Introduction
Solitary pulmonary capillary hemangioma (SPCH) is a
rare type of benign lung tumor that is difficult to diagnose due to
a lack of symptoms, small lesion size and inconspicuous
histological features (1). According
to our search of previous literature, only 12 SPCH cases have been
reported (1–10). However, each of these studies recorded
only 1 or 2 cases, and thus SPCH has not been studied in depth,
which may explain the absence of a description of SPCH in the World
Health Organization classification of lung tumors in 2015 (11).
SPCH is a benign lesion with an unclear etiology. In
the previously reported cases, all patients were asymptomatic and
the lesion was discovered incidentally rather than clinically
(1–10).
Additionally, tumor growth was universally slow and no patients
succumbed from the disease (1–10). Improvements in understanding and
recognition of SPCH by radiologists, surgeons and pathologists may
aid to elucidate its epidemiology.
SPCH exhibits ground glass opacity (GGO) on computed
tomography (CT), and the GGO lesion may be suspected to be an
adenocarcinoma in situ (AIS)/atypical adenomatous
hyperplasia (AAH) and/or to have focal inflammation (1). The prognosis and treatment of these
lesions vary; SPCH is a benign lesion and may be treated by
complete tumor resection, while AIS/AAH may be malignant or exhibit
malignant potential and require chemotherapy and/or radiotherapy in
addition to surgical resection. Meanwhile, inflammation is
typically treated with anti-inflammatory therapy (1,11). Thus, a
detailed pathological examination is key to ensure correct
diagnosis and subsequent treatment.
In the present study, a clinicopathological study of
an SPCH case has been reported alongside a review of the
literature. The purpose of the study was to define the clinical
manifestation, radiological, histological and immunohistochemical
features, and diagnosis of SPCH, as a currently unrecognized
lesion.
Case report
A 40-year-old Chinese woman was referred to the
Liaoning Cancer Hospital and Institute (Shenyang, China) with a
cough in October, 2015 without obvious inducement. The patient had
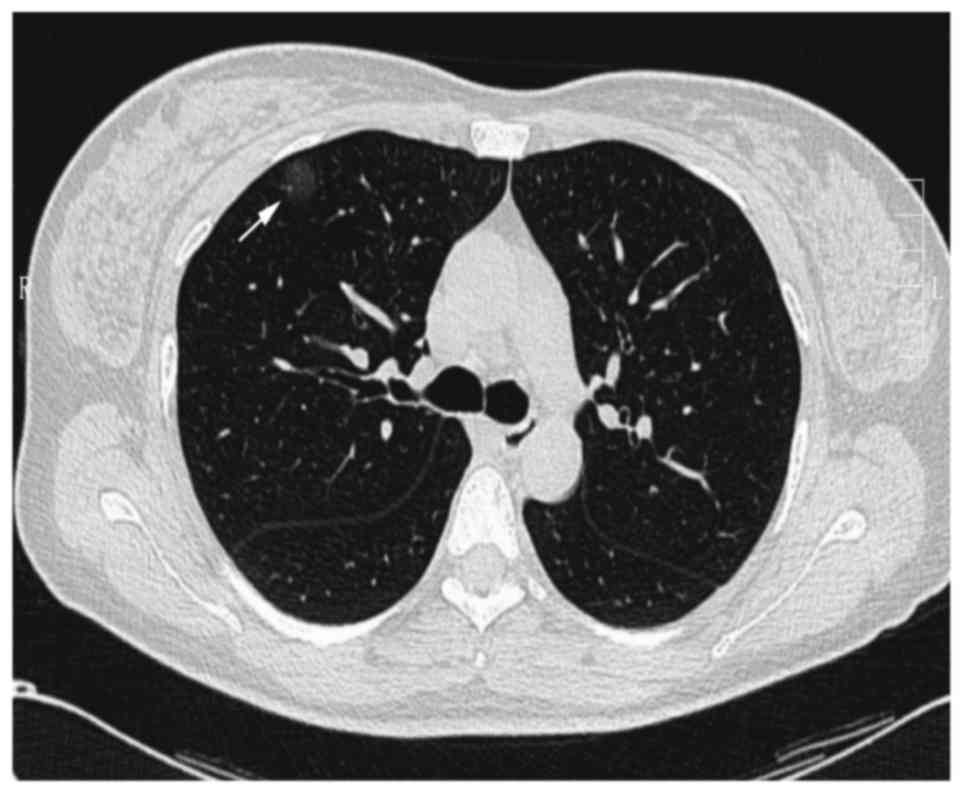
no phlegm or dyspnea chest discomfort. The chest CT indicated a
pure GGO lesion (size, 19×17 mm) in the right upper lung (Fig. 1). Laboratory findings, including tumor
markers, such as carcinoembryonic antigen and carbohydrate antigen
19-9 were within the normal ranges at 1.7 ng/ml (normal range 0–5
ng/ml) and 17.5 U/ml (normal range 0–37 U/ml), respectively
