Introduction
Endoscopic submucosal dissection (ESD) is a
minimally invasive treatment used to achieve the curative resection
of early-stage gastric cancer with almost no risk of lymph node
metastasis (1-5).
However, ESD continues to be associated with procedure-related
adverse events. In particular, post-operative bleeding is a major
adverse event associated with ESD. Post-operative bleeding, once it
occurs, can be a severe medical condition, requiring blood
transfusion or resulting in shock. It also prolongs hospital stays
and has a significant negative effect on the quality of life of
patients and healthcare economics. Therefore, it is a contingency
that should be prevented as far as possible.
In Japan, the publication of the Japan
Gastroenterological Endoscopy Society (JGES) guidelines for
gastroenterological endoscopy in patients undergoing antithrombotic
treatment (6) has led to more
opportunities for ESD with continued antithrombotic therapy. The
management of post-operative bleeding is therefore an increasingly
critical clinical issue associated with the use of ESD.
Hemostatic procedures, such as post-ESD coagulation
(PEC) (7), are performed for
post-ESD ulcers to prevent post-operative bleeding, which occurs in
3.1-5.5% of cases (7-9).
Mukai et al (10) and Azumi
et al (11) reported the
usefulness of coagulation plus artery-selective clipping (2C) and
search, coagulation and clipping (SCC), respectively. These
procedures reduced the post-operative bleeding rate compared with
the use of PEC alone (10,11). The present study further modified
this method of coagulation and clipping to reduce the risk of
post-operative bleeding. This approach requires the coagulation of
arteries and residual vessels at the margin of the ESD ulcer.
Additional clipping involving the muscular layers was performed to
provide additional strength and prevent delayed perforation due to
over-coagulation. This modified coagulation and clipping (MCC)
method was termed the ‘MCC method’.
The aim of the present retrospective study was to
evaluate the effectiveness of the MCC method in preventing
post-operative bleeding associated with ESD, and in addition, to
compare its usefulness with conventional hemostatic methods.
Patients and methods
Patients
Patients >18 years of age who underwent gastric
ESD for gastric cancer at Fukuchiyama City Hospital (Fukuchiyama,
Japan), Kyoto Kujo Hospital (Kyoto, Japan) and Saiseikai Suita
Hospital (Suita, Japan) between April, 2007 and July, 2020 were
enrolled in the present study. In patients who underwent ESD for
two or more lesions simultaneously, the largest lesion was selected
as the representative lesion. Patients with a history of
esophagectomy or gastrectomy were excluded from the study. The
protocol for the present study was approved by the Clinical Ethics
Committees on Human Experiments of Fukuchiyama City Hospital (IRB
registration no. 4-7), Kujo Hospital (IRB registration no. H291213)
and Saiseikai Suita Hospital (IRB registration no. 2020-12), and it
conforms to the provisions of the Declaration of Helsinki (as
revised in Fortaleza, Brazil, October 2013). Written informed
consent was obtained from all patients prior to their participation
in the study.
Patient characteristics
Patients medical records were reviewed and the
following data were collected: Age, sex, use of anti-thrombotic
agents, tumor location, tumor size, resected specimen size,
histology, invasion depth, the presence of ulcerations, duration of
surgery, number of clips used, time of clipping and ESD-related
complications.
Study design
Between July, 2010 and June, 2020, the MCC method
was introduced and performed in 321 patients undergoing ESD. The
incidence of post-operative bleeding was assessed following gastric
ESD and the clinical conditions affecting post-operative bleeding
during this period were also assessed. In addition, the incidence
of post-operative bleeding following gastric ESD and the clinical
conditions in the 3-year PEC period from April, 2007 to June, 2010
were assessed as a historical control. The bleeding rates were
compared between the PEC period and MCC period.
