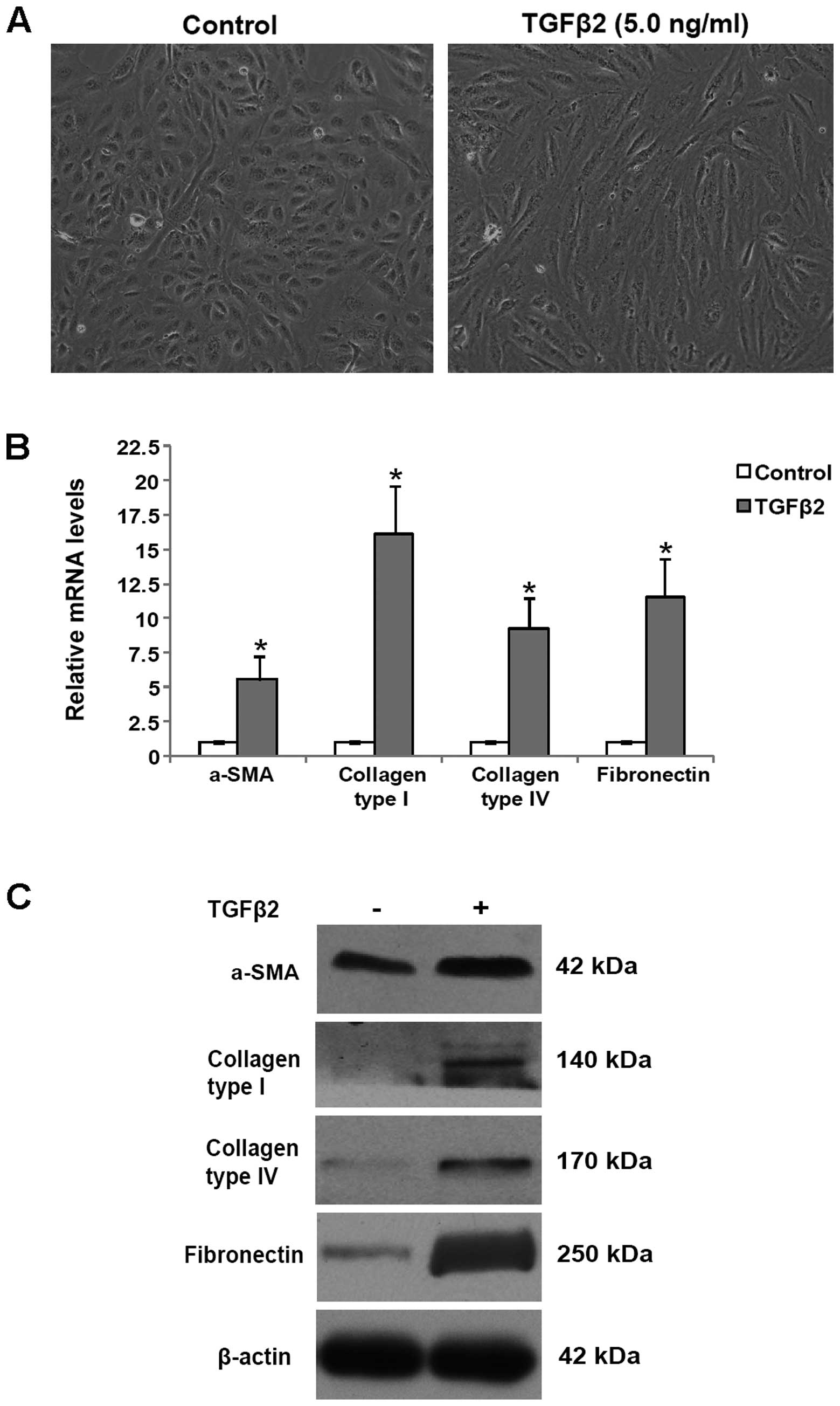
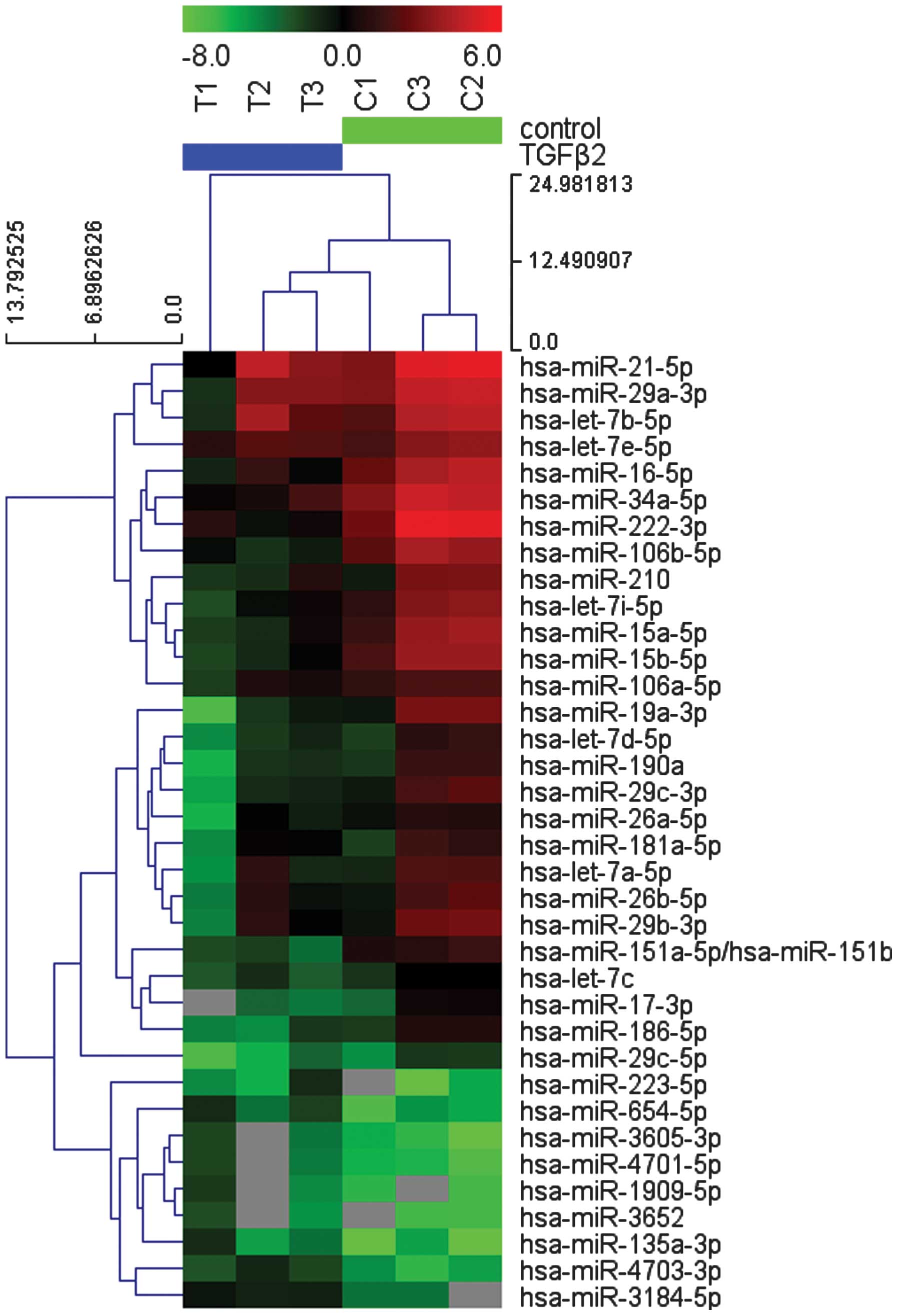
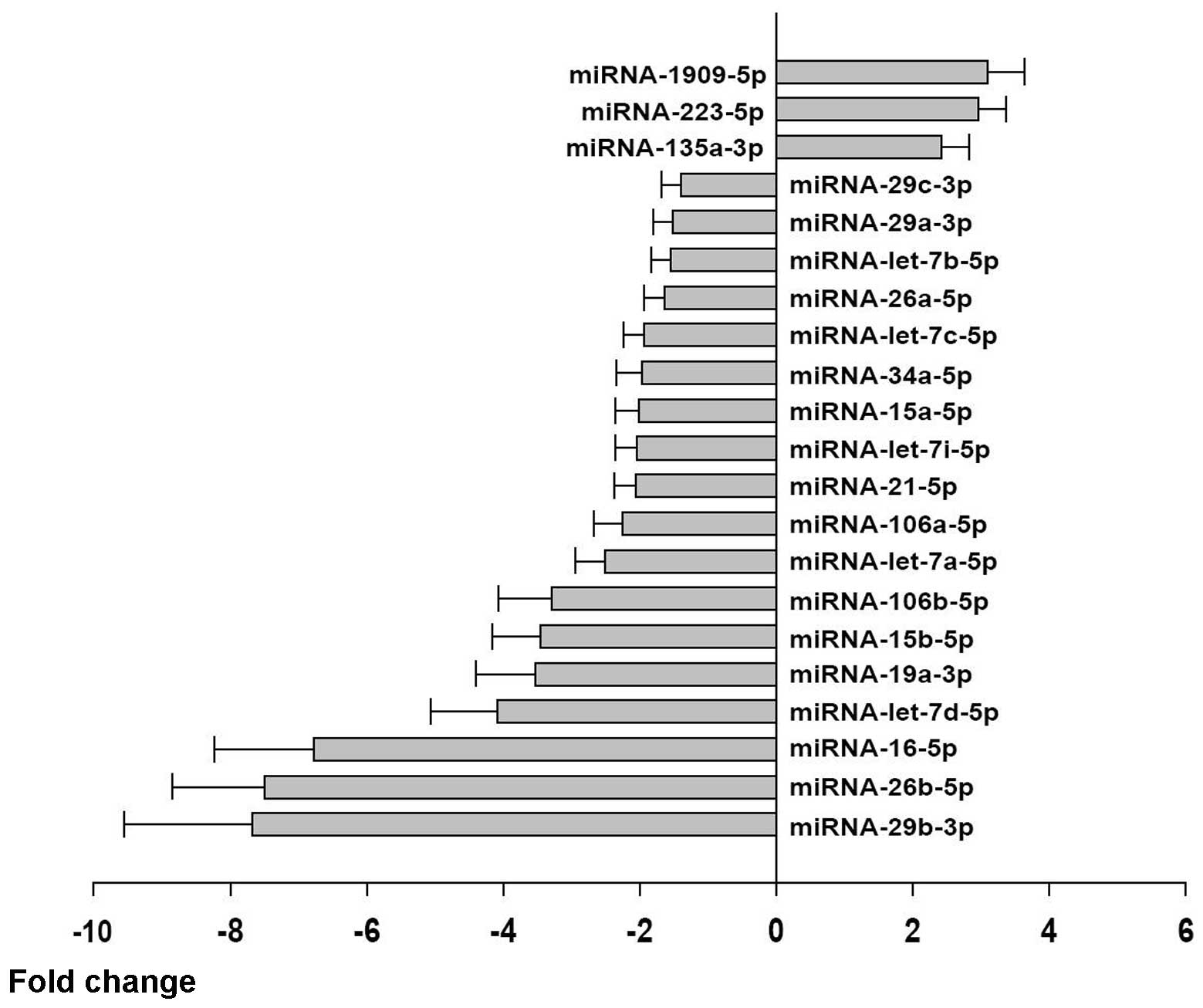
|
1
|
Kroll P, Rodrigues EB and Hoerle S:
Pathogenesis and classification of proliferative diabetic
vitreoretinopathy. Ophthalmologica. 221:78–94. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Pastor JC, de la Rúa ER and Martin F:
Proliferative vitreoretinopathy: risk factors and pathobiology.
Prog Retin Eye Res. 21:127–144. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Cui JZ, Chiu A, Maberley D, Ma P, Samad A
and Matsubara JA: Stage specificity of novel growth factor
expression during development of proliferative vitreoretinopathy.
Eye. 21:200–208. 2007. View Article : Google Scholar
|
|
4
|
Zheng XZ, Du LF and Wang HP: An
immunohistochemical analysis of a rat model of proliferative
vitreoretinopathy and a comparison of the expression of TGF-beta
and PDGF among the induction methods. Bosn J Basic Med Sci.
10:204–209. 2010.
|
|
5
|
Border WA and Noble NA: Transforming
growth factor beta in tissue fibrosis. N Engl J Med. 331:1286–1292.
1994. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kita T, Hata Y, Arita R, et al: Role of
TGF-beta in proliferative vitreoretinal diseases and ROCK as a
therapeutic target. Proc Natl Acad Sci USA. 105:17504–17509. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kita T, Hata Y, Kano K, et al:
Transforming growth factor-beta2 and connective tissue growth
factor in proliferative vitreoretinal diseases: possible
involvement of hyalocytes and therapeutic potential of Rho kinase
inhibitor. Diabetes. 56:231–238. 2007. View Article : Google Scholar
|
|
8
|
Wienholds E, Kloosterman WP, Miska E, et
al: MicroRNA expression in zebrafish embryonic development.