(12). Following systemic
anti-inflammatory therapy for 6 months, the size and density of the
lesion had not changed according to CT. Although no abnormal uptake
of fluorine-18-fluorodeoxyglucose in the lesion was observed during
positron emission tomography-CT, it was suspected to be an AIS or
AAH, and video-assisted thoracic surgery wedge resection was
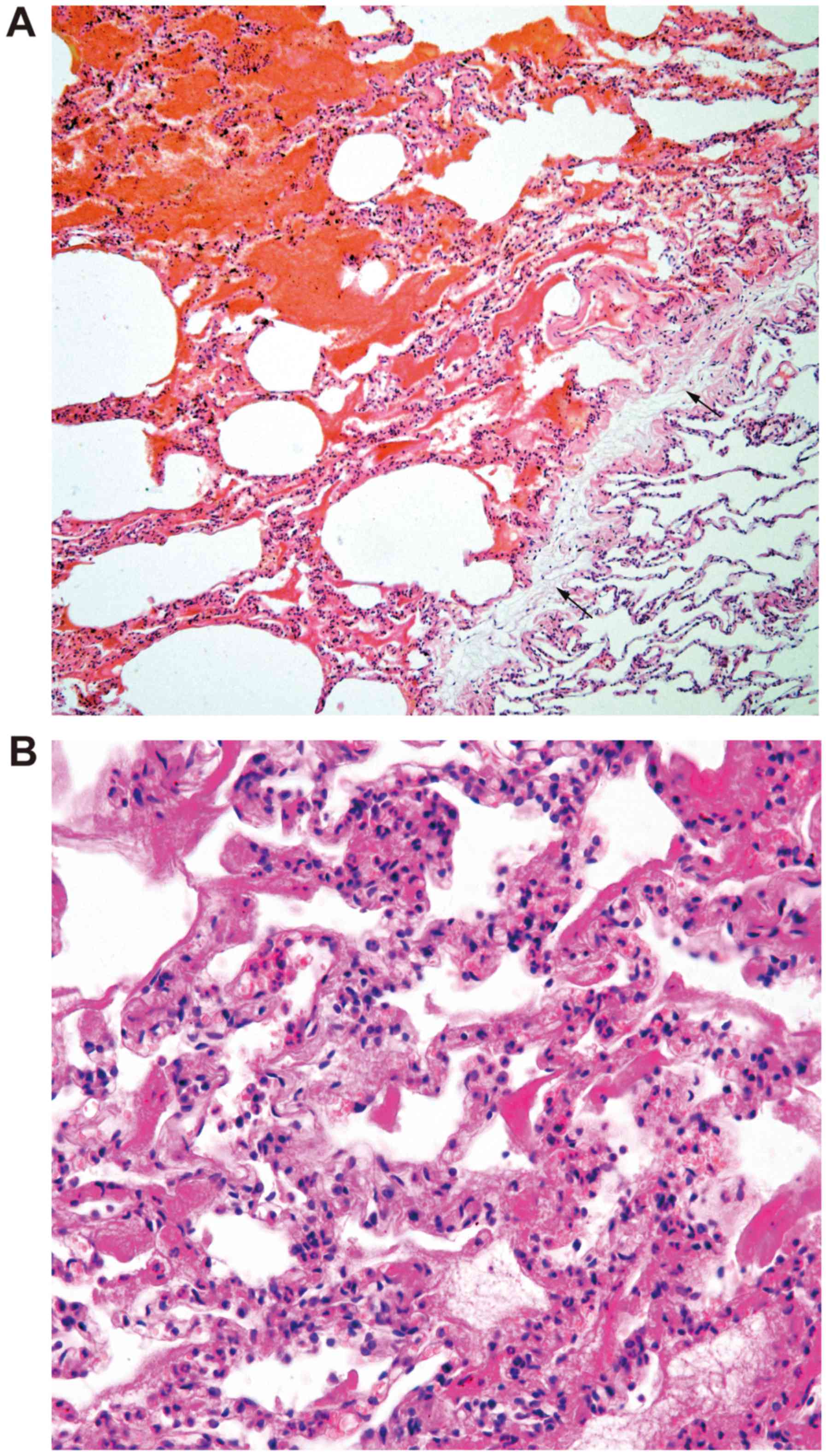
performed. Microscopically, a clear boundary between the tumor and
normal lung tissue was observed in the low-power field (Fig. 2A). The tumor consisted of a narrow
alveolar cavity, the alveolar septa was thickened with proliferated
lumens that were various sizes and the lumens were lined with
single-layer flat cells without atypical epithelial cells (Fig. 2B). Based on the above-mentioned
findings, the intraoperative frozen sections demonstrated that the
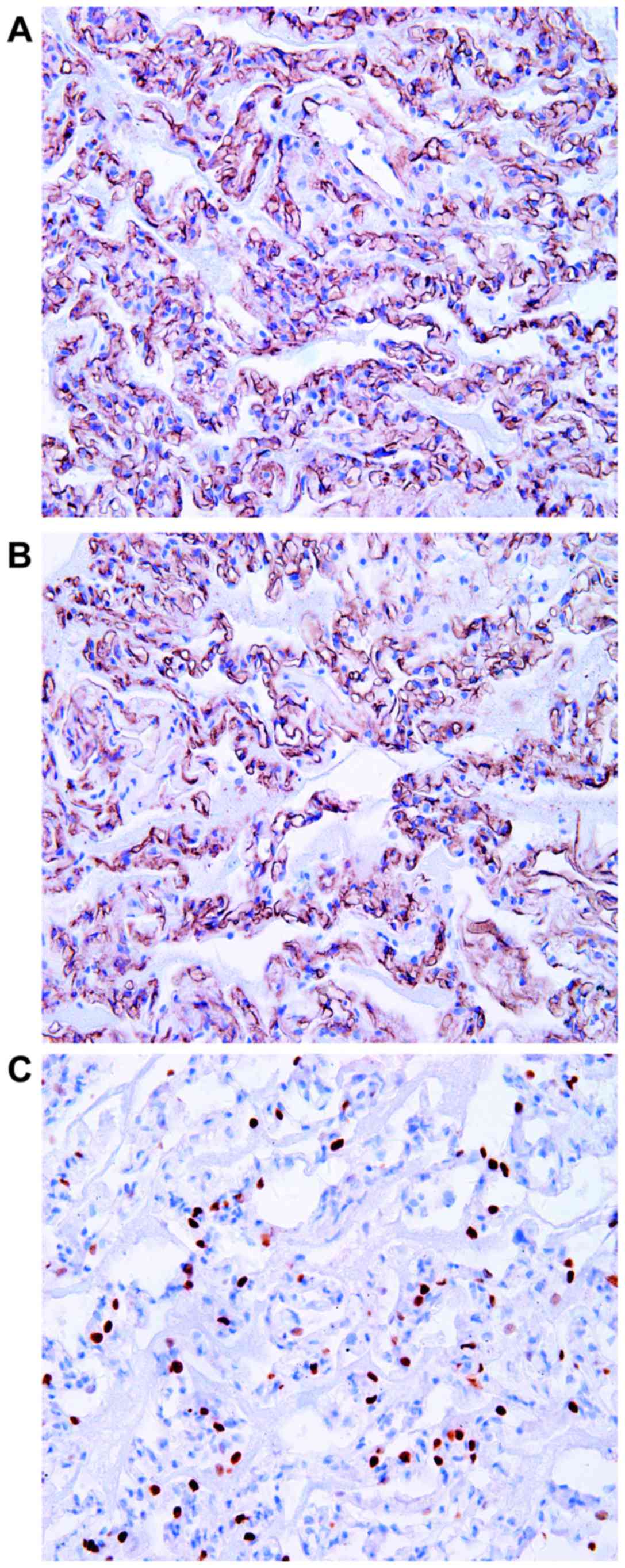
lesion was a benign tumor. Later, immunohistochemical staining of a
paraffin section revealed the flat cells were positive for cluster
of differentiation CD34 (Fig. 3A) and
CD31 (Fig. 3B), and negative for
D2-40, cytokeratin and thyroid transcription factor-1 (Fig. 3C). Therefore, the patient was diagnosed
with SPCH and was followed up for 13 months after surgery with no
recurrence.