ESD procedure and management following
ESD
Gastric ESD was performed with an insulated-tip
knife-2 (KD-611L, Olympus Corporation), a Flush knife (DK2620J;
FUJIFILM Wako Pure Chemical Corporation) and a clutch cutter
(DP2618DT; FUJIFILM Wako Pure Chemical Corporation) through a
conventional single-channel endoscope (EG-580RD, FUJIFILM Wako Pure
Chemical Corporation; and Q260J, Olympus Corporation). Marker dots
were made ~5 mm from the lesion. Following the submucosal injection
of hyaluronic acid, a circumferential mucosal incision was made
outside the marked region. Submucosal dissection was performed in
all cases. The hemostasis of active bleeding and the preventive of
coagulation of all visible vessels were performed during or
following ESD using hemostatic forceps (FD-411QR; Coagrasper,
Olympus Corporation) or hot biopsy forceps (HOYA Corporation,PENTAX
Lifecare Division).
A proton-pump inhibitor was intravenously injected
twice daily from the day prior to ESD to the day following ESD. The
patients were supplied with drinking water 2 h following ESD. A
second-look endoscopy was performed the following day. Liquid food
was also provided with no complications. Oral proton-pump
inhibitors were administered from 2 days following ESD to 8 weeks
thereafter.
Participating endoscopists
From April, 2007 to June, 2010, three endoscopists
performed ESD. They had experience with >50 gastric ESDs. From
July, 2010 and June, 2020, eight endoscopists performed the ESDs.
They had experience with >100 gastric ESDs.
Management of anti-thrombotic
agents
For patients receiving oral anti-thrombotic agents,
such as low-dose aspirin, thienopyridine, cilostazol, warfarin,
dabigatran, rivaroxaban, apixaban and edoxaban, the prescribing
doctor was consulted regarding the management of anti-thrombotic
agents prior to ESD. Prior to the publication of the JGES
guidelines for gastroenterological endoscopy in patients undergoing
anti-thrombotic treatment (6),
anti-thrombotic agents were almost always discontinued prior to
ESD. Following the publication of the JGES guidelines, the
management of anti-thrombotic agents was determined according to
the guidelines. The discontinued period of anti-thrombotic agents
prior to the ESD procedure was as follows: Aspirin, thienopyridine,
cilostazol, warfarin and direct oral anticoagulants were withdrawn
at 3-5, 5-7, 2, 3-4 and 1 day prior to ESD, respectively.
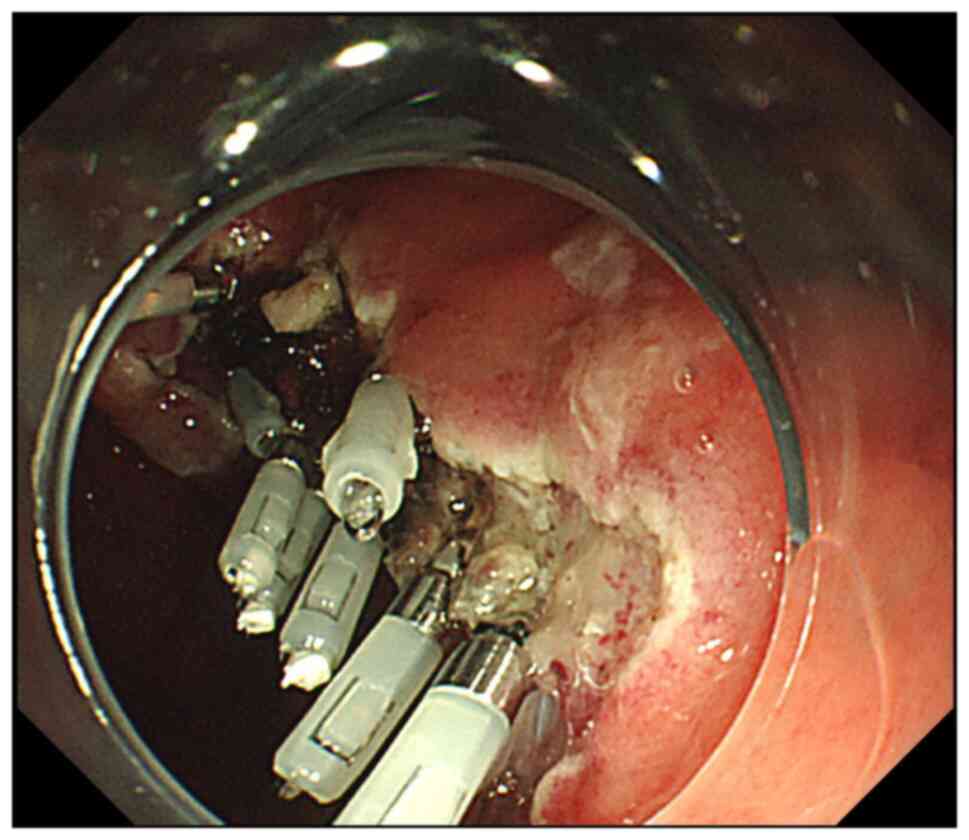
MCC method
For patients undergoing the MCC method, the operator
or assistant recorded the location of the blood vessels that caused
intraoperative bleeding and required hemostasis with hemostatic
forceps or in which preventive coagulation with hemostatic forceps
was performed.
