Introduction
Mycosis fungoides (MF) is a low-grade
lymphoproliferative disorder, and the most common MF type is
cutaneous T-cell lymphoma, accounting for 50% all cutaneous T-cell
lymphoma (1). Syringotropic MF (SMF)
is a rare variant of MF (2), which
is defined as MF with infiltration of eccrine glands by neoplastic
cells, according to the World Health Organization classification
(3). Typical disease characteristics
are patches, plaques, nodular masses, papules, scales and
lichenification (2). The shapes of
the skin lesions are often round, circular or irregular, the color
is often red, purple red or dark brown, and the lesions are
typically accompanied by pruritus or baldness (2). Early-stage SMF is treated with
narrowband (311 nm) ultraviolet therapy (2). To date, only a limited number of SMF
cases have been reported in the literature (4,5).
According to the current guidelines, the
syringotropic and folliculotropic forms of the disease are
classified as adnexotropic MF (4,6).
Solitary, erythematous, punctate and anhidrotic patches are often
clinically observed in the syringotropic form (4,6).
Currently, the main recommended treatments for SMF are
extracorporeal phototherapy and total skin electron beam therapy
(2,5,7). The
present study reported a case of rapidly progressive SMF with
alopecia and lymphadenopathy. A good response was observed
following one cycle of chemotherapy with vincristine sulfate,
etoposide, L-asparaginase and prednisone acetate, known as the VELP