Discussion
SPCH is a particularly rare type of benign tumor,
characterized by the proliferation of capillary vessels. It was
initially identified by Fugo et al (1) in 2006. Since then, only 13 cases have
been reported (including the present case; Table I). There were seven males and six
females, and the age ranged from 40 to 61 years (mean age, 52.8
years). The lesions were located on the left side in six cases and
on the right side in seven cases. Two were located in the upper
lobe, two in the middle lobe and nine cases were located in the
lower lobe. The tumor size ranged from 7 to 20 mm (mean, 12 mm).
SPCH tends to present as a single small lesion, which is located in
the lower lobe without any symptoms.
 | Table I.Patient profile of solitary pulmonary
capillary hemangioma. |
Table I.
Patient profile of solitary pulmonary
capillary hemangioma.
| Case | Author, year | Sex | Age, years | Detected | CT finding | Size (mm) | Location | Surgery | Pathological
finding | (Refs) |
|---|
| 1 | Fugo et al
2006 | M | 59 | Medical check up | Mixed GGO | 19×11 | LL peripheral | Seg | The lesion
showed | 1 |
| 2 | Fugo et al
2006 | F | 48 | Medical check up | Mixed GGO | 13×12 | RM peripheral | WR | thickening of
the | 1 |
| 3 | Kato et al
2009 | M | 55 | Medical check up | Mixed GGO | 11 | RL peripheral | Seg | alveolar septa
caused | 2 |
| 4 | Hakiri et al
2010 | M | 45 | Medical check up | Mixed GGO | 12×11 | LL peripheral | Seg | by the
proliferation | 3 |
| 5 | Sakaguchi et
al 2014 | F | 53 | Medical check up | Pure GGO | 20×20 | LU not
peripheral | Lob | of the capillary | 4 |
| 6 | Taniguchi et
al 2010 | F | 59 | Medical check up | Pure GGO | 11×5 | RL peripheral | WR | vessels without | 5 |
| 7 | Yanagawa et al
2007 | M | 58 | Medical check up | Pure GGO | 8 | RL | WR | cytological atypia
or | 6 |
| 8 | Uegami et al
2008 | F | 54 | Medical check up | Solid nodule | 12×11 | RM peripheral | WR | inflammatory | 7 |
| 9 | Shimada et al
2012 | M | 61 | Medical check up | Mixed GGO | 10 | LL peripheral | Lob | background. | 8 |
| 10 | Shimada et al
2012 | M | 42 | Medical check up | Pure GGO | 7 | RL peripheral | WR |
| 8 |
| 11 | Matsushita et
al 2012 | M | 58 | Medical check up | Mixed GGO | 7 | LL | N.D. |
| 9 |
| 12 | Isaka et al
2013 | F | 55 | Medical check up | Mixed GGO | 7×4.6 | LL peripheral | WR |
| 10 |
| 13 | Yanmei et al
2017 | F | 40 | Cough | Pure GGO | 19×17 | RU peripheral | WR |
| Present case |
Observation of GGO is typical upon imaging of cases
of SPCH (1). In 13 cases, four were
pure GGO, and eight were mixed GGO and one case exhibited a solid
nodule. The pure GGO may represent a limited proliferation of
capillary vessels in the alveolar septum, which is followed by a
thickened alveolar septum and a narrowed alveolar space, although
the space was preserved and still contained air. The solid aspect
of the GGO may be the result of the capillary vessel congestion,
which may cause alveolar collapse and a resulting lack of space to
contain air.
When GGO in the peripheral lung is observed, AIS/AAH
may be highly suspected. It is difficult to distinguish AIS/AAH
from SPCH by imaging examination alone. During surgery, Shimada
et al (8) observed that the
SPCH became impalpable following reiterative palpation. This
finding may be caused by squeezing the blood in capillary vessels
followed by constriction of the thickened alveolar septum and the
lesion feeling the same as normal lung tissue. However, in AIS/AAH,
based upon the different histopathological manifestations, the
nodule would not be impalpable, as there are collagenous fibers or
elastic fibers in the alveolar septa rather than capillary vessels.