Following the ESD procedure, additional coagulation
was performed at the sites where the location was recorded, at the
penetrating blood vessels in the muscular layer and at the margin
of the ESD ulcer. Additional hemostatic clips (HX-610-135; Olympus
Corporation) were applied to include the muscular layer for the
additional strength of the vessels and to prevent delayed
perforation owing to over-coagulation (Figs. 1 and 2).
Definition of post-operative
bleeding
Post-operative bleeding was defined as an event
requiring urgent endoscopic hemostasis within 1 month following
gastric ESD, such as hematemesis, melena or a decrease in the
hemoglobin concentration by >2 g/dl. Endoscopic hemostasis was
performed when active bleeding or blood coagulation was observed.
Preventive hemostasis of visible vessels without bleeding was not
regarded as post-operative bleeding.
Statistical analysis
For descriptive statistics, continuous variables are
presented as the mean ± standard deviation or median, whereas
discrete variables are presented as the frequency and proportion.
For statistical analyses, the paired Student's t-test or Wilcoxon
signed-rank test were used for discrete variables. The bleeding
rates in the MCC and PEC groups were examined using odds ratios.
When the event occurrence was 0, the continuity correction was
conducted according to the Cornfield formula described in the study
by Schlesselman (12). A two-sided
P-value <0.05 was considered to indicate a statistically
significant difference.
Propensity score matching (PSM) was performed in
order to adjust for confounding variables, where possible to reduce
background differences due to differences in the period of
treatment indication. The scores were calculated as the log-odds
obtained by the logistic regression model, with response variables
including the PEC or MCC groups. Explanatory variables consisted of
tumor location, tumor size, resected specimen size, invasion depth,
histological types, use of anti-thrombotic agents, and procedure
time. Matching was performed using a 1:1 matching protocol with
nearest-neighbor matching within a caliper width of 0.01 without
replacement. The receiver operating characteristic and area under
the curve were used to measure the balance of covariates. The
matched datasets were examined for balance in terms of an absolute
standardized difference. The propensity score was rounded from
three to two decimal places. Matching was performed using three
decimal places for the PEC and MCC groups.
Following PSM, the post-operative bleeding rates
between the PEC and MCC groups were compared.
Statistical analyses were performed using STATA
version 12.1 (StataCorp LP) and EZR (Saitama Medical Center, Jichi
Medical University, Saitama, Japan), R version 2.13.0 (The R
Foundation for Statistical Computing, Vienna, Austria).
Results
A total of 481 (349 males and 132 females) patients
were enrolled in the present study. The clinicopathological
characteristics of these patients are summarized in Table I. The mean age was 71.1±10.0
years.
 | Table IClinical characteristics of all the
patients in the present study and gastric lesions. |
Table I
Clinical characteristics of all the
patients in the present study and gastric lesions.