chemotherapy regimen.
Case report
In May 2014, a 19-year-old man presented at Lanzhou
General Hospital (Lanzhou, China), complaining of systemic
infiltrative dark red skin plaques with itching that persisted for
5 months. At 5 months prior to the presentation at Lanzhou General
Hospital, the patient had developed red plaques with itching on the
trunk. The number of skin lesions increased gradually and they were
dispersed across the entire body, being particularly prominent on
the head and trunk. Physical examination revealed stable vital
signs and enlarged bilateral submandibular, neck and armpit lymph
nodes. The lymph nodes were soybean-sized, and had a tenacious
texture, moderate mobility and no evident tenderness.
Cardiac, pulmonary and abdominal examinations
revealed no marked abnormalities. A skin examination revealed skin
lesions scattered over the entire body, which were particularly
prominent on the head, trunk and lower extremities. The lesions
were manifested as dark red infiltrative plaques and tumors, with
diameters of 1–4 cm, clear boundaries and a hard texture. Certain
skin lesions were ulcerated, and there were clear ulcerative
exudates on the trunk. The patient suffered from alopecia, which
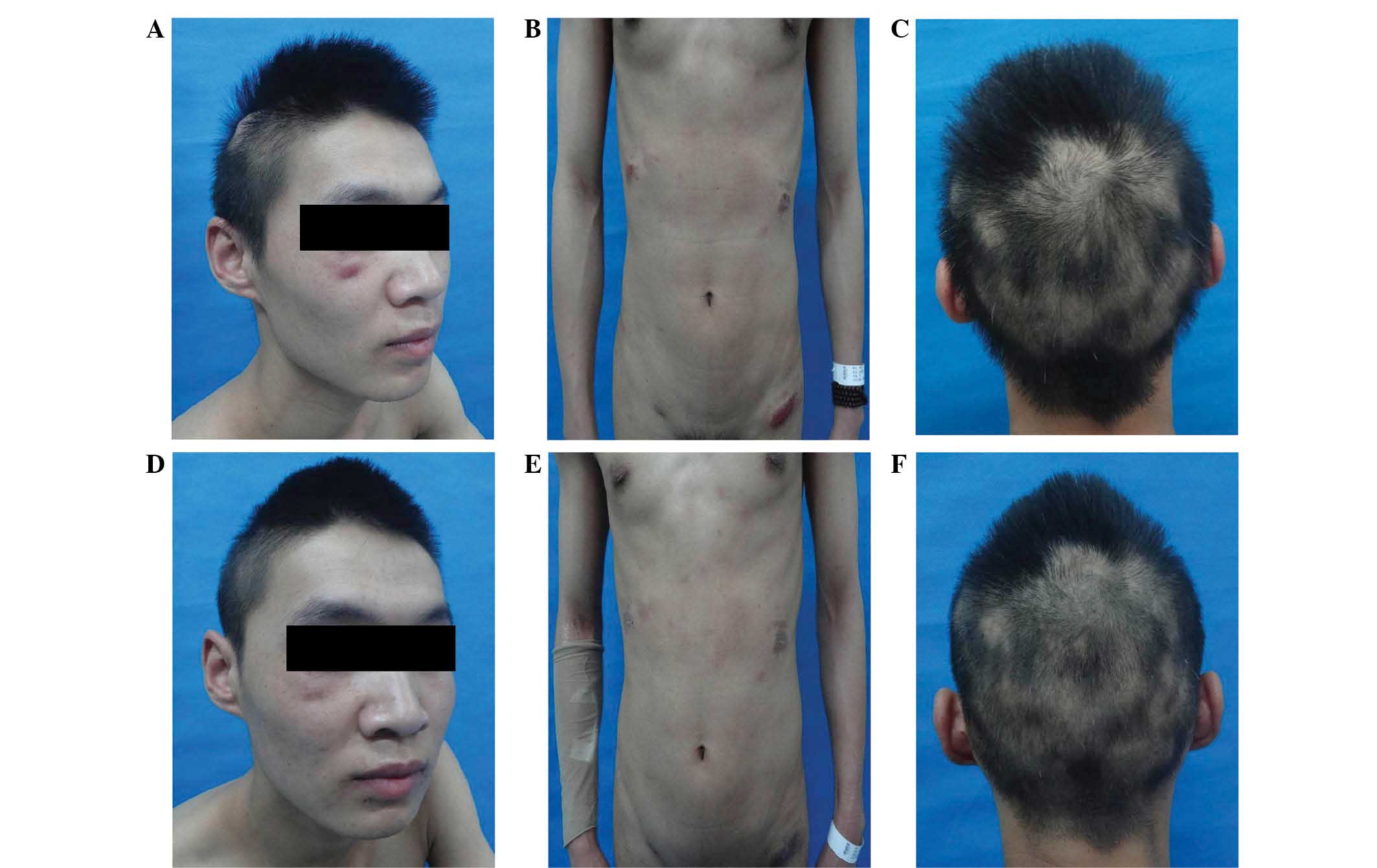
was particularly evident on the occipital scalp (Fig. 1A–C).
The patient underwent skin biopsy twice within a
period of 2 months; the first was performed on admission to
