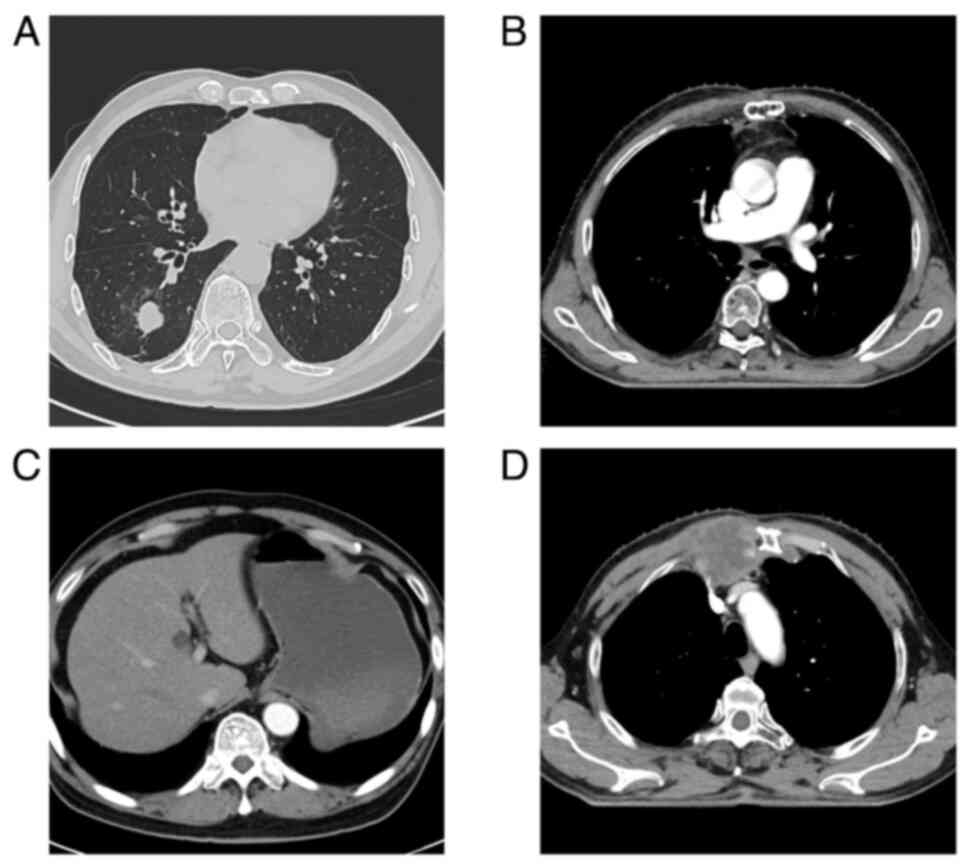
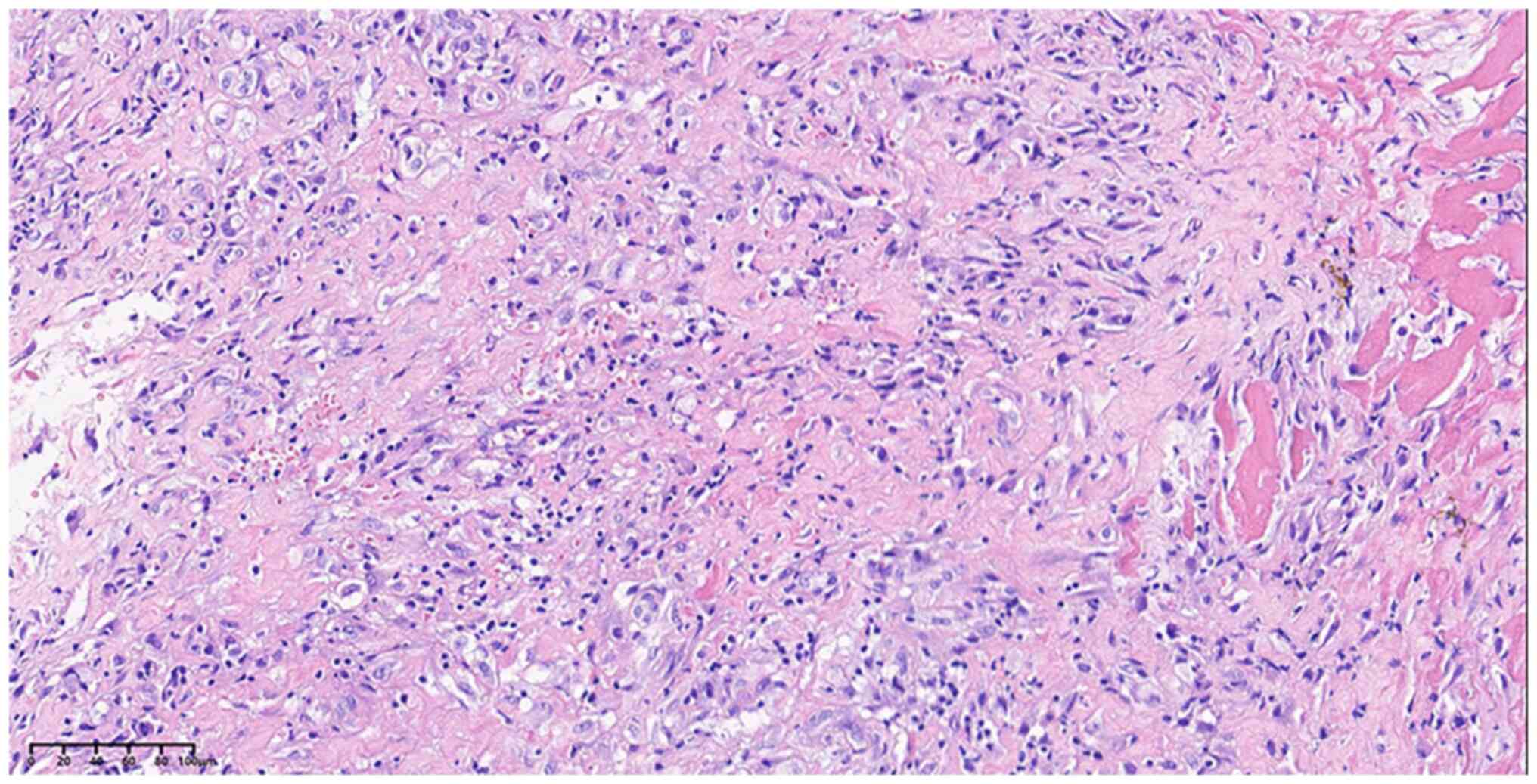
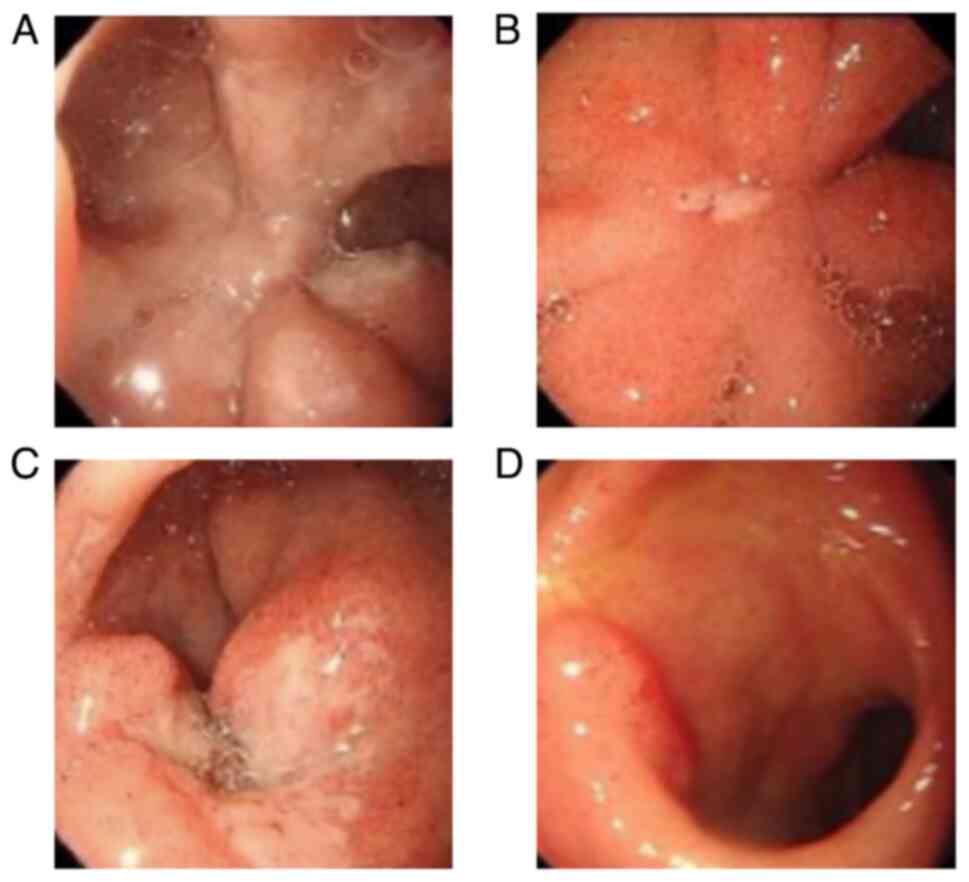
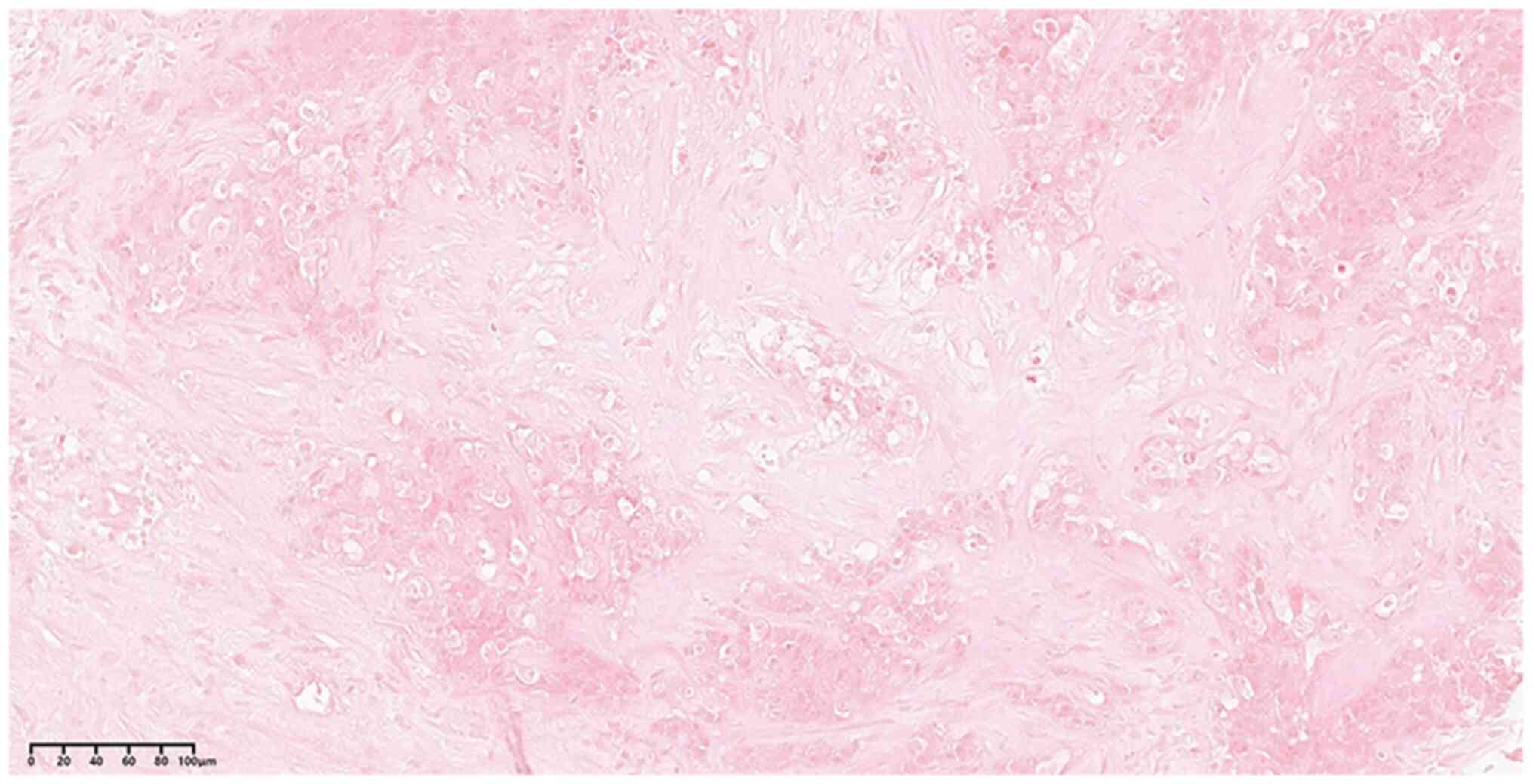
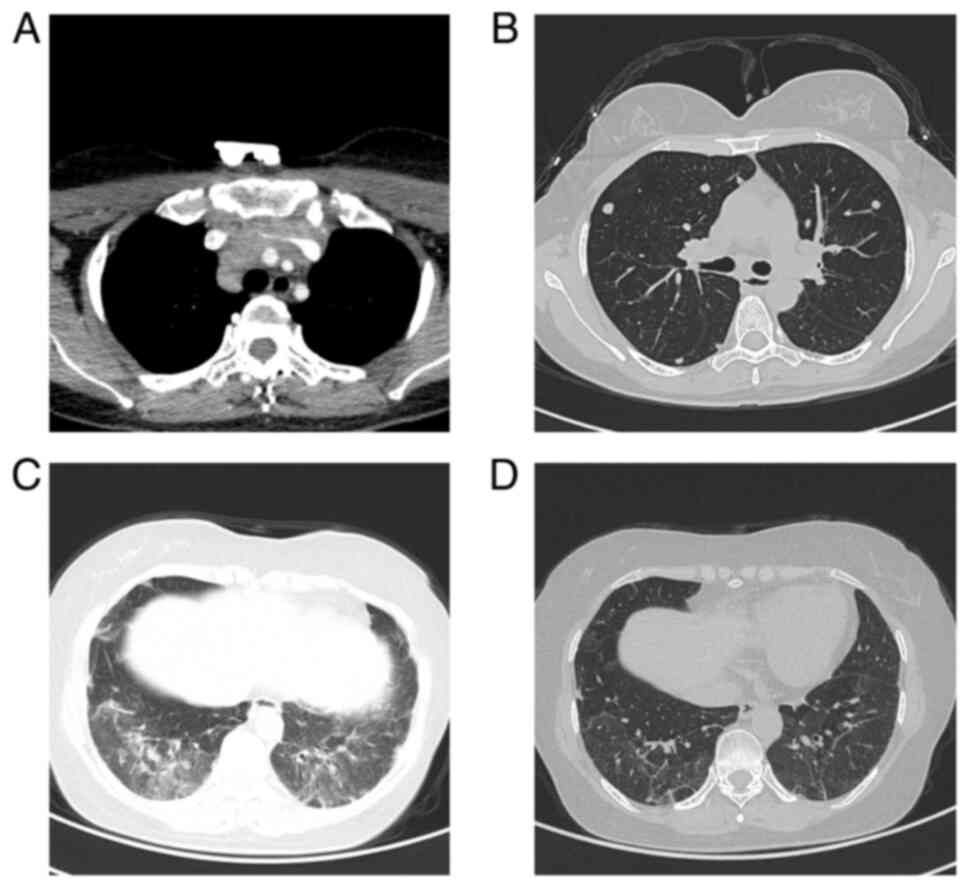
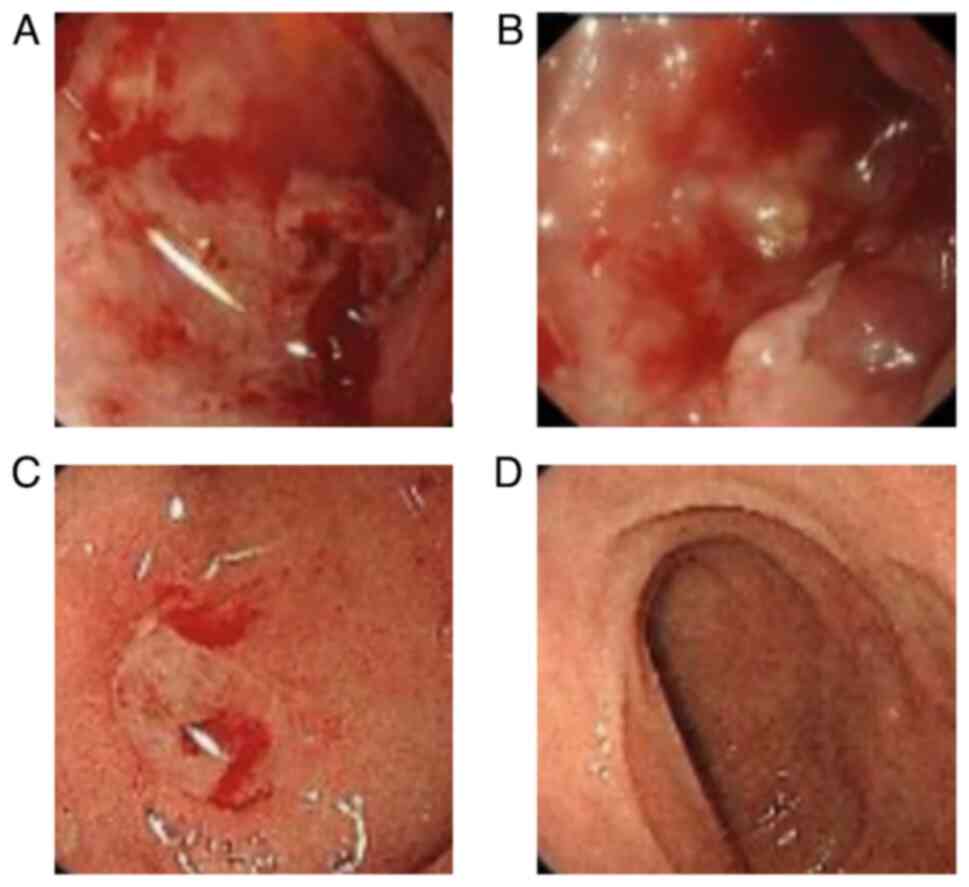