| Variable | All patients
(n=481) |
|---|
| Sex (male/female), n
(%) | 349/132
(72.6/27.4) |
| Age (mean ± SD)
(years) | 71.1±10.0 |
| Antithrombotic agent,
n (%) | 89 (18.5) |
| Tumor location, n
(%) | |
|
Upper third
of the stomach | 74 (15.4) |
|
Middle third
of the stomach | 133 (27.7) |
|
Lower third
of the stomach | 274 (56.9) |
| Tumor size (mean ±
SD) (mm) | 14.7±10.3 |
| Resected specimen
size (mean ± SD) (mm) | 35.9±12.7 |
| Histological type of
the tumor, n (%) | |
|
Differentiated | 465 (96.7) |
|
Undifferentiated | 16 (3.3) |
| Invasion depth, n
(%) | |
|
T1a | 438 (91.1) |
|
T1b or
greater | 43 (8.9) |
| Presence of
ulceration, n (%) | 37 (7.7) |
| Duration of surgery
time (mean ± SD) (min) | 75.7±72.9 |
Out of the 481 patients, 321 underwent MCC and 160
underwent PEC, respectively. The clinicopathological
characteristics of the groups are presented in Table II. Significant differences between
the two groups were found in tumor size (P=0.034), resected
specimen size (P=0.019), the duration of surgery (P<0.001) and
post-operative bleeding (P<0.001). In total, nine patients
developed postoperative bleeding; however, all of these patients
were in the PEC group, with no patient in the MCC group. All cases
of postoperative bleeding were successfully treated by endoscopic
hemostasis. No major hemorrhaging occurred. In total, 2 cases of
delayed perforation occurred in the PEC group, but not in the MCC
group (Table II). The 2 cases of
delayed perforation were endoscopically closed with clips.
 | Table IIClinicopathological characteristics of
the patients in the PEC and MCC groups. |
Table II
Clinicopathological characteristics of
the patients in the PEC and MCC groups.
| Variable | PEC group
(n=160) | MCC group
(n=321) | P-value |
|---|
| Sex (male/female), n
(%) | 123/37
(76.9/23.1) | 226/95
(70.4/29.6) | 0.159 |
| Age (mean ± SD)
(years) | 70.1±10.2 | 71.3±9.9 | 0.859 |
| Antithrombotic agent,
n (%) | 25 (15.6) | 64 (19.9) | 0.264 |
| Heparin replacement
therapy, n (%) | 8 (5.0) | 9 (2.8) | 0.222 |
| Tumor location, n
(%) | | | 0.404 |
|
Upper third
of the stomach | 29 (18.0) | 45 (14.0) | |
|
Middle third
of the stomach | 46 (28.7) | 87 (27.1) | |
|
Lower third
of the stomach | 85 (53.1) | 189 (58.9) | |
| Tumor size (mean ±
SD) (mm) | 15.7±9.8 | 14.2±10.5 | 0.034 |
| Resected specimen
size (mean ± SD) (mm) | 37.8±13.0 | 34.9±12.4 | 0.019 |
| Histological type of
the tumors, n (%) | | | 0.103 |
|
Differentiated | 158 (98.8) | 307 (95.6) | |
|
Undifferentiated | 2 (1.2) | 14 (4.4) | |
| Invasion depth, n
(%) | | | 0.127 |
|
T1a | 141 (88.1) | 297 (92.5) | |
|
T1b or
greater | 19 (11.9) | 24 (7.5) | |
| Presence of
ulceration, n (%) | 12 (7.5) | 25 (7.8) | 0.999 |
| Operation time
median (range) (min) | 101.6±100.1 | 62.8±49.8 | <0.001 |
| Number of clips
(mean ± SD) | 0 | 7.9±4.4 | |
| Time of clipping
(mean ± SD) | 0 | 13.5±9.2 | |
| Delayed
perforation, n (%) | 2 (1.3) | 0 (0) | 0.110 |
| Post-operative
bleeding, n (%) | 9 (5.6) | 0 (0) | <0.001 |
Out of the 481 patients, 89 (18.5%) were on
anti-thrombotic therapy for their comorbidities. The proportion of
patients in each treatment group on anti-thrombotic therapy was
19.9% (64/321) in the MCC group and 15.6% (25/160) in the PEC
group. Post-operative bleeding occurred in 0% (0/64) of the
patients in the MCC group and in 4% (1/25) of the patients in the
PEC group. No significant difference was observed between the two
groups (P=0.281), although the incidence of post-operative bleeding
was lower in the MCC group than in the PEC group (Table III).
