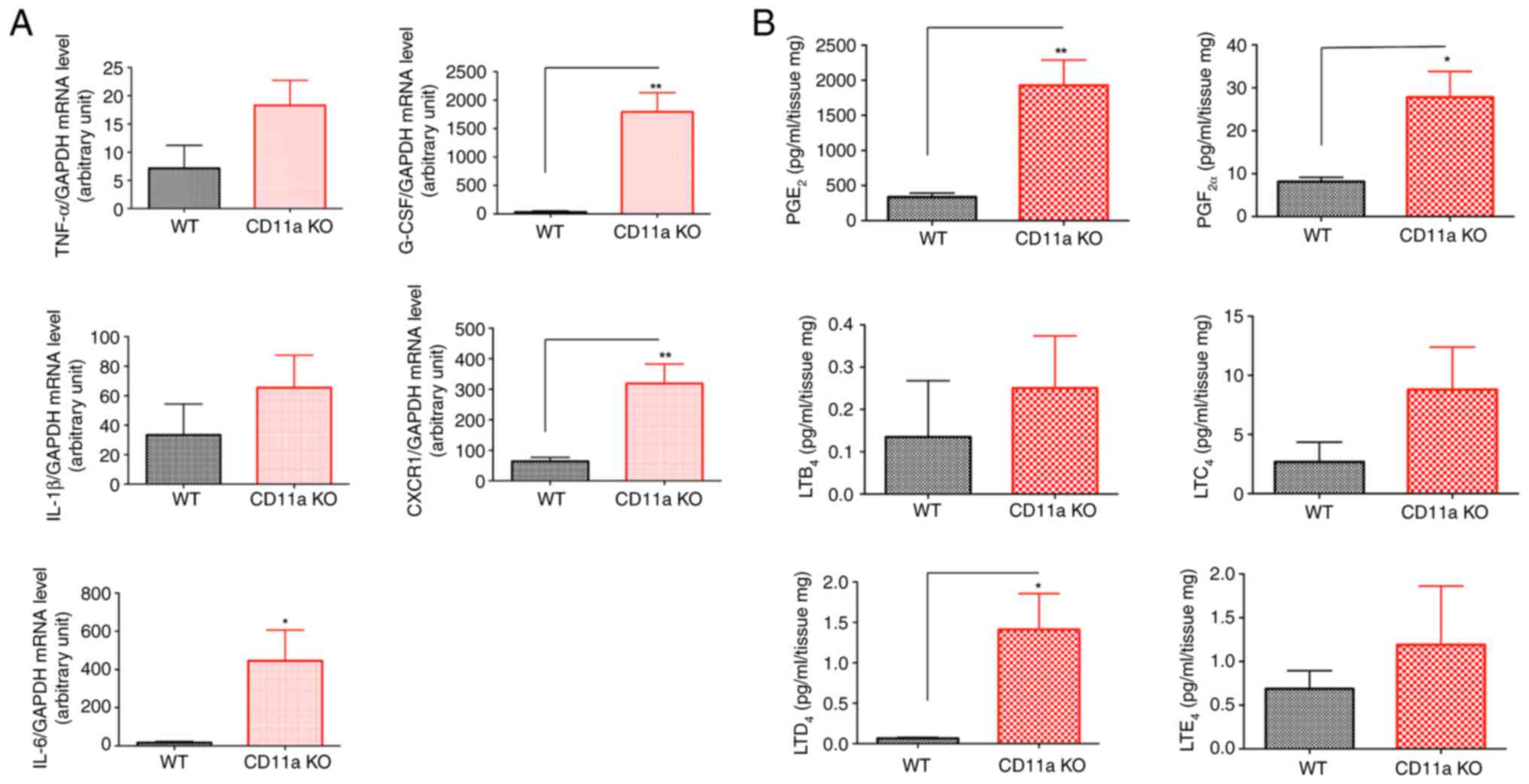 | Figure 3.Cytokine and eicosanoid levels in the
tumor bed tissues. No anesthetic exposure was performed in the
experiments presented here. (A) Tumor bed tissues at 2 weeks after
implantation were subjected to reverse transcription-quantitative
PCR. GAPDH was used as the internal control housekeeping gene. Data
are presented as the mean ± SD of quadruplicates. An unpaired
Student's t-test was performed. *P<0.05 and **P<0.01. (B)
Eicosanoid levels of tumor tissues were measured. Data are
presented as the mean ± SD of quadruplicates. An unpaired Student's
t-test was performed. *P<0.05 and **P<0.01. CXCR1, C-X-C
motif chemokine receptor 1; G-CSF, granulocyte colony stimulating
factor; KO, knockout; LTB4, leukotriene B4;
LTC4, leukotriene C4; LTD4,
leukotriene D4; LTE4, leukotriene
E4; PGE2, prostaglandin E2;
PGF2α, prostaglandin F2α; WT, wild-type. |
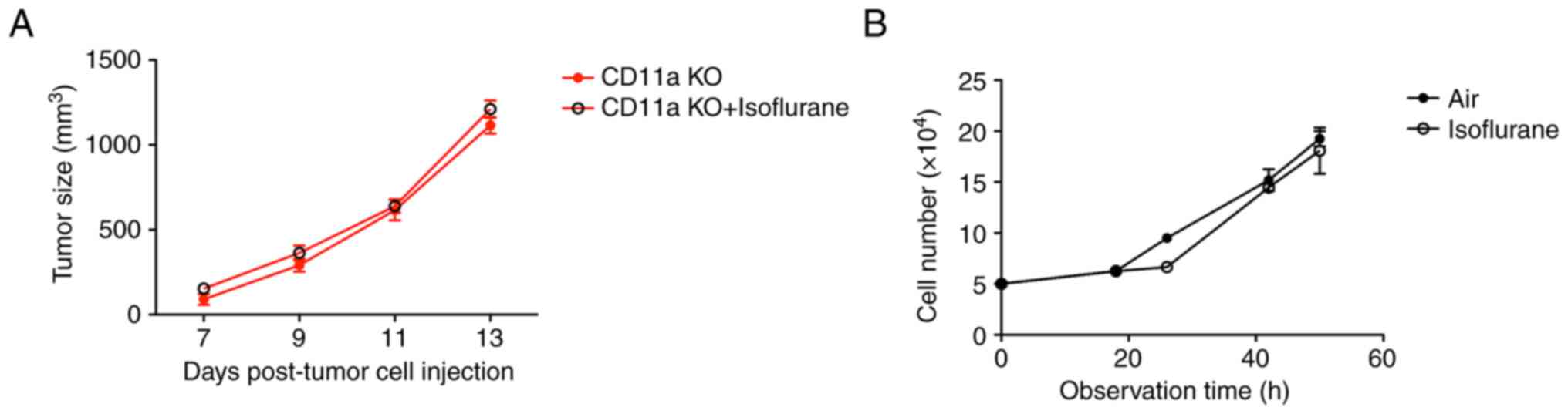
Isoflurane did not further increase
breast cancer size
The data so far indicated that LFA-1 would be
isoflurane target to modulate tumor size. To test this hypothesis,
we examined the effect of isoflurane in CD11a KO mice. Supportive
of our hypothesis, isoflurane did not significantly affect the size
of tumor in CD11a KO mice (tumor size at day 3, 1,115.0 +/- 1,07.7
mm3, maximum 1,304 mm3 for CD11a KO mice, and
1,210.6 +/- 115.0 mm3, maximum 1,326 mm3 for
CD11a KO mice with isoflurane exposure) (Fig. 4A). LFA-1 is exclusively expressed on
leukocytes. Thus, we also tested if isoflurane directly affected
tumor size in vitro. As expected, isoflurane did not
increase the EO771 cell number (Fig.
4B). This suggests that LFA-1 would be a major isoflurane
target to modulate breast cancer growth in vivo.
Discussion
In this study, we showed that volatile anesthetic
isoflurane and sevoflurane exposure significantly enhanced breast
cancer growth. We also suggested that LFA-1 facilitate breast
cancer growth via affecting LFA-1.