hospital and the second biopsy was performed as a diagnosis could
not be confirmed. Biopsy tissues were fixed in 10% formalin
(Shanghai Baoman Biotechnology Co., Ltd., Shanghai, China) for 12
h, then dehydrated and embedded in paraffin (Shanghai Hualing
Equipment Factory, Shanghai, China) and sectioned (4 µm) using a
Leica section station (RM2016; Leica Microsystems GmbH, Wetzlar,
Germany). The sections were then bleached using a Pathological
Bleaching and Drying Machine (Changzhou Electronic Instrument Co.,
Ltd., Changzhou, China), dewaxed and stained with hematoxylin
(Shanghai Chemical Reagent Co., Ltd., Tianjin, China) and eosin
(HE; Third Factory of Shanghai Reagent Chemicals, Shanghai, China)
for 15 min. The HE-stained sections were observed with an optical
microscope (BX53; Olympus Corporation, Tokyo, China) and diagnosed
by a pathologist. Histopathological examination revealed mild
epidermal spongiosis, unremarkable lymphocyte epidermotropism,
abundant infiltration of lymphocytes into the dermis, no obvious
heterotypic lymphocytes, granulomas formed by epithelioid cells and
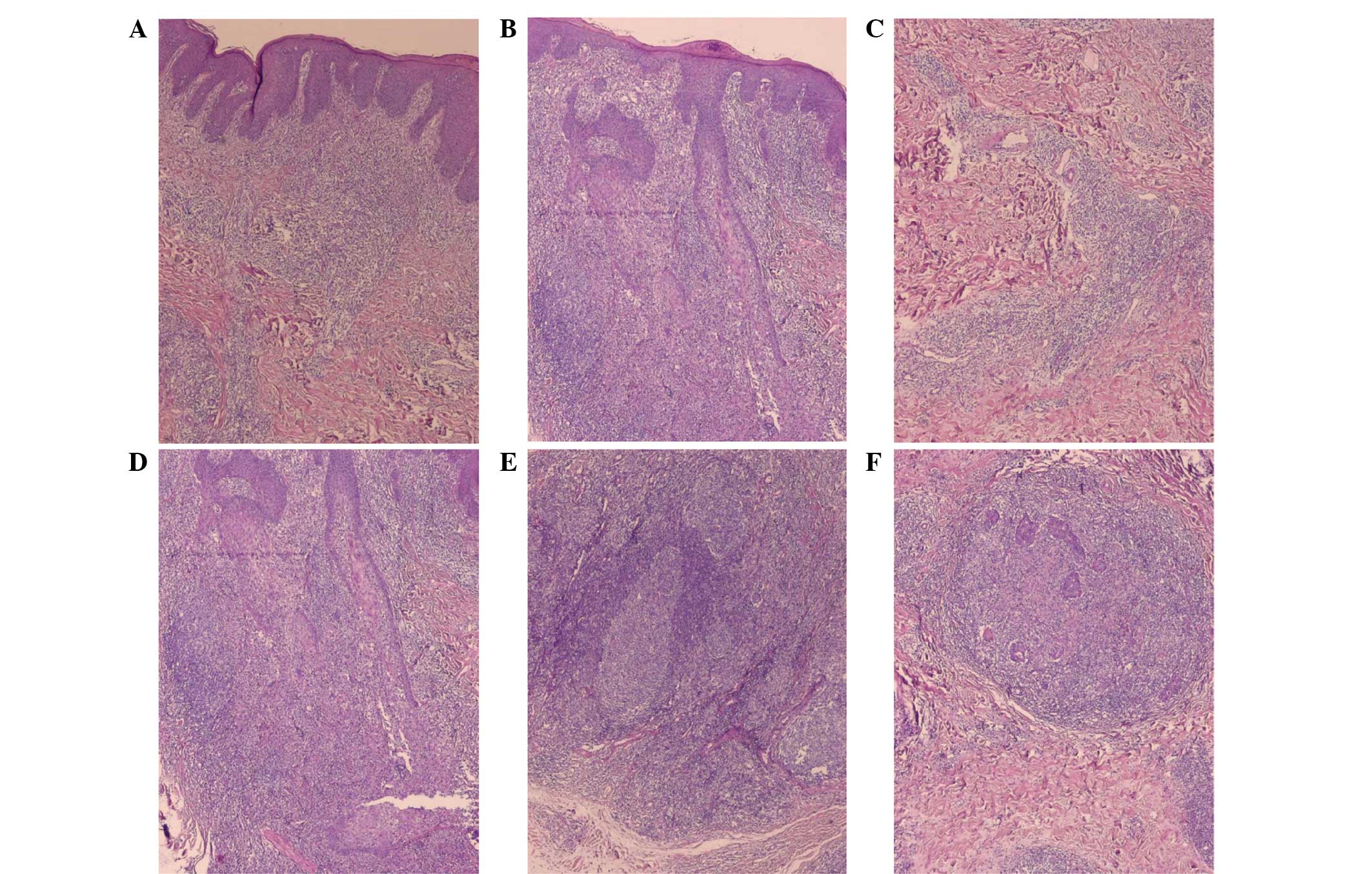
macrophages in the dermis (Fig. 2A and
B), and abundant lymphocyte infiltration around the metaplastic
glands (Fig. 2C and D). There was
abundant lymphocyte infiltration around hair follicles. Nodule-like
structures, abundant macrophages and lymphoid follicle formation
were visible in the lower dermis (Fig.
2E and F).