Therefore, when tumors are located in the peripheral lung, present
with a GGO image and cannot be palpated following reiterative
palpation during surgery, a diagnosis of SPCH may be determined.
SPCH is a benign tumor, whereas AIS/AAH are malignant or have
malignant potential. Therefore, it is critical to differentiate
these lesions in the clinical setting.
Histopathology is an important method, as the
histological appearance between SPCH and AIS/AAH is completely
different (11). AIS often occurs in
the peripheral lung, very close to the pleura. AIS is a small (≤3
cm) adenocarcinoma with growth restricted to neoplastic cells along
pre-existing alveolar structure (lepidic growth), lacking stroma,
vascular, alveolar space or pleural invasion. Grossly, it is a
poorly defined nodule measuring up to 3 cm in size, with a tan or
pale cut surface. Microscopically, AIS typically exhibits type II
pneumocytes and/or Clara cell differentiation. The cells
demonstrate marked cytologic atypia, such as dark and big
nucleolus, coarse granular chromatin and pathologic mitosis.
Alveolar septa widening is common in AIS, which is caused by
sclerosis/elastosis rather than proliferation of capillary vessels
(11). Macroscopically, AAH is a
millimeter-sized, poorly defined, tan-yellowed nodule. It is
characterized by mildly to moderately atypical type II pneumocyte
and/or Clara cell proliferation along the alveolar walls. Double
nuclei are common, but mitoses are extremely rare. There are gaps
along the surface of the basement membrane between the cells.
Alveolar septa widening is not obvious and there is no capillary
vessel proliferation (11).
Pulmonary capillary hemangiomatosis (PCH) is a
multiple-lesion disease with numerous GGO nodules observed on CT
(13). Although the histopathological
characteristics are similar between PCH and SPCH, their clinical
manifestations are completely different (14). PCH is a multiple-lesion disease with
numerous GGO nodules on CT and is associated with pulmonary
hypertension, whereas SPCH is a solitary nodular lesion with only
solitary nodules observed on CT and the patient may present with
mild or no symptoms (13). However,
the underlying mechanisms have not been clarified (1). Evidence indicates that the increased
expression levels of vascular endothelial growth factor and
platelet-derived growth factor may be associated with PCH (1). However, the association between PCH and
SPCH remains unknown. Based upon the similarities in histological
manifestation, whether SPCH is the precursor lesion of PCH or
whether they are independent of each other requires further
investigation.
Pulmonary vein fibrosis is the main histological
change observed in pulmonary veno-occlusive disease (PVOD) that
leads to the complete blocking of pulmonary veins, venous
stagnation and alveolar wall capillary dilatation (14). The histological features of PCH are the
proliferation of capillaries within the alveolar walls, as well as
muscularization of arterioles and medial hypertrophy of muscular
pulmonary arteries. Therefore, PCH causes pulmonary hypertension
(14). PCH and PVOD are similar in
clinical manifestation, though are fundamentally different diseases
due to their histopathological features.
Focal inflammation also exhibits a GGO on CT
(15). Clinically, it may decrease or
disappear following antibiotic therapy. Histopathologically,
inflammation results in different types of inflammatory cell, such
as lymphocytes, plasmocytes, neutrophil granulocytes and
proliferation of fibroblasts, rather than the proliferation of
capillary vessels in widened alveolar septa (11).
In conclusion, different types of lesion exhibit GGO
on CT, and the GGO lesion may be SPCH rather than AIS/AAH. The
prognosis of these types of lesions is entirely different,
therefore, a pathological examination must be conducted to ensure
the correct diagnosis.
Acknowledgements
The current study was supported by the Dr.
Scientific Research Start Fund Project in Liaoning Province, China
(grant no. 20170520046).