 | Table IIIClinicopathological characteristics
of the patients on anti-thrombotic therapy in the PEC and MCC
groups. |
Table III
Clinicopathological characteristics
of the patients on anti-thrombotic therapy in the PEC and MCC
groups.
| Characteristic | PEC group
(n=25) | MCC group
(n=64) | P-value |
|---|
| Post-operative
bleeding, n (%) | 1(4) | 0 (0) | 0.281 |
| Anti-thrombotic
agent, n (%) | | | |
|
Single
antiplatelet agent | 16(64) | 46 (71.8) | 0.197 |
|
Dual
antiplatelet agents | 2(8) | 4 (6.3) | 0.999 |
|
Anticoagulant
agents | 7(28) | 14 (21.9) | 0.999 |
| Heparin replacement
therapy, n (%) | 8(32) | 9 (14.1) | 0.293 |
Propensity score matching was used to create matched
pairs of the MCC method and PEC method, and comparisons were made
between matched pairs. Following PSM, there were 143 matched pairs
of patients between the two groups. The details of the propensity
score-matched patients are presented in Table IV. A significant difference in
post-operative bleeding was identified between the two groups
(P=0.004).
 | Table IVClinicopathological characteristics
of the patients in the PEC and MCC groups following propensity
score matching. |
Table IV
Clinicopathological characteristics
of the patients in the PEC and MCC groups following propensity
score matching.
| | Following
propensity score matching (n=286) |
|---|
| Variable | PEC method
(n=143) | MCC method
(n=143) | ASD | P-value |
|---|
| Sex (male/female),
n (%) | 109/34
(76.2/23.8) | 100/43
(69.9/30.1) | 0.14 | 0.286 |
| Age (mean ± SD),
years | 71.6±9.6 | 71.9±9.5 | 0.03 | 0.686 |
| Anti-thrombotic
agent, n (%) | 23 (16.1) | 26 (18.2) | 0.05 | 0.754 |
| Heparin replacement
therapy, n | 8 | 4 | | 0.380 |
| Tumor location, n
(%) | | | 0.00 | 0.683 |
|
Upper third
of the stomach | 23 (16.1) | 19 (13.3) | | |
|
Middle third
of the stomach | 41 (28.7) | 47 (32.9) | | |
|
Lower third
of the stomach | 79 (55.2) | 77 (53.8) | | |
| Tumor size (mean ±
SD), mm | 15.1±9.6 | 15.6±12.1 | 0.04 | 0.716 |
| Resected specimen
size (mean ± SD), mm | 36.4±12.4 | 36.2±12.8 | 0.02 | 0.870 |
| Histological type
of the tumors, n (%) | | | 0.14 | 0.498 |
|
Differentiated | 141 (98.6) | 143(100) | | |
|
Undifferentiated | 2 (1.4) | 0 (0) | | |
| Invasion depth, n
(%) | | | 0.13 | 0.441 |
|
T1a | 133 (93.0) | 137 (95.8) | | |
|
T1b or
greater | 10 (7.0) | 6 (4.2) | | |
| Presence of
ulceration, n (%) | 11 | 16 | | 0.419 |
| Duration of
surgery, median (range) (min) | 78.1±66.1 | 77.2±64.3 | 0.01 | 0.908 |
| Number of clips
(mean ± SD) | 0 | 8.6±4.4 | | |
| Time of clipping
(mean ± SD) | 0 | 14.3±9.3 | | |
| Delayed
perforation, n (%) | 2 (1.4) | 0 (0) | | 0.500 |
| Postoperative
bleeding, n (%) | 8 (5.6) | 0 (0) | | 0.004 |
Discussion
The present study introduced the MCC method and
confirmed its efficacy in preventing post-operative bleeding
associated with gastric ESD, with good results: Not a single case
of post-operative bleeding was observed in a multicenter setting
over a 10-year period.
