|
1
|
Echt G, Jepson J, Steel J, Langholz B,
Luxton G, Hernandez W, Astrahan M and Petrovich Z: Treatment of
uterine sarcomas. Cancer. 66:35–39. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Kim WY, Chang SJ, Chang KH, Yoon JH, Kim
JH, Kim BG, Bae DS and Ryu HS: Uterine leiomyosarcoma: 14-year
two-center experience of 31 cases. Cancer Res Treat. 41:24–28.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Tirumani SH, Deaver P, Shinagare AB,
Tirumani H, Hornick JL, George S and Ramaiya NH: Metastatic pattern
of uterine leiomyosarcoma: Retrospective analysis of the predictors
and outcome in 113 patients. J Gynecol Oncol. 25:306–312. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Chern JY, Boyd LR and Blank SV: Uterine
sarcomas: The latest approaches for these rare but potentially
deadly tumors. Oncology (Williston Park). 31:229–236.
2017.PubMed/NCBI
|
|
5
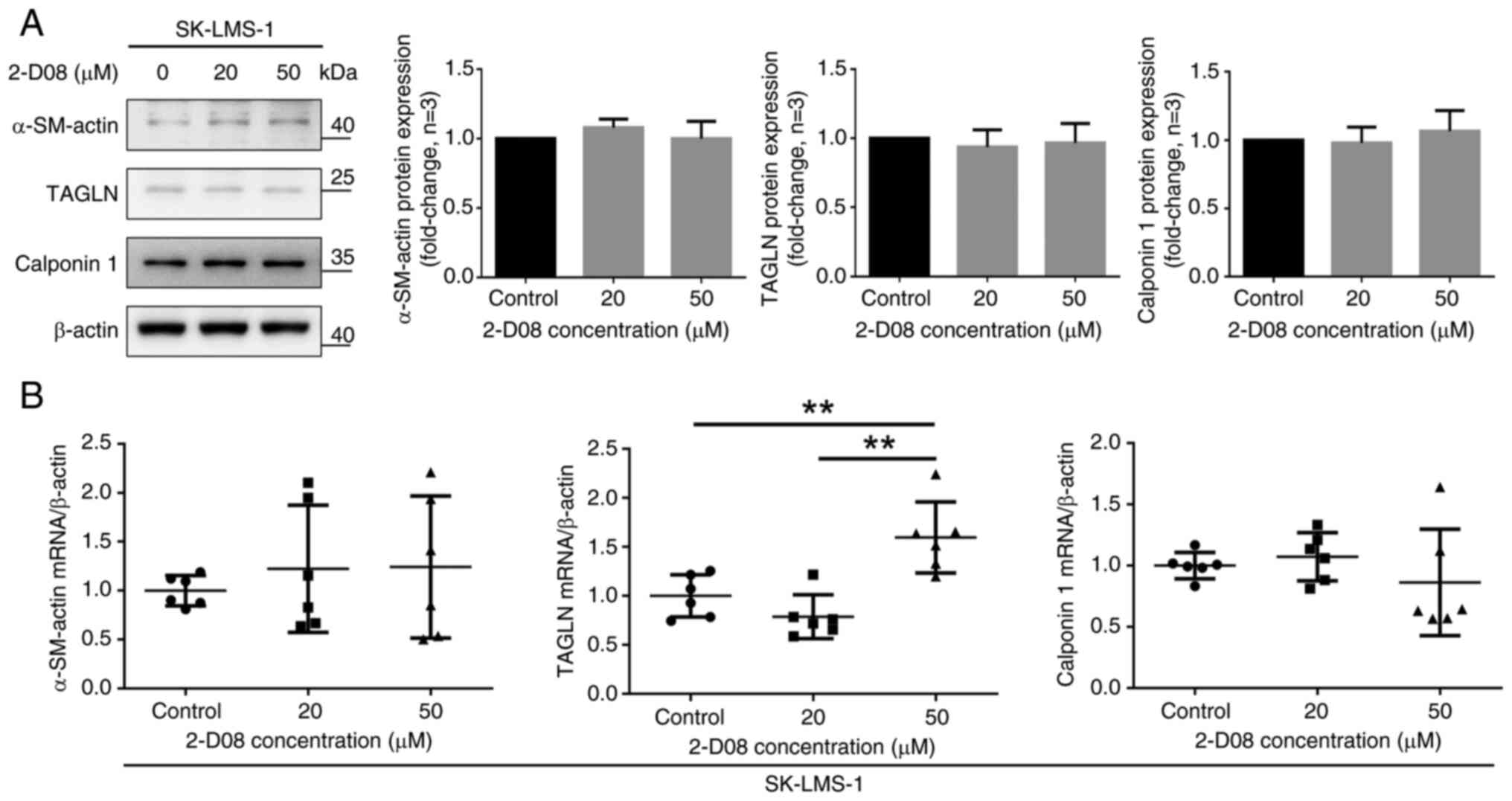
|
Murakami M, Ichimura T, Kasai M, Matsuda
M, Kawamura N, Fukuda T and Sumi T: Examination of the use of
needle biopsy to perform laparoscopic surgery safely on uterine
smooth muscle tumors. Oncol Lett. 15:8647–8651. 2018.PubMed/NCBI
|
|
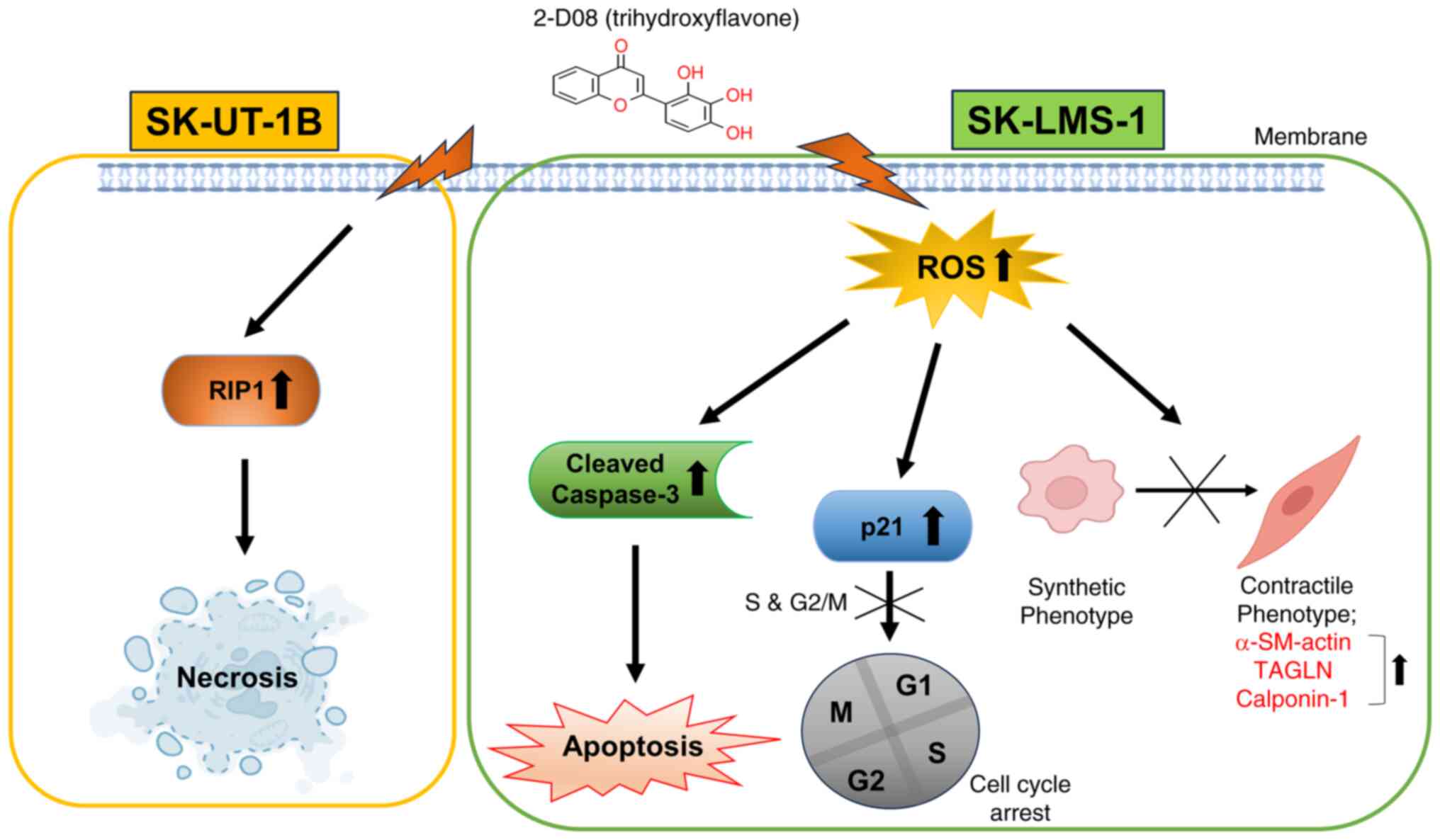
6
|
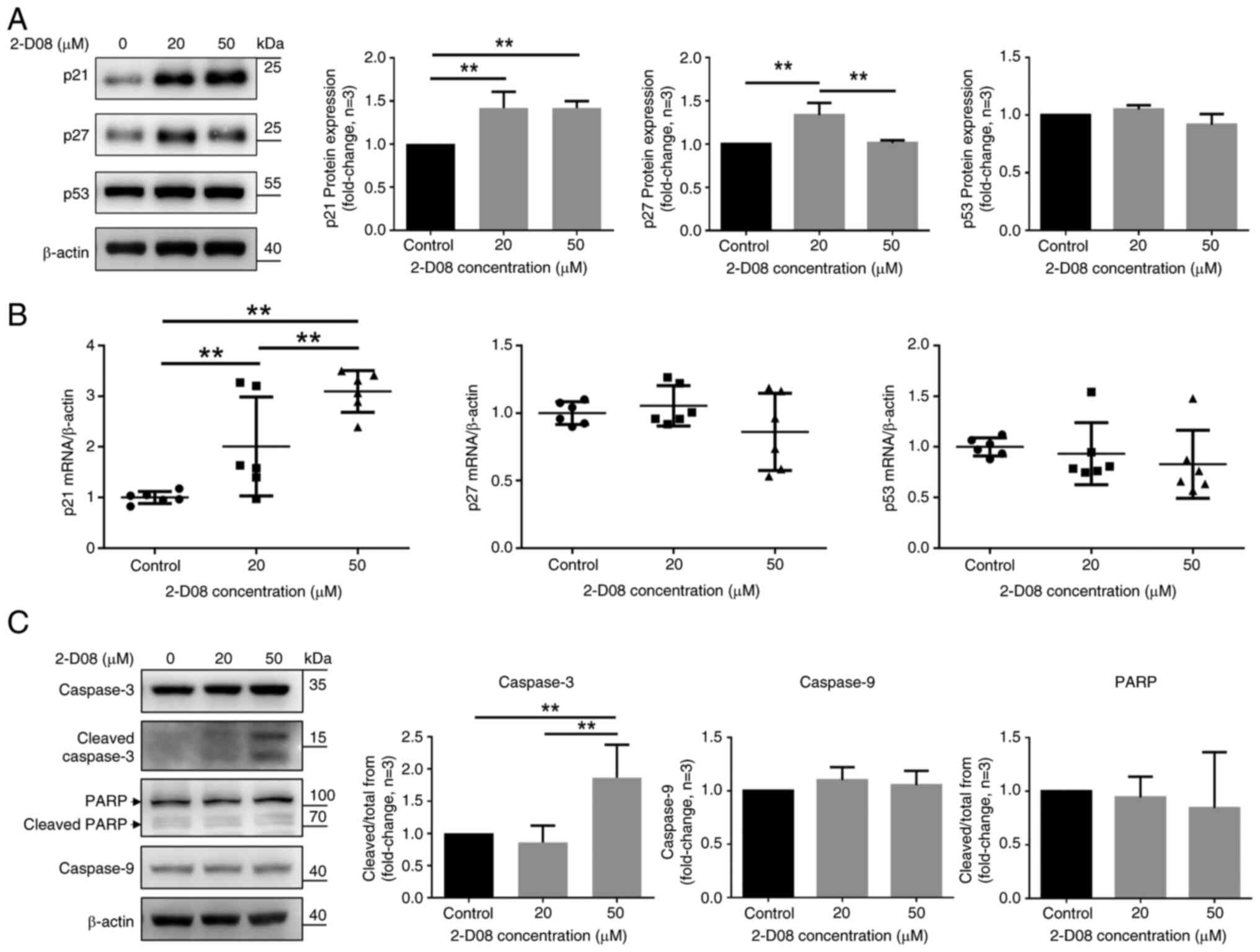
Hernando E, Charytonowicz E, Dudas ME,
Menendez S, Matushansky I, Mills J, Socci ND, Behrendt N, Ma L,