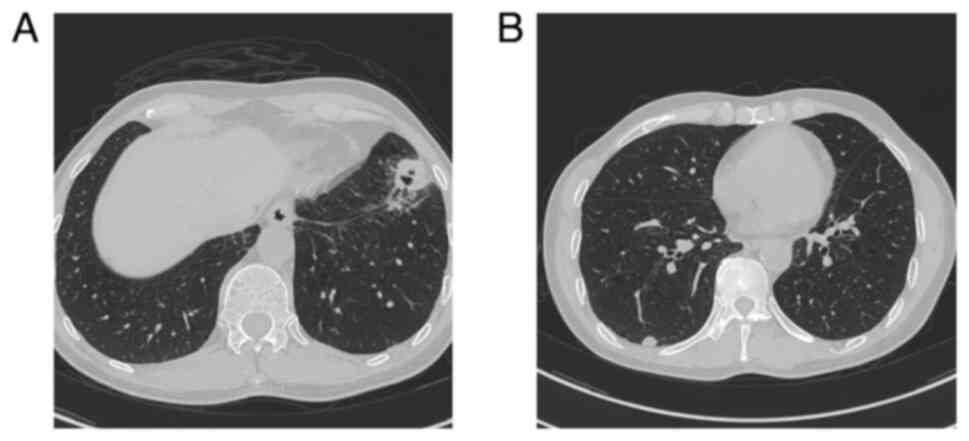
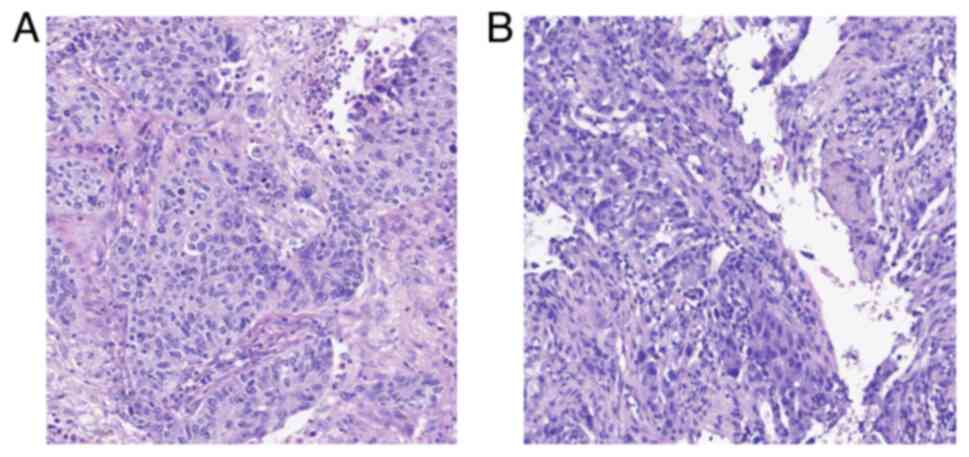
|
1
|
Pardoll DM: The blockade of immune
checkpoints in cancer immunotherapy. Nat Rev Cancer. 12:252–264.
2012.PubMed/NCBI View
Article : Google Scholar
|
|
2
|
Som A, Mandaliya R, Alsaadi D, Farshidpour
M, Charabaty A, Malhotra N and Mattar MC: Immune checkpoint
inhibitor-induced colitis: A comprehensive review. World J Clin
Cases. 7:405–418. 2019.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Durrechou Q, Domblides C, Sionneau B,
Lefort F, Quivy A, Ravaud A, Gross-Goupil M and Daste A: Management
of immune checkpoint inhibitor toxicities. Cancer Manag Res.
12:9139–9158. 2020.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Coutzac C, Adam J, Soularue E, Collins M,
Racine A, Mussini C, Boselli L, Kamsukom N, Mateus C and Charrier
M: Colon immune-related adverse events: Anti-CTLA-4 and Anti-PD-1
blockade induce distinct immunopathological entities. J Crohns
Colitis. 11:1238–1246. 2017.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Darnell EP, Mooradian MJ, Baruch EN,
Yilmaz M and Reynolds KL: Immune-related adverse events (irAEs):
Diagnosis, management, and clinical pearls. Curr Oncol Rep.