The role of anesthetic selection in cancer resection
surgery has been a hot topic. Wigmore et al (1) reported that the use of intravenous
anesthetics was significantly associated with better overall
survival and less cancer recurrence than the use of volatile
anesthetics. This landmark paper ignited the discussion of whether
or not intravenous or volatile anesthetics should be used for
general anesthesia for cancer resection surgery. A number of
retrospective studies examined various type of cancer surgeries,
largely favoring the use of intravenous anesthetics (28). In parallel, many investigators
examined the effect of anesthetics using in vitro cell
culture system and in vivo animal models. However, the
mechanism of anesthetic-mediated tumor growth has been less studied
in vivo. We previously demonstrated that commonly used
volatile anesthetics isoflurane and sevoflurane directly bind to
and inhibit LFA-1 (10–12), while an intravenous anesthetic
propofol did not affect LFA-1 function at clinically relevant doses
(29). We previously demonstrated
the importance of LFA-1 as a volatile anesthetic target relevance
in K562 cells, leukemia cells by showing that the inhibition of
LFA-1 by isoflurane and sevoflurane attenuated natural killer (NK)
cell- mediated cytotoxicity (30).
In our data, we showed both isoflurane and sevoflurane facilitated
breast cancer growth. While isoflurane also bound to and blocked
Mac-1 (11,12), sevoflurane did not bind to and
inhibit Mac-1 (10,13). Taken together, our data suggested
that LFA-1 served as a target for both isoflurane and sevoflurane
to facilitate breast cancer growth.
As LFA-1 is exclusively expressed on leukocytes, we
expect that isoflurane significantly enhanced tumor growth via
altering the phenotype of leukocytes. The analysis of tumor beds
showed an increase in PGE2 and LTD4 levels in
CD11a KO mice compared to WT mice. However, we still do not know
what triggered this change. As LFA-1 is ubiquitously expressed on
leukocytes, it is imperative to examine the role of LFA-1 in all
leukocyte types. For example, LFA-1 is involved in NK cell-mediated
tumor cytotoxicity (31,32). LFA-1 activation enriches
tumor-specific T cells to improve anti-tumor responses (33). Both neutrophils and macrophages play
a significant role in cancer immunity (34,35).
However, how LFA-1 on neutrophils and macrophages affect cancer
growth is not known. In the future, it would be critical to
determine how LFA-1 affects cancer growth via leukocytes in
vivo. In line with an increase in PGE2 in CD11a KO
mice, we previously showed that PGE2 levels were
significantly increased by isoflurane (21).
We need to note a few issues. While we measured the
size of tumor to calculate the volume in the same way throughout
the study, we did not use IVIG for additional confirmation.
Although it is very common to study the effect of anesthetics on
tumor growth as in our case, it is important to point out that
anesthetics are usually given for tumor resection. To be completely
in line with this scenario, it would be important to examine the
effect of anesthetics using tumor resection model. However, a
simple tumor resection and recurrence model has not been reported
in breast cancer yet. In the model using 4T1 breast cancer cells, a
nephrectomy has also been done to see tumor recurrence (36). In this study, we used the EO771 cell
model, but it would be important to examine different types of
cancer given each cancer is very different. LFA-1 binds to its
ligand intercellular adhesion molecule-1 (ICAM-1). The expression
of ICAM-1 can vary. For example, ICAM-1 is expressed more in triple
negative breast cancer cells compared with other types (37). Although our data highly supported
that both isoflurane and sevoflurane affected LFA-1 and facilitated
tumor growth based on our previous structural studies (10–12),
we did not show the direct binding of volatile anesthetics in
vivo. Therefore, confirmatory experiment is needed to
explicitly support the direct interaction between LFA-1 and
isoflurane (sevoflurane) in vivo.
In summary, we showed that isoflurane significantly
facilitated breast cancer growth via affecting LFA-1.
Acknowledgements
The authors would like to thank Ms. Janice Bautista
(Department of Anesthesiology, Critical Care and Pain Medicine,
Boston Children's Hospital) for technical support.
Funding
The present study was supported by the National Institute of
General Medical Sciences (grant no. K08GM101345).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
SK performed experiments and wrote the manuscript.
WW performed experiments. LH performed experiments. TO performed
experiments and edited the manuscript. KY designed the study,
performed experiments and wrote the manuscript. SK, LH and KY
confirmed the authenticity of all the raw data. All authors read
and approved the final version of the manuscript.
Ethics approval and consent to
participate
For animal experiments, Boston Children's Hospital
IACUC approval (approved protocol nos. 16-03-3120 and 00001574;
Boston, MA, USA) was obtained.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Wigmore TJ, Mohammed K and Jhanji S:
Long-term survival for patients undergoing volatile versus IV
anesthesia for cancer surgery: A retrospective analysis.