Maki RG, et al: The AKT-mTOR pathway plays a critical role in the
development of leiomyosarcomas. Nat Med. 13:748–753. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Babichev Y, Kabaroff L, Datti A, Uehling
D, Isaac M, Al-Awar R, Prakesch M, Sun RX, Boutros PC, Venier R, et
al: PI3K/AKT/mTOR inhibition in combination with doxorubicin is an
effective therapy for leiomyosarcoma. J Transl Med. 14:672016.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Gaumann AK, Drexler HC, Lang SA,
Stoeltzing O, Diermeier-Daucher S, Buchdunger E, Wood J, Bold G and
Breier G: The inhibition of tyrosine kinase receptor signalling in
leiomyosarcoma cells using the small molecule kinase inhibitor
PTK787/ZK222584 (Vatalanib®). Int J Oncol. 45:2267–2277.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Hayashi T, Horiuchi A, Sano K, Hiraoka N,
Kanai Y, Shiozawa T, Tonegawa S and Konishi I: Molecular approach
to uterine leiomyosarcoma: LMP2-deficient mice as an animal model
of spontaneous uterine leiomyosarcoma. Sarcoma. 2011:4764982011.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Cancer Genome Atlas Research Network.
Electronic address, . simpleelizabeth.demicco@sinaihealthsystem.ca
and Cancer Genome Atlas Research Network: Comprehensive and
integrated genomic characterization of adult soft tissue sarcomas.