Post-operative bleeding associated with gastric ESD
requires invasive intervention and, to date, has been difficult to
eliminate. Mukai et al (10) and Azumi et al (11) reported that the 2C and SCC methods,
respectively, reduced the post-operative bleeding rate compared
with PEC alone. In the present study, no patient in the MCC group
developed post-operative bleeding associated with gastric ESD.
There were no complications associated with the MCC method. On the
other hand, 9 patients developed post-operative bleeding in the PEC
group. In many of these cases, sufficient coagulation to the deeper
layer of the muscle may not be achieved. This may cause
post-operative bleeding.
The main difference between the MCC method and the
other two clipping methods is the application of coagulation and
clipping. The 2C and SCC methods coagulate vessels and clip for
additional strength. By contrast, the MCC method coagulates vessels
and applies clips not only to provide additional strength, but also
to prevent delayed perforation due to over-coagulation. After the
high-risk vessels and residual vessels at the margin of the ESD
ulcer were coagulated, additional clipping involving the muscular
layers surrounding the vessels was performed with sufficient clips.
Accordingly, more clips are used in the MCC method (mean, 7.9
clips) than in either of the other two methods (mean, 3.8-4.8
clips). These factors may aid in preventing post-operative bleeding
associated with gastric ESD. In the PEC group, 2 cases of delayed
perforation occurred, but none in the MCC group. In the PEC group,
vessels were coagulated sufficiently to prevent post-operative
bleeding, although this may lead to over-coagulation, resulting in
delayed perforation. No statistically significant differences
between the PEC group and MCC group were identified due to the
small number of cases of delayed perforation.
In the hyper-aged society of Japan, the number of
patients undergoing ESD and receiving anti-thrombotic therapy is
significantly increasing. In addition, the JGES guidelines for
gastroenterological endoscopy in patients undergoing
anti-thrombotic treatment (6) have
made it commonplace to perform ESD with continued antithrombotic
therapy. On the other hand, anti-thrombotic therapy is a definite
risk factor for post-operative bleeding, particularly in patients
receiving dual antiplatelet agents and heparin replacement therapy
(13-17).
In the PEC implementation period between 2007 and
2010, anti-thrombotic drugs were withdrawn during ESD in the
majority of cases, whereas after the guidelines were published in
2012, in the MCC implementation period between 2010 and 2020, the
majority of patients continued their anti-thrombotic therapy or
underwent heparin replacement intraoperatively according to the
guidelines. Nevertheless, during the MCC introduction period,
post-operative bleeding was successfully prevented in all cases
(100%). These findings suggest that the MCC method is useful in
preventing post-operative bleeding in patients receiving
anti-thrombotic therapy.
From an economic point of view, one hemostatic clip
costs 7.5 USD/825 JPY. Mukai et al (10) and Azumi et al (11) reported that 3.8 and 4.9 clips were
required on average, respectively. In the present study, 7.9 clips
were used on average. Thus, the cost was 59.2 USD/6,517.5 JPY.
Considering that the incidence of post-operative bleeding is ~5%,
it may be considered excessive to perform the MCC method for all
patients undergoing gastric ESD. On the other hand, the cost is
considered to be worth the benefit in cases with a high risk of
bleeding with concomitant anti-thrombotic therapy and for the
prevention of severe conditions and mortality in elderly patients
who are vulnerable due to underlying diseases.
The present study had certain limitations which
should be mentioned. First, the present study was conducted in
three institutions with a limited number of patients. Second, the
present study was a retrospective study, and was not conducted with
a simultaneous control. Therefore, there was selection bias. It is
not easy to estimate the effects of the MCC method alone, as
advancements in devices and principles, and improvements in
antacids may also have an impact on reducing the risk of
post-operative bleeding. Intraoperative bleeding is slightly higher
with an insulated-tip knife-2 and Flush knife than with a clutch
cutter in ESD. Intraoperative bleeding may affect post-operative
bleeding. Third, a longer enrollment period and significant
differences in terms of tumor size, resected specimen size and
duration of surgery between the PEC and MCC groups are further
limitations. Significant differences in the duration of surgery
between the two groups are due an improvement of technical skills
with time. The propensity score We was calculated, matched pairs
were constructed and these effects were equalized as much as
possible. Comparisons on this basis also indicated that the MCC
method was superior to the PEC method in preventing bleeding.