Anesthesiology. 124:69–79. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Jun IJ, Jo JY, Kim JI, Chin JH, Kim WJ,
Kim HR, Lee EH and Choi IC: Impact of anesthetic agents on overall
and recurrence-free survival in patients undergoing esophageal
cancer surgery: A retrospective observational study. Sci Rep.
7:140202017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wu ZF, Lee MS, Wong CS, Lu CH, Huang YS,
Lin KT, Lou YS, Lin C, Chang YC and Lai HC: Propofol-based total
intravenous anesthesia is associated with better survival than
desflurane anesthesia in colon cancer surgery. Anesthesiology.
129:932–941. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ferlay J, Colombet M, Soerjomataram I,
Mathers C, Parkin DM, Piñeros M, Znaor A and Bray F: Estimating the
global cancer incidence and mortality in 2018: GLOBOCAN sources and
methods. Int J Cancer. 144:1941–1953. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Sakamoto K, Schmidt JW and Wagner KU:
Mouse models of breast cancer. Methods Mol Biol. 1267:47–71. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Casey AE, Laster WR Jr and Ross GL:
Sustained enhanced growth of carcinoma EO771 in C57 black mice.
Proc Soc Exp Biol Med. 77:358–362. 1951. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Johnstone CN, Smith YE, Cao Y, Burrows AD,
Cross RS, Ling X, Redvers RP, Doherty JP, Eckhardt BL, Natoli AL,
et al: Functional and molecular characterisation of EO771.LMB
tumours, a new C57BL/6-mouse-derived model of spontaneously
metastatic mammary cancer. Dis Model Mech. 8:237–251.
2015.PubMed/NCBI
|
|
8
|
Stollings LM, Jia LJ, Tang P, Dou H, Lu B
and Xu Y: Immune modulation by volatile anesthetics.
Anesthesiology. 125:399–411. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yuki K and Eckenhoff RG: Mechanisms of the
immunological effects of volatile anesthetics: A review. Anesth
Analg. 123:326–335. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yuki K, Astrof NS, Bracken C, Soriano SG
and Shimaoka M: Sevoflurane binds and allosterically blocks
integrin lymphocyte function-associated antigen-1. Anesthesiology.
113:600–609. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yuki K, Astrof NS, Bracken C, Yoo R,
Silkworth W, Soriano SG and Shimaoka M: The volatile anesthetic
isoflurane perturbs conformational activation of integrin LFA-1 by
binding to the allosteric regulatory cavity. FASEB J. 22:4109–4116.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Yuki K, Bu W, Xi J, Sen M, Shimaoka M and
Eckenhoff RG: Isoflurane binds and stabilizes a closed conformation
of the leukocyte function-associated antigen-1. FASEB J.
26:4408–4417. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Jung S and Yuki K: Differential effects of
volatile anesthetics on leukocyte integrin macrophage-1 antigen. J
Immunotoxicol. 13:148–156. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Shimaoka M and Springer TA: Therapeutic
antagonists and conformational regulation of integrin function. Nat
Rev Drug Discov. 2:703–716. 2003. View
Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ho MK and Springer TA: Mac-1 antigen:
quantitative expression in macrophage populations and tissues, and
immunofluorescent localization in spleen. J Immunol. 128:2281–2286.
1982. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ding ZM, Babensee JE, Simon SI, Lu H,
Perrard JL, Bullard DC, Dai XY, Bromley SK, Dustin ML, Entman ML,
et al: Relative contribution of LFA-1 and Mac-1 to neutrophil
adhesion and migration. J Immunol. 163:5029–5038. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Coxon A, Rieu P, Barkalow FJ, Askari S,
Sharpe AH, von Andrian UH, Arnaout MA and Mayadas TN: A novel role
for the beta 2 integrin CD11b/CD18 in neutrophil apoptosis: A
homeostatic mechanism in inflammation. Immunity. 5:653–666. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Berrueta L, Bergholz J, Munoz D, Muskaj I,
Badger GJ, Shukla A, Kim HJ, Zhao JJ and Langevin HM: Stretching
reduces tumor growth in a mouse breast cancer model. Sci Rep.
8:78642018. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Tomayko MM and Reynolds CP: Determination
of subcutaneous tumor size in athymic (nude) mice. Cancer Chemother
Pharmacol. 24:148–154. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Sonner JM, Gong D, Li J, Eger EI II and
Laster MJ: Mouse strain modestly influences minimum alveolar
anesthetic concentration and convulsivity of inhaled compounds.