22(39)2020.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Shieh C, Chalikonda D, Block P, Shinn B
and Kistler CA: Gastrointestinal toxicities of immune checkpoint
inhibitors: A multicenter retrospective analysis. Ann
Gastroenterol. 34:46–52. 2021.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Thompson JA, Schneider BJ, Brahmer J,
Andrews S, Armand P, Bhatia S, Budde LE, Costa L, Davies M,
Dunnington D, et al: Management of immunotherapy-related
toxicities, version 1.2019. J Natl Compr Canc Netw. 17:255–289.
2019.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Pernot S, Ramtohul T and Taieb J:
Checkpoint inhibitors and gastrointestinal immune-related adverse
events. Curr Opin Oncol. 28:264–268. 2016.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Kähler KC, Hassel JC, Heinzerling L,
Loquai C, Mössner R, Ugurel S, Zimmer L and Gutzmer R: ‘Cutaneous
Side Effects’ Committee of the Work Group Dermatological Oncology
(ADO). Management of side effects of immune checkpoint blockade by
anti-CTLA-4 and anti-PD-1 antibodies in metastatic melanoma. J
Dtsch Dermatol Ges. 14:662–681. 2016.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Brahmer JR, Lacchetti C, Schneider BJ,
Atkins MB, Brassil KJ, Caterino JM, Chau I, Ernstoff MS, Gardner
JM, Ginex P, et al: Management of immune-related adverse events in
patients treated with immune checkpoint inhibitor therapy: American
society of clinical oncology clinical practice guideline. J Clin
Oncol. 36:1714–1768. 2018.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Puzanov I, Diab A, Abdallah K, Bingham CO
III, Brogdon C, Dadu R, Hamad L, Kim S, Lacouture ME, LeBoeuf NR,
et al: Managing toxicities associated with immune checkpoint
inhibitors: Consensus recommendations from the society for
immunotherapy of cancer (SITC) toxicity management working group. J
Immunother Cancer. 5(95)2017.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Haanen J, Carbonnel F, Robert C, Kerr KM,
Peters S, Larkin J and Jordan K: ESMO Guidelines Committee.
Management of toxicities from immunotherapy: ESMO clinical practice
guidelines for diagnosis, treatment and follow-up. Ann Oncol. 28
(Suppl 4):iv119–iv142. 2017.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Grover S, Rahma OE, Hashemi N and Lim RM:
Gastrointestinal and hepatic toxicities of checkpoint inhibitors:
Algorithms for management. Am Soc Clin Oncol Educ Book. 38:13–19.
2018.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Collins M, Soularue E, Marthey L and
Carbonnel F: Management of patients with immune checkpoint
inhibitor-induced enterocolitis: A systematic review. Clin
Gastroenterol Hepatol. 18:1393–1403.e1. 2020.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Dougan M, Wang Y, Rubio-Tapia A and Lim
JK: AGA clinical practice update on diagnosis and management of
immune checkpoint inhibitor colitis and hepatitis: Expert review.
Gastroenterology. 160:1384–1393. 2021.PubMed/NCBI View Article : Google Scholar
|
|
16
|
American Joint Committee on Cancer (AJCC):
AJCC Cancer Staging Manual. 8th edition. Springer, Cham, p1032,
2017.
|
|
17
|
Kamada T, Satoh K, Itoh T, Ito M, Iwamoto
J, Okimoto T, Kanno T, Sugimoto M, Chiba T, Nomura S, et al:
Evidence-based clinical practice guidelines for peptic ulcer
disease 2020. J Gastroenterol. 56:303–322. 2021.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Cheng Y, Ling F, Li J, Chen Y, Xu M, Li S
and Zhu L: An updated review of gastrointestinal toxicity induced
by PD-1 inhibitors: From mechanisms to management. Front Immunol.