The prevention of bleeding is of utmost importance
in very elderly individuals on anti-thrombotic therapy, whose
general health is fragile and they have underlying diseases, and
are thus at a risk of developing life-threatening subsequent
bleeding. Furthermore, the healthcare costs of dealing with this
phenomena can be exorbitant. The MCC method would be beneficial,
particularly for these high-risk patients undergoing ESD, as no
post-operative bleeding was observed in the present study.
To determine the size of the bleeding prevention
effect of the MCC method alone, it is necessary to assess the
effect size of the MCC method separately from the impact of
advancements in ESD devices and antacid performance on bleeding
prevention. For this purpose, evaluation using randomized control
trials is desirable. Moreover, with the post-bleeding rate of
<5% observed herein, which is not very high, a highly validated
cost-effectiveness analysis that rigorously evaluates cost and
effectiveness needs to be conducted to determine which subjects
should be indicated for the MCC method.
In conclusion, the present study introduced a novel
hemostatic prophylaxis technique termed the MCC method and the
results are noteworthy: No post-operative bleeding was observed in
a multicenter 10-year period. The MCC method was thus suggested to
be a reliable bleeding prevention technique in ESD in the era of
concomitant anti-coagulant therapy and in very elderly patients.
Randomized controlled trials are required however, to fully clarify
the role of the MCC method in the prevention of post-ESD
bleeding.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
KT performed the research, analyzed the data and
wrote the manuscript. KO designed the study. KS analyzed the data.
All the other authors (TaS, SA, TN, TM, NI, SF, HO, TK, KM, TOkuda,
JM, YM, CM, MM, SM, ToS, YI and TOkanoue) contributed to the
collection and interpretation of data for the study, and critically
reviewed the manuscript. All authors have read and approved the
final version of the manuscript and agree to be accountable for all
aspects of the work in ensuring that questions related to the
accuracy or integrity of any part of the work are appropriately
investigated and resolved. KT and KS confirmed the authenticity of
all the raw data.
Ethics approval and consent to
participate
The protocol for the present study was approved by
the Clinical Ethics Committees on Human Experiments of Fukuchiyama
City Hospital (IRB registration no. 4-7), Kujo Hospital (IRB
registration no. H291213) and Saiseikai Suita Hospital (IRB
registration no. 2020-12), and it conforms to the provisions of the
Declaration of Helsinki (as revised in Fortaleza, Brazil, October
2013). Written informed consent and patient consent for publication
were obtained from all patients in the present study.
Patient consent for publication
Written informed consent was obtained from all
patients in the present study for the publication of their data and
any related images.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Abe N, Yamaguchi Y, Takeuchi H, Izumisato
Y, Yanagida O, Masaki T, Mori T, Sugiyama N and Atomi Y: Key
factors for successful en bloc endoscopic submucosal dissection of
early stage gastric cancer using an insulation-tipped diathermic
knife. Hepatogastroenterology. 53:639–642. 2006.PubMed/NCBI
|
|
2
|
Gotoda T: Endoscopic resection of early
gastric cancer. Gastric Cancer. 10:1–11. 2007.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Kim BJ, Chang TH, Kim JJ, Min BH, Lee JH,
Son HJ, Rhee PL, Rhee JC, Kim KM and Park CK: Efficacy and safety
of endoscopic submucosal dissection for early gastric cancer in
patients with comorbid diseases. Gut Liver. 4:186–191.
2010.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Oda I, Saito D, Tada M, Iishi H, Tanabe S,
Oyama T, Doi T, Otani Y, Fujisaki J, Ajioka Y, et al: A multicenter
retrospective study of endoscopic resection for early gastric
cancer. Gastric Cancer. 9:262–270. 2006.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Ono H, Kondo H, Gotoda T, Shirao K,
Yamaguchi H, Saito D, Hosokawa K, Shimoda T and Yoshida S:
Endoscopic mucosal resection for treatment of early gastric cancer.