Cell. 171:950–965.e28. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Mäkinen N, Aavikko M, Heikkinen T, Taipale
M, Taipale J, Koivisto-Korander R, Bützow R and Vahteristo P: Exome
sequencing of uterine leiomyosarcomas identifies frequent mutations
in TP53, ATRX, and MED12. PLoS Genet. 12:e10058502016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Tsuyoshi H and Yoshida Y: Molecular
biomarkers for uterine leiomyosarcoma and endometrial stromal
sarcoma. Cancer Sci. 109:1743–1752. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hanahan D and Weinberg RA: Hallmarks of
cancer: The next generation. Cell. 144:646–674. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
DeCensi A, Guerrieri-Gonzaga A, Gandini S,
Serrano D, Cazzaniga M, Mora S, Johansson H, Lien EA, Pruneri G,
Viale G and Bonanni B: Prognostic significance of Ki-67 labeling
index after short-term presurgical tamoxifen in women with
ER-positive breast cancer. Ann Oncol. 22:582–587. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Davey MG, Hynes SO, Kerin MJ, Miller N and
Lowery AJ: Ki-67 as a prognostic biomarker in invasive breast
cancer. Cancers (Basel). 13:44552021. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Tollefson MK, Karnes RJ, Kwon ED, Lohse
CM, Rangel LJ, Mynderse LA, Cheville JC and Sebo TJ: Prostate