Biopsy tissues underwent immnohistochemical staining
as follows: Sections (1.5 cmx1.5 cmx4 µm) were cut (RM2235; Leica
Microsystems GmbH), dewaxed in xylene, boiled in 0.01 mol/l citrate
buffer (pH 9.0) (both purchased from Beijing Zhongshan Jinqiao
Biotechnology Co., Ltd., Beijing, China) for 3 min, cooled and
washed with phosphate buffer solution (PBS) 3 times for 3 min,
incubated with 3% H2O2 at 42°C for 10 min,
washed and incubated in PBS for 5 min at 37°C, and incubated with
the following primary antibodies at 37°C for 1 h: Monoclonal mouse
anti-AE1/AE3 (1:100 dilution; IR053), monoclonal mouse anti-Bcl6
(1:50 dilution; IR625), polyclonal rabbit anti-CD3 (1:100 dilution;
IR503), monoclonal mouse anti-CD4 (1:100 dilution; IR649),
monoclonal mouse anti-CD8 (1:150 dilution; IR623), monoclonal mouse
anti-CD20 (1:100 dilution; IR604), monoclonal mouse anti-CD30 (1:50
dilution; IR602), monoclonal mouse anti-CD68 (1:200; IR613) and
monoclonal mouse anti-Ki67 (1:100; IR626) (all purchased from Dako,
Glostrup, Denmark). The sections were washed for 5 min with PBS
twice, incubated with biotinylated goat anti-rabbit/mouse
polyclonal secondary antibody (1:500 dilution; K0675; Dako) at 37°C
for 30 min, and washed for 5 min with PBS twice. The sections were
then incubated with diaminobenzidine solution (Beijing Zhongshan
Jinqiao Biotechnology Co., Ltd.) at 37°C for 5 min, washed with
distilled water for 1 min, stained with hematoxylin for 1 min and
washed with distilled water for 1 min. The sections were incubated
in hydrochloric acid ethanol solution for 10 sec, washed with
distilled water for 1 min and sealed. The sections were then
observed under an Olympus BX53 microscope (Olympus Corporation).
Immunohistochemical tests were positive for CD3 and CD4 and
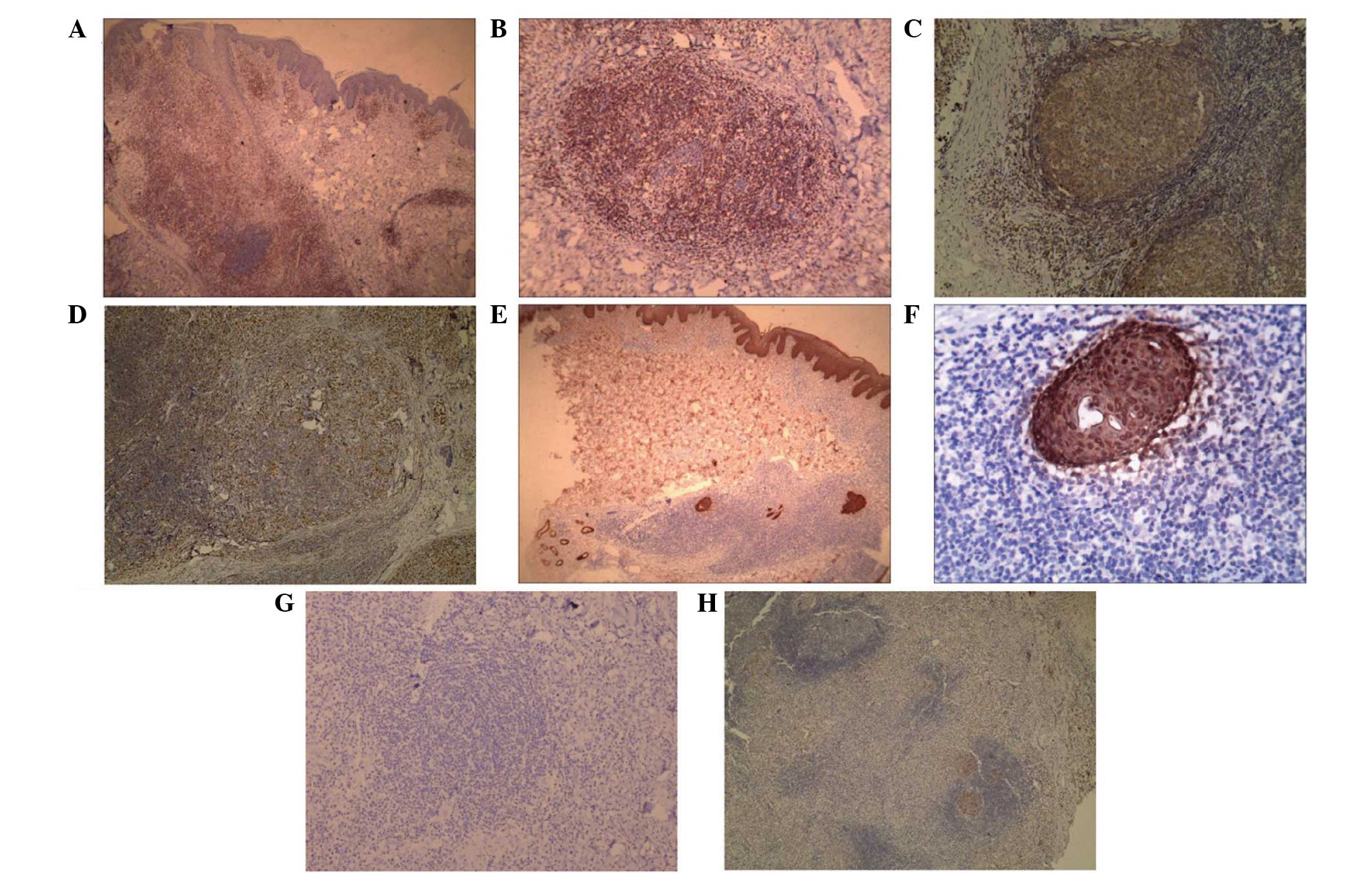
negative for CD8 in the newly formed T lymphocytes (Fig. 3A). In addition, the analysis revealed
an abundance of CD3-positive lymphocytes infiltrating around the
hair follicles. CD3-positive lymphocytes (Fig. 3B), abundance of CD20-positive B cells
(Fig. 3C), and a small quantity of
CD68-positive cells in the nodule-like structures (Fig. 3D) were also observed. The
accumulating infiltrative lymphocytes were negative for CD30
(Fig. 3E), S-100 and CD1a (Fig. 3F). Cytokeratin AE1/AE3 staining
showed an abundance of lymphocytes infiltrating around
brown-staining sweat ducts and arrangement of lymphocytes