Gut. 48:225–229. 2001.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Fujimoto K, Fujishiro M, Kato M, Higuchi
K, Iwakiri R, Sakamoto C, Uchiyama S, Kashiwagi A, Ogawa H,
Murakami K, et al: Guidelines for gastroenterological endoscopy in
patients undergoing antithrombotic treatment. Dig Endosc. 26:1–14.
2014.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Takizawa K, Oda I, Gotoda T, Yokoi C,
Matsuda T, Saito Y, Saito D and Ono H: Routine coagulation of
visible vessels may prevent delayed bleeding after endoscopic
submucosal dissection-An analysis of risk factors. Endoscopy.
40:179–183. 2008.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Oda I, Suzuki H, Nonaka S and Yoshinaga S:
Complications of gastric endoscopic submucosal dissection. Dig
Endosc. 25 (Suppl 1):S71–S78. 2013.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Okada K, Yamamoto Y, Kasuga A, Omae M,
Kubota M, Hirasawa T, Ishiyama A, Chino A, Tsuchida T, Fujisaki J,
et al: Risk factors for delayed bleeding after endoscopic
submucosal dissection for gastric neoplasm. Surg Endosc. 25:98–107.
2011.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Mukai S, Cho S, Nakamura S, Hatano Y,
Kotachi T, Shimizu A, Matsuura G, Azakami T, Takaba A, Hamada T, et
al: Postprocedural combined treatment using the coagulation plus
artery-selective clipping (2C) method for the prevention of delayed
bleeding after ESD. Surg Endosc. 27:1292–1301. 2013.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Azumi M, Takeuchi M, Koseki Y, Kumagai M,
Kobayashi Y, Takatsuna M, Yoshioka A, Yoshikawa S, Miura T and
Terai S: The search, coagulation, and clipping (SCC) method
prevents delayed bleeding after gastric endoscopic submucosal
dissection. Gastric Cancer. 22:567–575. 2019.PubMed/NCBI View Article : Google Scholar
|
|
12
|
James J: Schlesselman: Case control
studies: Design, conduct, analysis (Monographs in Epidemiology and
Biostatistics), January 21, 1982.
|
|
13
|
Terasaki K, Dohi O, Naito Y, Azuma Y,
Ishida T, Kitae H, Matsumura S, Ogita K, Takayama S, Mizuno N, et
al: Effects of guidelines for gastroenterological endoscopy in
patients undergoing antithrombotic treatment on postoperative
bleeding after endoscopic submucosal dissection for early gastric
cancer: A propensity score-matching analysis. Digestion.
102:256–264. 2021.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Shindo Y, Matsumoto S, Miyatani H, Yoshida
Y and Mashima H: Risk factors for postoperative bleeding after
gastric endoscopic submucosal dissection in patients under
antithrombotics. World J Gastrointest Endosc. 8:349–356.
2016.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Sanomura Y, Oka S, Tanaka S, Yorita N,
Kuroki K, Kurihara M, Mizumoto T, Yoshifuku Y and Chayama K: Taking
warfarin with heparin replacement and direct oral anticoagulant is
a risk factor for bleeding after endoscopic submucosal dissection
for early gastric cancer. Digestion. 97:240–249. 2018.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Igarashi K, Takizawa K, Kakushima N,
Tanaka M, Kawata N, Yoshida M, Ito S, Imai K, Hotta K, Ishiwatari
H, et al: Should antithrombotic therapy be stopped in patients
undergoing gastric endoscopic submucosal dissection? Surg Endosc.
31:1746–1753. 2017.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Furuhata T, Kaise M, Hoteya S, Iizuka T,
Yamada A, Nomura K, Kuribayashi Y, Kikuchi D, Matsui A, Ogawa O, et
al: Postoperative bleeding after gastric endoscopic submucosal
dissection in patients receiving antithrombotic therapy. Gastric
Cancer. 20:207–214. 2017.PubMed/NCBI View Article : Google Scholar
|