cancer Ki-67 (MIB-1) expression, perineural invasion, and gleason
score as biopsy-based predictors of prostate cancer mortality: The
mayo model. Mayo Clin Proc. 89:308–318. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Maia R, Santos GAD, Reis S, Viana NI,
Pimenta R, Guimarães VR, Recuero S, Romão P, Leite KRM, Srougi M
and Passerotti CC: Can we use Ki67 expression to predict prostate
cancer aggressiveness? Rev Col Bras Cir. 49:e20223200. 2022.(In
English, Portuguese). View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zhang F, Zhang F, Liu Z, Wu K, Zhu Y and
Lu Y: Prognostic role of Ki-67 in adrenocortical carcinoma after
primary resection: A retrospective mono-institutional study. Adv
Ther. 36:2756–2768. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Akhan SE, Yavuz E, Tecer A, Iyibozkurt CA,
Topuz S, Tuzlali S, Bengisu E and Berkman S: The expression of
Ki-67, p53, estrogen and progesterone receptors affecting survival
in uterine leiomyosarcomas. A clinicopathologic study. Gynecol
Oncol. 99:36–42. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Travaglino A, Raffone A, Catena U, De Luca
M, Toscano P, Del Prete E, Vecchione ML, Lionetti R, Zullo F and
Insabato L: Ki67 as a prognostic marker in uterine leiomyosarcoma:
A quantitative systematic review. Eur J Obstet Gynecol Reprod Biol.