coinciding with the coiling direction of the sweat ducts, while an
abundance of infiltrated lymphocytes was observed around the
metaplastic eccrine glands (Fig. 3G and
H).
Acid-fast staining was performed as follows: Samples
were dewaxed and stained with 5% alkaline fuchsin solution (diluted
in 100% ethanol; Tianjin Zhiyuan Chemical Reagent Co., Ltd.,
Tianjin, China) for 5 min, heated to ~100°C and washed with
distilled water. Then, 3% hydrochloric acid and ethanol solution
was added until the sections were no longer faded by the water, and
the samples were double staining with 0.5% methylene blue solution
(Hangzhou Baisi Biotechnology Co., Ltd., Hangzhou, China) for 30–60
sec, washed with distilled water and air-dried. The samples were
then examined under an optical microscope (BX53; Olympus
Corporation). The background of the stained sections was blue, and
no red-stained acid-fast bacillus was observed, therefore excluding
leprosy as a diagnosis.
Based on the aforementioned findings, SMF was
diagnosed, and the clinical stage of the disease was determined as
IIB (8). Subsequently, the patient
received narrowband (311 nm) ultraviolet therapy (SS06 Phototherapy
Unit; Sigma-Aldrich, St. Louis, MO, USA) for 1 month; however, the
skin lesions were not improved and new skin lesions appeared, with
enlargement of the neck, armpit and submandibular lymph nodes also
observed. The patient was then administered VELP chemotherapy
(9). The VELP chemotherapy regimen
was as follows: 2 mg/day intravenous vincristine sulfate (Zhejiang
Hisun Pharmaceutical Co., Ltd., Taizhou, China) for 1 day; 0.1
g/day intravenous etoposide (Jiangsu Hengrui Medicine Co., Ltd.,
Jiangsu, China) for 1 day; 10,000 U/day intravenous L-asparaginase
(Changzhou Qianhong Biochemical Pharmaceutical Co., Ltd.,
Changzhou, China) for 6 days; and 5 mg/day oral prednisone acetate
(Tianjin Lisheng Pharmaceutical Co. Ltd., Tianjin, China), three
times per day for 7 days. After 7 days of VELP chemotherapy, the
skin lesions were clearly ameliorated, the lesion color became
darker, and hair loss was decreased (Fig. 1D–F). No new skin lesions appeared
during the 6 months of follow-up; skin lesions were recovered and
hair was regrown.
Written informed consent was obtained from the
patient for the publication of the present case study.
Discussion
MF, a low-malignant T lymphoma originating from the
skin, is the most common type of skin T lymphoma, accounting for
50% of all the skin lymphomas (3).