Science. 309:310–311. 2005. View Article : Google Scholar
|
|
9
|
Yi R, O’Carroll D, Pasolli HA, et al:
Morphogenesis in skin is governed by discrete sets of
differentially expressed microRNAs. Nat Genet. 38:356–362. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Allegra A, Alonci A, Campo S, et al:
Circulating microRNAs: New biomarkers in diagnosis, prognosis and
treatment of cancer (Review). Int J Oncol. 41:1897–1912.
2012.PubMed/NCBI
|
|
11
|
He J, Jing Y, Li W, et al: Roles and
mechanism of miR-199a and miR-125b in tumor angiogenesis. PLoS One.
8:e566472013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Rana TM: Illuminating the silence:
understanding the structure and function of small RNAs. Nat Rev Mol
Cell Biol. 8:23–36. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
13
|
Filipowicz W, Bhattacharyya SN and
Sonenberg N: Mechanisms of post-transcriptional regulation by
microRNAs: are the answers in sight? Nat Rev Genet. 9:102–114.
2008. View
Article : Google Scholar : PubMed/NCBI
|
|
14
|
Gregory PA, Bert AG, Paterson EL, et al:
The miR-200 family and miR-205 regulate epithelial to mesenchymal
transition by targeting ZEB1 and SIP1. Nat Cell Biol. 10:593–601.
2008. View
Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ru P, Steele R, Newhall P, Phillips NJ,
Toth K and Ray RB: miRNA-29b suppresses prostate cancer metastasis
by regulating epithelial-mesenchymal transition signaling. Mol
Cancer Ther. 11:1166–1173. 2012. View Article : Google Scholar
|
|
16
|
Siemens H, Jackstadt R, Hunten S, et al:
miR-34 and SNAIL form a double-negative feedback loop to regulate
epithelial-mesenchymal transitions. Cell Cycle. 10:4256–4271. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zhou Q, Fan J, Ding X, et al:
TGF-{beta}-induced MiR-491–5p expression promotes Par-3 degradation
in rat proximal tubular epithelial cells. J Biol Chem.
285:40019–40027. 2010.
|
|
18
|
Jackson SJ, Zhang Z, Feng D, et al: Rapid
and widespread suppression of self-renewal by microRNA-203 during
epidermal differentiation. Development. 140:1882–1891. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Luningschror P, Hauser S, Kaltschmidt B
and Kaltschmidt C: MicroRNAs in pluripotency, reprogramming and
cell fate induction. Biochim Biophys Acta. 1833:1894–1903. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Khan AA, Penny LA, Yuzefpolskiy Y, Sarkar
S and Kalia V: MicroRNA-17~92 regulates effector and memory CD8
T-cell fates by modulating proliferation in response to infections.
Blood. 121:4473–4483. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lin XZ, Luo J, Zhang LP, Wang W, Shi HB
and Zhu JJ: MiR-27a suppresses triglyceride accumulation and
affects gene mRNA expression associated with fat metabolism in
dairy goat mammary gland epithelial cells. Gene. 521:15–23. 2013.
View Article : Google Scholar
|
|
22
|
Qiu J, Zhou XY, Zhou XG, Cheng R, Liu HY
and Li Y: Neuroprotective effects of microRNA-210 against
oxygen-glucose deprivation through inhibition of apoptosis in PC12
cells. Mol Med Rep. 7:1955–1959. 2013.
|
|
23
|
Peng WJ, Tao JH, Mei B, et al:
MicroRNA-29: a potential therapeutic target for systemic sclerosis.
Expert Opin Ther Targets. 16:875–879. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
van Rooij E, Sutherland LB, Thatcher JE,
et al: Dysregulation of microRNAs after myocardial infarction
reveals a role of miR-29 in cardiac fibrosis. Proc Natl Acad Sci
USA. 105:13027–13032. 2008.
|
|
25
|
Vettori S, Gay S and Distler O: Role of
microRNAs in fibrosis. Open Rheumatol J. 6:130–139. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Rothschild SI, Tschan MP, Federzoni EA, et
al: MicroRNA-29b is involved in the Src-ID1 signaling pathway and
is dysregulated in human lung adenocarcinoma. Oncogene.