266:119–124. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kim YS, Keyser SG and Schneekloth JS Jr:
Synthesis of 2′,3′,4′-trihydroxyflavone (2-D08), an inhibitor of
protein sumoylation. Bioorg Med Chem Lett. 24:1094–1097. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Marsh DT, Das S, Ridell J and Smid SD:
Structure-activity relationships for flavone interactions with
amyloid beta reveal a novel anti-aggregatory and neuroprotective
effect of 2′,3′,4′-trihydroxyflavone (2-D08). Bioorg Med Chem.
25:3827–3834. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Choi BH, Philips MR, Chen Y, Lu L and Dai
W: K-Ras Lys-42 is crucial for its signaling, cell migration, and
invasion. J Biol Chem. 293:17574–17581. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zhou P, Chen X, Li M, Tan J, Zhang Y, Yuan
W, Zhou J and Wang G: 2-D08 as a SUMOylation inhibitor induced ROS
accumulation mediates apoptosis of acute myeloid leukemia cells
possibly through the deSUMOylation of NOX2. Biochem Biophys Res
Commun. 513:1063–1069. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Liu H, Lee SM and Joung H: 2-D08 treatment
regulates C2C12 myoblast proliferation and differentiation via the
Erk1/2 and proteasome signaling pathways. J Muscle Res Cell Motil.
42:193–202. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Dobin A, Davis CA, Schlesinger F, Drenkow
J, Zaleski C, Jha S, Batut P, Chaisson M and Gingeras TR: STAR:
Ultrafast universal RNA-seq aligner. Bioinformatics. 29:15–21.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Li B and Dewey CN: RSEM: Accurate
transcript quantification from RNA-Seq data with or without a
reference genome. BMC Bioinformatics. 12:3232011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Love MI, Huber W and Anders S: Moderated
estimation of fold change and dispersion for RNA-seq data with
DESeq2. Genome Biol. 15:5502014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Robinson MD, McCarthy DJ and Smyth GK:
edgeR: A bioconductor package for differential expression analysis
of digital gene expression data. Bioinformatics. 26:139–140. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Guo C, Sun L, Chen X and Zhang D:
Oxidative stress, mitochondrial damage and neurodegenerative
diseases. Neural Regen Res. 8:2003–2014. 2013.PubMed/NCBI
|
|
32
|
Nishikawa M, Hashida M and Takakura Y:
Catalase delivery for inhibiting ROS-mediated tissue injury and
tumor metastasis. Adv Drug Deliv Rev. 61:319–326. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Zhitkovich A: N-Acetylcysteine:
Antioxidant, aldehyde scavenger, and more. Chem Res Toxicol.
32:1318–1319. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Beamish JA, He P, Kottke-Marchant K and
Marchant RE: Molecular regulation of contractile smooth muscle cell
phenotype: Implications for vascular tissue engineering. Tissue Eng
Part B Rev. 16:467–491. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Xie C, Ritchie RP, Huang H, Zhang J and
Chen YE: Smooth muscle cell differentiation in vitro: Models and
underlying molecular mechanisms. Arterioscler Thromb Vasc Biol.
31:1485–1494. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Amada S, Nakano H and Tsuneyoshi M:
Leiomyosarcoma versus bizarre and cellular leiomyomas of the
uterus: A comparative study based on the MIB-1 and proliferating
cell nuclear antigen indices, p53 expression, DNA flow cytometry,
and muscle specific actins. Int J Gynecol Pathol. 14:134–142. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Sprogøe-Jakobsen S and Hølund B:
Immunohistochemistry (Ki-67 and p53) as a tool in determining
malignancy in smooth muscle neoplasms (exemplified by a myxoid
leiomyosarcoma of the uterus). APMIS. 104:705–708. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Horiuchi A, Nikaido T, Ito K, Zhai Y, Orii
A, Taniguchi S, Toki T and Fujii S: Reduced expression of calponin
h1 in leiomyosarcoma of the uterus. Lab Invest. 78:839–846.