Glossary
Abbreviations
Abbreviations:
|
SPCH
|
solitary pulmonary capillary
hemangioma
|
|
CT
|
computed tomography
|
|
GGO
|
ground glass opacity
|
|
AIS
|
adenocarcinoma in situ
|
|
AAH
|
atypical adenomatous hyperplasia
|
|
TTF1
|
thyroid transcription factor-1
|
|
CK
|
cytokeratin
|
References
|
1
|
Fugo K, Matsuno Y, Okamoto K, Kusumoto M,
Maeshima A, Kaji M, Takabatake H, Kondo H and Moriyama N: Solitary
capillary hemangioma of the lung: Report of 2 resected cases
detected by high-resolution CT. Am J Surg Pathol. 30:750–753. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Kato H, Oizumi H, Kanauchi N and Sadahiro
M: A case of pulmonary capillary hemangioma diagnosed by
thoraco-scopic segmentectomy. J Jpn Assoc Chest Surg. 23:932–935.
2009. View Article : Google Scholar
|
|
3
|
Hakiri S, Agatsuma H and Yoshioka H: A
resected case of capillary hemangioma of the lung suspected to be
lung cancer on chest computed tomography. Haigan. 50:841–845. 2010.
View Article : Google Scholar
|
|
4
|
Sakaguchi Y, Isowa N, Tokuyasu H and Miura
H: A resected case of solitary pulmonary capillary hemangioma
showing pure ground glass opacity. Ann Thorac Cardiovasc Surg.
20:578–581. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Taniguchi D, Taniguchi H, Sano I, Tamura
K, Shindou H, Shimizu K, Hamasaki K, Nakazaki T, Shigematsu K and
Takahara O: Solitary capillary hemangioma in the lung: Report of a
case. Kyobu Geka. 63:423–425. 2010.(In Chinese). PubMed/NCBI
|
|
6
|
Yanagawa N, Kato H and Kanauchi N: Two
cases of solitary peripheral small lung tumor needed to
differentiate from small lung adenocarcinoma. Jpn J Diagn Pathol.
24:426–429. 2007.
|
|
7
|
Uegami S, Hirai S, Mitsui N, Matsuura Y
and Hamanaka Y: A case of solitary capillary hemangioma of the
lung. J Jpn Assoc Chest Surg. 22:641–644. 2008. View Article : Google Scholar
|
|
8
|
Shimada Y, Murakawa T, Sano A, Fukami T,
Yoshida Y, Inoue Y, Morita S, Fukayama M and Nakajima J: Capillary
hemangiomas of the lung presenting as ground glass opacities by
high resolution computed tomography. Kyobu Geka. 65:1038–1043.
2012.(In Chinese). PubMed/NCBI
|
|
9
|
Matsushita M, Kawakami S, Matsushita T,
Sugiyama Y, Endo M, Shimojo H, Toishi M and Kadoya M: Changes in CT
density of solitary capillary hemangioma of the lung upon varying
patient position. Jpn J Radiol. 30:772–776. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Isaka T, Yokose T, Ito H, Washimi K,
Imamura N, Watanabe M, Imai K, Nishii T, Yamada K, Nakayama H, et
al: Case of solitary pulmonary capillary hemangioma: Pathological
features based on frozen section analysis. Pathol Int. 63:615–618.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Travis WD, Brambilla E, Nicholson AG,
Yatabe Y, Austin JHM, Beasley MB, Chirieac LR, Dacic S, Duhig E,
Flieder DB, et al: WHO Panel: The 2015 World Health Organization
Classification of Lung Tumors: Impact of Genetic, Clinical and
Radiologic Advances Since the 2004 Classification. J Thorac Oncol.
10:1243–1260. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Chen XL and Chen XY: The significance of
serum tumor markers in lung cancer diagnosis. J Clin Pulm Med.
9:590–592. 2004.
|
|
13
|
Li X, Jin ML, Wei P, Dai HP, Cui A, Zhang
YG, Diao XL and Zhao HY: Pulmonary capillary hemangiomatosis: A
clinicopathologic analysis of 2 cases with review of literature.
Zhonghua Bing Li Xue Za Zhi. 41:16–19. 2012.(In Chinese).
PubMed/NCBI
|
|
14
|
Frazier AA, Franks TJ, Mohammed TL,
Ozbudak IH and Galvin JR: From the archives of the AFIP: Pulmonary
veno-occlusive disease and pulmonary capillary hemangiomatosis.
Radiographics. 27:867–882. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Lv YG, Bao JH, Xu DU, Yan QH, Li YJ, Yuan
DL and Ma JH: Characteristic analysis of pulmonary ground-glass
lesions with the help of 64-slice CT technology. Eur Rev Med
Pharmacol Sci. 21:3212–3217. 2017.PubMed/NCBI
|