Anesth Analg. 89:1030–1034. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Dahan A, Sarton E, Teppema L, Olievier C,
Nieuwenhuijs D, Matthes HW and Kieffer BL: Anesthetic potency and
influence of morphine and sevoflurane on respiration in mu-opioid
receptor knockout mice. Anesthesiology. 94:824–832. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Okuno T, Koutsogiannaki S, Hou L, Bu W,
Ohto U, Eckenhoff RG, Yokomizo T and Yuki K: Volatile anesthetics
isoflurane and sevoflurane directly target and attenuate Toll-like
receptor 4 system. FASEB J. 33:14528–14541. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zha H, Matsunami E, Blazon-Brown N,
Koutsogiannaki S, Hou L, Bu W, Babazada H, Odegard KC, Liu R,
Eckenhoff RG and Yuki K: Volatile anesthetics affect macrophage
phagocytosis. PLoS One. 14:e02161632019. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Yuki K, Bu W, Shimaoka M and Eckenhoff R:
Volatile anesthetics, not intravenous anesthetic propofol bind to
and attenuate the activation of platelet receptor integrin αIIbβ3.
PLoS One. 8:e604152013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Walker OL, Dahn ML, Power Coombs MR and
Marcato P: The prostaglandin E2 pathway and breast cancer stem
cells: Evidence of increased signaling and potential targeting.
Front Oncol. 11:7916962022. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Akaydin S, Ramazanoğlu S, Salihoğlu EM,
Karanlik H and Demokan S: Leukotriene D4 levels in patients with
breast cancer. FABAD J Pharm Sci. 47:331–338. 2022.
|
|
28
|
Yuki K: The role of general anesthetic
drug selection in cancer outcome. Biomed Res Int. 2021:25630932021.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Koutsogiannaki S, Schaefers MM, Okuno T,
Ohba M, Yokomizo T, Priebe GP, DiNardo JA, Sulpicio SG and Yuki K:
From the cover: Prolonged exposure to volatile anesthetic
isoflurane worsens the outcome of polymicrobial abdominal sepsis.
Toxicol Sci. 156:402–411. 2017.PubMed/NCBI
|
|
30
|
Tazawa K, Koutsogiannaki S, Chamberlain M
and Yuki K: The effect of different anesthetics on tumor
cytotoxicity by natural killer cells. Toxicol Lett. 266:23–31.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Barber DF, Faure M and Long EO: LFA-1
contributes an early signal for NK cell cytotoxicity. J Immunol.
173:3653–3659. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Gao N, Wang C, Yu Y, Xie L, Xing Y, Zhang
Y, Wang Y, Wu J and Cai Y: LFA-1/ICAM-1 promotes NK cell
cytotoxicity associated with the pathogenesis of ocular
toxoplasmosis in murine model. PLoS Negl Trop Dis. 16:e00108482022.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Hickman A, Koetsier J, Kurtanich T,
Nielsen MC, Winn G, Wang Y, Bentebibel SE, Shi L, Punt S, Williams
L, et al: LFA-1 activation enriches tumor-specific T cells in a
cold tumor model and synergizes with CTLA-4 blockade. J Clin
Invest. 132:e1541522022. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Hedrick CC and Malanchi I: Neutrophils in
cancer: Heterogeneous and multifaceted. Nat Rev Immunol.
22:173–187. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
DeNardo DG and Ruffell B: Macrophages as
regulators of tumour immunity and immunotherapy. Nat Rev Immunol.
19:369–382. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Tai LH, Tanese de Souza C, Sahi S, Zhang
J, Alkayyal AA, Ananth AA and Auer RA: A mouse tumor model of
surgical stress to explore the mechanisms of postoperative
immunosuppression and evaluate novel perioperative immunotherapies.
J Vis Exp. 512532014.PubMed/NCBI
|
|
37
|
Guo P, Huang J, Wang L, Jia D, Yang J,
Dillon DA, Zurakowski D, Mao H, Moses MA and Auguste DT: ICAM-1 as
a molecular target for triple negative breast cancer. Proc Natl
Acad Sci USA. 111:14710–14715. 2014. View Article : Google Scholar : PubMed/NCBI
|