1998.PubMed/NCBI
|
|
39
|
Owens GK, Kumar MS and Wamhoff BR:
Molecular regulation of vascular smooth muscle cell differentiation
in development and disease. Physiol Rev. 84:767–801. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Kimura Y, Morita T, Hayashi K, Miki T and
Sobue K: Myocardin functions as an effective inducer of growth
arrest and differentiation in human uterine leiomyosarcoma cells.
Cancer Res. 70:501–511. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Wang B, Wang Y, Zhang J, Hu C, Jiang J, Li
Y and Peng Z: ROS-induced lipid peroxidation modulates cell death
outcome: Mechanisms behind apoptosis, autophagy, and ferroptosis.
Arch Toxicol. 97:1439–1451. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Nakamura H and Takada K: Reactive oxygen
species in cancer: Current findings and future directions. Cancer
Sci. 112:3945–3952. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Philipp-Staheli J, Kim KH, Liggitt D,
Gurley KE, Longton G and Kemp CJ: Distinct roles for p53, p27Kip1,
and p21Cip1 during tumor development. Oncogene. 23:905–913. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
O'Neill CJ, McBride HA, Connolly LE and
McCluggage WG: Uterine leiomyosarcomas are characterized by high
p16, p53 and MIB1 expression in comparison with usual leiomyomas,
leiomyoma variants and smooth muscle tumours of uncertain malignant
potential. Histopathology. 50:851–858. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Schaefer IM, Hornick JL, Sholl LM, Quade
BJ, Nucci MR and Parra-Herran C: Abnormal p53 and p16 staining
patterns distinguish uterine leiomyosarcoma from inflammatory
myofibroblastic tumour. Histopathology. 70:1138–1146. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Bunz F, Dutriaux A, Lengauer C, Waldman T,
Zhou S, Brown JP, Sedivy JM, Kinzler KW and Vogelstein B:
Requirement for p53 and p21 to sustain G2 arrest after DNA damage.
Science. 282:1497–1501. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Le Guen L, Marchal S, Faure S and de Santa
Barbara P: Mesenchymal-epithelial interactions during digestive
tract development and epithelial stem cell regeneration. Cell Mol
Life Sci. 72:3883–3896. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Scirocco A, Matarrese P, Carabotti M,
Ascione B, Malorni W and Severi C: Cellular and molecular
mechanisms of phenotypic switch in gastrointestinal smooth muscle.
J Cell Physiol. 231:295–302. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Chakraborty R, Chatterjee P, Dave JM,
Ostriker AC, Greif DM, Rzucidlo EM and Martin KA: Targeting smooth
muscle cell phenotypic switching in vascular disease. JVS Vasc Sci.
2:79–94. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Kim HR, Cho KS, Kim E, Lee OH, Yoon H, Lee
S, Moon S, Park M, Hong K, Na Y, et al: Rapid expression of RASD1
is regulated by estrogen receptor-dependent intracellular signaling
pathway in the mouse uterus. Mol Cell Endocrinol. 446:32–39. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Hong K and Choi Y: Role of estrogen and
RAS signaling in repeated implantation failure. BMB Rep.
51:225–229. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Vaidyanathan G, Cismowski MJ, Wang G,
Vincent TS, Brown KD and Lanier SM: The Ras-related protein
AGS1/RASD1 suppresses cell growth. Oncogene. 23:5858–5863. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Park DJ, Nakamura H, Chumakov AM, Said JW,
Miller CW, Chen DL and Koeffler HP: Transactivational and DNA
binding abilities of endogenous p53 in p53 mutant cell lines.
Oncogene. 9:1899–1906. 1994.PubMed/NCBI
|
|
54
|
Smardová J, Pavlová S, Svitáková M,
Grochová D and Ravcuková B: Analysis of p53 status in human cell
lines using a functional assay in yeast: detection of new non-sense
p53 mutation in codon 124. Oncol Rep. 14:901–907. 2005.PubMed/NCBI
|
|
55
|
Mills J, Matos T, Charytonowicz E, Hricik
T, Castillo-Martin M, Remotti F, Lee FY and Matushansky I:
Characterization and comparison of the properties of sarcoma cell
lines in vitro and in vivo. Hum Cell. 22:85–93. 2009. View Article : Google Scholar : PubMed/NCBI
|