Beside from typical MF, there are numerous clinical and
pathological variants, including folliculotropic MF (10), SMF (2,6,7,11) and
granulomatous MF (12). The present
case is a rare SMF with reactive B cell proliferation and lymphoid
follicle formation (5).
SMF was first described by Sarkany (13) in 1969, after which, reports of SMF
have increased gradually. For instance, in 2011, Pileri et
al (14) reported 14 cases of
SMF. Clinically, the major symptoms of SMF are skin patches,
plaques, nodule-like masses, papules, scales and lichenification.
The skin lesions may be round, circular or irregular in shape, and
are frequently red, dark red or dark brown in color. The lesions
are frequently accompanied by pruritus and occasionally by alopecia
(2,4). Santucci et al (15) proposed that the presence of
medium-large cerebriform cells in the epidermis or in clusters in
the dermis is a highly reliable feature for early diagnosis of MF.
SMF is histologically characterized by infiltration of the eccrine
glands by atypical lymphocytes in association with syringolymphoid
hyperplasia.
The current patient showed mild epidermal
spongiosis, unremarkable lymphocyte epidermotropism, massive
lymphocyte infiltration in the dermis, no heterotypical
lymphocytes, granulomas formed by epithelioid cells and macrophages
in the dermis, and evident lymphocyte infiltration around the
metaplastic glands. In addition, abundant lymphocyte infiltration
was observed around the hair follicles. Immunohistochemical
examination showed CD3/CD4 positivity and CD8 negativity in newly
formed T lymphocytes, and CD3-positive lymphocyte infiltration
around the hair follicles. Furthermore, CD3-positive lymphocytes,
CD20-positive B cells and CD68-positive cells were also observed in
the nodule-like structures. Accumulating infiltrative lymphocytes
were negative for CD30, S100 and CD1a. Cytokeratin AE1/AE3 staining
revealed abundant lymphocyte infiltration around the brown-staining
sweat ducts and abundant lymphocyte infiltration around the
metaplastic eccrine glands. Based on these findings, a diagnosis of
SMF with B cell proliferation and lymphoid follicle formation was
established (5).
Subsequent to reviewing a total of 23 SMF cases in
the literature using PubMed (http://www.ncbi.nlm.nih.gov/pubmed; keywords,
syringotropic mycosis fungoides; date of search, December 2015),
the disease often occurred in young people, particularly
individuals with an age of approximately 20 years (2). In the current case, the patient was 19
years old. The patient presented pruritus, and leprosy was excluded
based on histopathological examination, immunohistochemical studies
and acid-fast staining. Subsequently, the patient was diagnosed
with SMF, which is reported to be responsive to phototherapy
(16). Therefore, the patient was
initially given 311-nm ultraviolet therapy for 1 month; however,
the skin lesions were not mitigated and new skin lesions appeared,
with neck, armpit and submandibular lymph nodes becoming
larger.
A number of studies have shown that radiotherapy
achieved an appreciable efficacy in the treatment of SMF (2,17).
However, the patient refused radiotherapy, and thus VELP
chemotherapy was administered. After 7 days of chemotherapy, the
skin lesions clearly remitted and hair loss was improved. During 6
months of follow-up, no new skin lesions or lymphadenopathy were
observed, which suggested a good response to VELP chemotherapy. To
the best of our knowledge, no studies have reported the use of VELP
chemotherapy for the treatment of SMF thus far. Therefore, the
present case provides novel findings for the use and efficacy of
VELP chemotherapy in SMF patients with lymphadenopathy. However,
the underlying mechanism for the efficacy of VELP chemotherapy for
SMF remains unknown and requires further investigation.
In conclusions, the present study reported a case of
SMF with reactive B cell proliferation, lymphoid follicle
formation, hair loss and lymphadenopathy. The patient did not
respond to 311-nm ultraviolet treatment; however, a good response
to VELP chemotherapy was observed. VELP chemotherapy significantly
improved the symptoms, and no new symptoms were observed during the
6-month follow-up period.