31:4221–4232. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wang C, Bian Z, Wei D and Zhang JG:
miR-29b regulates migration of human breast cancer cells. Mol Cell
Biochem. 352:197–207. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Zhang W, Qian JX, Yi HL, et al: The
microRNA-29 plays a central role in osteosarcoma pathogenesis and
progression. Mol Biol. 46:622–627. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Liu Y, Taylor NE, Lu L, et al: Renal
medullary microRNAs in Dahl salt-sensitive rats: miR-29b regulates
several collagens and related genes. Hypertension. 55:974–982.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Maurer B, Stanczyk J, Jungel A, et al:
MicroRNA-29, a key regulator of collagen expression in systemic
sclerosis. Arthritis Rheum. 62:1733–1743. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Li J, Kong X, Zhang J, Luo Q, Li X and
Fang L: MiRNA-26b inhibits proliferation by targeting PTGS2 in
breast cancer. Cancer Cell Int. 13:72013. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Liu XX, Li XJ, Zhang B, et al:
MicroRNA-26b is underexpressed in human breast cancer and induces
cell apoptosis by targeting SLC7A11. FEBS Lett. 585:1363–1367.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Wu N, Zhao X, Liu M, et al: Role of
microRNA-26b in glioma development and its mediated regulation on
EphA2. PLoS One. 6:e162642011. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Pandit KV, Corcoran D, Yousef H, et al:
Inhibition and role of let-7d in idiopathic pulmonary fibrosis. Am
J Respir Crit Care Med. 182:220–229. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Smith AL, Iwanaga R, Drasin DJ, et al: The
miR-106b-25 cluster targets Smad7, activates TGF-beta signaling,
and induces EMT and tumor initiating cell characteristics
downstream of Six1 in human breast cancer. Oncogene. 31:5162–5171.
2012. View Article : Google Scholar
|
|
36
|
Savita U and Karunagaran D:
MicroRNA-106b-25 cluster targets beta-TRCP2, increases the
expression of Snail and enhances cell migration and invasion in
H1299 (non small cell lung cancer) cells. Biochem Biophys Res
Commun. 434:841–847. 2013. View Article : Google Scholar
|
|
37
|
Kan T, Sato F, Ito T, et al: The
miR-106b-25 polycistron, activated by genomic amplification,
functions as an oncogene by suppressing p21 and Bim.
Gastroenterology. 136:1689–1700. 2009. View Article : Google Scholar
|
|
38
|
Li F, Liu J and Li S: MicorRNA 106b
approximately 25 cluster and gastric cancer. Surg Oncol. 22:e7–e10.
2013. View Article : Google Scholar
|
|
39
|
Slaby O, Jancovicova J, Lakomy R, et al:
Expression of miRNA-106b in conventional renal cell carcinoma is a
potential marker for prediction of early metastasis after
nephrectomy. J Exp Clin Cancer Res. 29:902010. View Article : Google Scholar
|
|
40
|
Calin GA, Dumitru CD, Shimizu M, Bichi R,
Zupo S, Noch E, Aldler H, Rattan S, Keating M, Rai K, Rassenti L,
Kipps T, Negrini M, Bullrich F and Croce CM: Frequent deletions and
down-regulation of micro- RNA genes miR15 and miR16 at 13q14 in
chronic lymphocytic leukemia. Proc Natl Acad Sci USA.
99:15524–15529. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Calin GA, Ferracin M, Cimmino A, Di Leva
G, Shimizu M, Wojcik SE, Iorio MV, Visone R, Sever NI, Fabbri M,
Iuliano R, Palumbo T, Pichiorri F, Roldo C, Garzon R, Sevignani C,
Rassenti L, Alder H, Volinia S, Liu CG, Kipps TJ, Negrini M and
Croce CM: A MicroRNA signature associated with prognosis and
progression in chronic lymphocytic leukemia. N Engl J Med.
353:1793–1801. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Linsley PS, Schelter J, Burchard J, et al:
Transcripts targeted by the microRNA-16 family cooperatively
regulate cell cycle progression. Mol Cell Biol. 27:2240–2252. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Yuan JY, Wang F, Yu J, Yang GH, Liu XL and
Zhang JW: MicroRNA-223 reversibly regulates erythroid and
megakaryocytic differentiation of K562 cells. J Cell Mol Med.
13:4551–4559. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Sugatani T and Hruska KA: MicroRNA-223 is
a key factor in osteoclast differentiation. J Cell Biochem.
101:996–999. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Johnnidis JB, Harris MH, Wheeler RT, et
al: Regulation of progenitor cell proliferation and granulocyte
function by microRNA-223. Nature. 451:1125–1129. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Li J, Guo Y, Liang X, et al: MicroRNA-223
functions as an oncogene in human gastric cancer by targeting
FBXW7/hCdc4. J Cancer Res Clin Oncol. 138:763–774. 2012. View Article : Google Scholar : PubMed/NCBI
|