14(1190850)2023.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Abu-Sbeih H, Ali FS, Qiao W, Lu Y, Patel
S, Diab A and Wang Y: Immune checkpoint inhibitor-induced colitis
as a predictor of survival in metastatic melanoma. Cancer Immunol
Immunother. 68:553–561. 2019.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Wang Y, Abu-Sbeih H, Mao E, Ali N, Ali FS,
Qiao W, Lum P, Raju G, Shuttlesworth G, Stroehlein J and Diab A:
Immune-checkpoint inhibitor-induced diarrhea and colitis in
patients with advanced malignancies: Retrospective review at MD
Anderson. J Immunother Cancer. 6(37)2018.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Tang L, Wang J, Lin N, Zhou Y, He W, Liu J
and Ma X: Immune checkpoint inhibitor-associated colitis: From
mechanism to management. Front Immunol. 12(800879)2021.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Molina GE, Allen IM, Hughes MS, Zubiri L,
Lee H, Mooradian MJ, Reynolds KL, Dougan M and Chen ST: Prognostic
implications of co-occurring dermatologic and gastrointestinal
toxicity from immune checkpoint inhibition therapy for advanced
malignancies: A retrospective cohort study. J Am Acad Dermatol.
82:743–746. 2020.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Johnson DH, Hailemichael Y, Foo WC, Hess
KR, Haymaker CL, Wani KM, Lazar AJ, Saberian CM, Bentebibel SE,
Burton EM, et al: Interleukin-6 is potential target to de-couple
checkpoint inhibitor-induced colitis from antitumor immunity. J
Clin Oncol. 37(2616)2019.
|
|
24
|
Miossec P, Korn T and Kuchroo VK:
Interleukin-17 and type 17 helper T cells. N Engl J Med.
361:888–898. 2009.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Valpione S, Pasquali S, Campana LG, Piccin
L, Mocellin S, Pigozzo J and Chiarion-Sileni V: Sex and
interleukin-6 are prognostic factors for autoimmune toxicity
following treatment with anti-CTLA4 blockade. J Transl Med.
16(94)2018.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Hailemichael Y, Johnson DH, Abdel-Wahab N,
Foo WC, Bentebibel SE, Daher M, Haymaker C, Wani K, Saberian C,
Ogata D, et al: Interleukin-6 blockade abrogates immunotherapy
toxicity and promotes tumor immunity. Cancer Cell. 40:509–523.e6.
2022.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Fa'ak F, Buni M, Falohun A, Lu H, Song J,
Johnson DH, Zobniw CM, Trinh VA, Awiwi MO, Tahon NH, et al:
Selective immune suppression using interleukin-6 receptor
inhibitors for management of immune-related adverse events. J
Immunother Cancer. 11(e006814)2023.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Young K, Lin E, Chen E, Brinkerhoff B,
Scott G and Yu J: Small bowel hemorrhage from check point inhibitor
enteritis: A case report. BMC Gastroenterol. 21(345)2021.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Trystram N, Laly P, Bertheau P, Baroudjian
B, Aparicio T and Gornet JM: Haemorrhagic shock secondary to a
diffuse ulcerative enteritis after Ipilimumab and Nivolumab
treatment for metastatic melanoma: A case report. Ann Palliat Med.
11:837–842. 2022.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Liu WT, Li YF and Hsieh TY: Unusual severe
gastritis and gastric ulcers caused by pembrolizumab. J Postgrad
Med. 68:38–40. 2022.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Kato R, Hayashi H, Sano K, Handa K, Kumode
T, Ueda H, Okuno T, Kawakami H, Matsumura I, Kudo M and Nakagawa K:
Nivolumab-induced hemophilia A presenting as gastric ulcer bleeding
in a patient with NSCLC. J Thorac Oncol. 13:e239–e241.
2018.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Desai M, Chitnavis V and Grider D: Gastric
ulcer in a patient with metastatic lung cancer. Gastroenterology.
156:1572–1573. 2019.PubMed/NCBI View Article : Google Scholar
|