References
|
1
|
Nashan D, Faulhaber D, Ständer S, Luger TA
and Stadler R: Mycosis fungoides: A dermatological masquerader. Br
J Dermatol. 156:1–10. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
de Masson A, Battistella M,
Vignon-Pennamen MD, Cavelier-Balloy B, Mouly F, Rybojad M, Bouaziz
JD, Petit A, Saussine A, Ronceray S, et al: Syringotropic mycosis
fungoides: Clinical and histologic features, response to treatment
and outcome in 19 patients. J Am Acad Dermatol. 71:926–934. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Burg G, Kempf W, Cozzio A, Feit J,
Willemze R, Jaffe SE, Dummer R, Berti E, Cerroni L, Chimenti S, et
al: WHO/EORTC classification of cutaneous lymphomas 2005:
Histological and molecular aspects. J Cutan Pathol. 32:647–674.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bakar O, Seckin D, Demirkesen C, Baykal C
and Buyukbabani N: Two clinically unusual cases of folliculotropic
mycosis fungoides: One with and the other without syringotropism.
Ann Dermatol. 26:385–391. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Wang L, Wang G and Gao T: Granulomatous
syringotropic mycosis fungoides with two lesions having reactive
B-cell proliferation. J Cutan Pathol. 41:400–406. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Willemze R, Jaffe ES, Burg G, Cerroni L,
Berti E, Swerdlow SH, Ralfkiaer E, Chimenti S, Diaz-Perez JL,
Duncan LM, et al: WHO-EORTC classification for cutaneous lymphomas.
Blood. 105:3768–3785. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Jennings L, Campbell SM, Yaar R,
Mahalingam M, Sahni D, Lerner A and Rünger TM: Generalized
syringotropic mycosis fungoides responsive to extracorporeal
photopheresis. Br J Dermatol. 170:200–202. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Trautinger F, Knobler R, Willemze R, Peris
K, Stadler R, Laroche L, D'Incan M, Ranki A, Pimpinelli N,
Ortiz-Romero P, et al: EORTC consensus recommendations for the
treatment of mycosis fungoides/Sézary syndrome. Eur J Cancer.
42:1014–1030. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Pectasides D, Aravantinos G, Visvikis A,
Bakoyiannis C, Halikia A, Kalofonos C, Kosmidis P, Skarlos D and
Fountzilas G: Platinum-based chemotherapy of primary extragonadal
germ cell tumours: The hellenic cooperative oncology group
experience. Oncology. 57:1–9. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lehman JS, Cook-Norris RH, Weed BR, Weenig
RH, Gibson LE, Weaver AL and Pittelkow MR: Folliculotropic mycosis
fungoides: Single-center study and systematic review. Arch
Dermatol. 146:607–613. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yost JM, Do TT, Kovalszki K, Su L,
Anderson TF and Gudjonsson JE: Two cases of syringotropic cutaneous
T-cell lymphoma and review of the literature. J Am Acad Dermatol.
61:133–138. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kempf W, Ostheeren-Michaelis S, Paulli M,
Lucioni M, Wechsler J, Audring H, Assaf C, Rüdiger T, Willemze R,
Meijer CJ, et al: Granulomatous mycosis fungoides and granulomatous
slack skin: A multicenter study of the Cutaneous lymphoma
histopathology task force group of the European organization for
research and treatment of cancer (EORTC). Arch Dermatol.
144:1609–1617. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Sarkany I: Patchy alopecia, anhidrosis,
eccrine gland wall hypertrophy and vasculitis. Proc R Soc Med.
62:157–159. 1969.PubMed/NCBI
|
|
14
|
Pileri A, Facchetti F, Rütten A, Zumiani
G, Boi S, Fink-Puches R and Cerroni L: Syringotropic mycosis
fungoides: A rare variant of the disease with peculiar
clinicopathologic features. Am J Surg Pathol. 35:100–109. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Santucci M, Biggeri A, Feller AC, Massi D
and Burg G: Efficacy of histologic criteria for diagnosing early
mycosis fungoides: An EORTC cutaneous lymphoma study group
investigation. European organization for research and treatment of
cancer. Am J Surg Pathol. 24:40–50. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Hodak E, Feinmesser M, Segal T,
Yosipovitch G, Lapidoth M, Maron L, Bergman R, Sahar D and David M:
Follicular cutaneous T-cell lymphoma: A clinicopathological study
of nine cases. Br J Dermatol. 141:315–322. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Jacob R, Scala M and Fung MA: A case of
syringotropic cutaneous T-cell lymphoma treated with local
radiotherapy. J Am Acad Dermatol. 60:152–154. 2009. View Article : Google Scholar : PubMed/NCBI
|