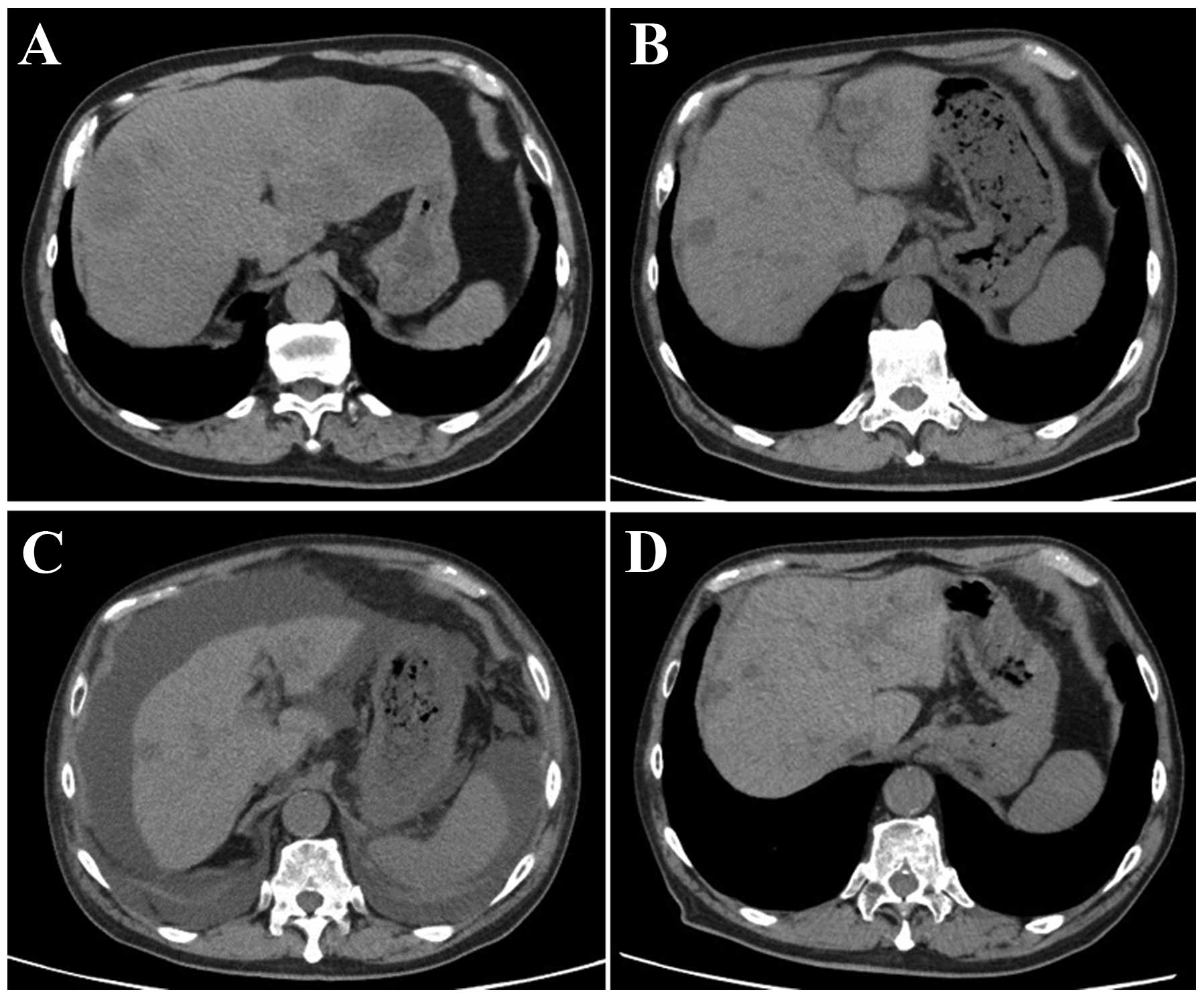
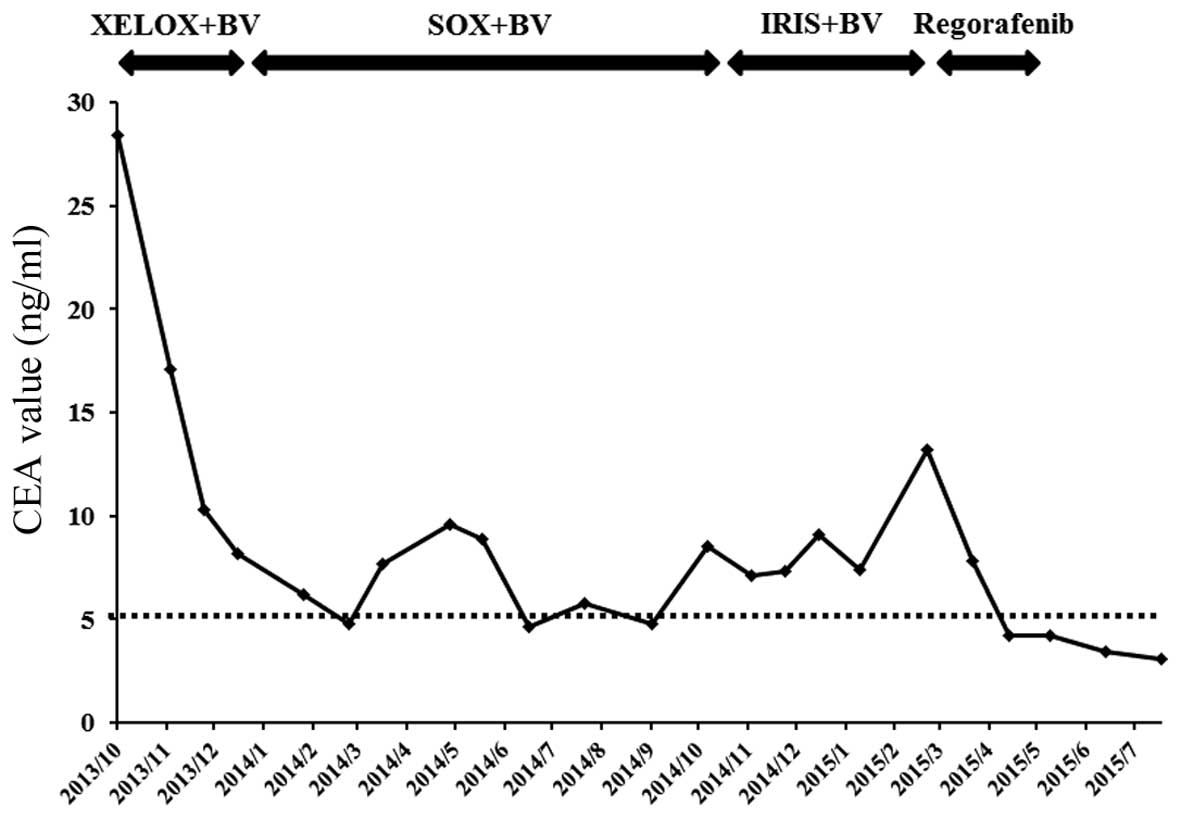
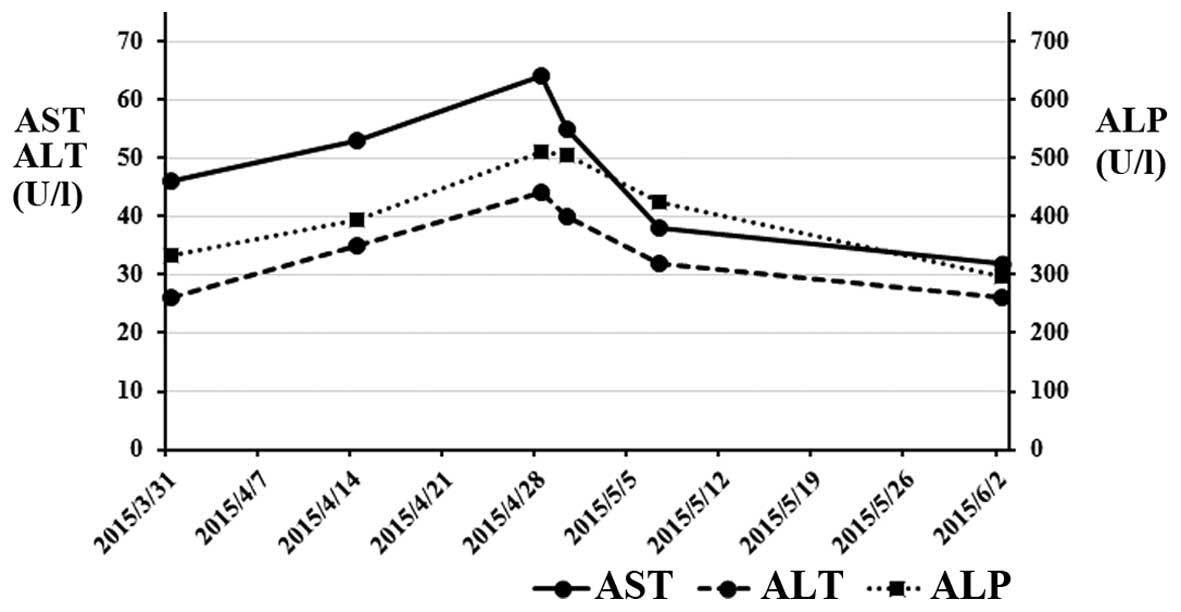
|
1
|
Wilhelm SM, Dumas J, Adnane L, Lynch M,
Carter CA, Schütz G, Thierauch KH and Zopf D: Regorafenib (BAY
73–4506): A new oral multikinase inhibitor of angiogenic, stromal
and oncogenic receptor tyrosine kinases with potent preclinical
antitumor activity. Int J Cancer. 129:245–255. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Grothey A, Van Cutsem E, Sobrero A, Siena
S, Falcone A, Ychou M, Humblet Y, Bouché O, Mineur L, Barone C, et
al: CORRECT Study Group: Regorafenib monotherapy for previously
treated metastatic colorectal cancer (CORRECT): An international,
multicentre, randomised, placebo-controlled, phase 3 trial. Lancet.
381:303–312. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Li J, Qin S, Xu R, Yau TC, Ma B, Pan H, Xu
J, Bai Y, Chi Y, Wang L, et al: CONCUR Investigators: Regorafenib
plus best supportive care versus placebo plus best supportive care
in Asian patients with previously treated metastatic colorectal
cancer (CONCUR): A randomised, double-blind, placebo-controlled,
phase 3 trial. Lancet Oncol. 16:619–629. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kim ST, Kim TW, Kim KP, Kim TY, Han SW,
Lee JY, Lim SH, Lee MY, Kim H and Park YS: Regorafenib as Salvage
Treatment in Korean Patients with Refractory Metastatic Colorectal
Cancer. Cancer Res Treat. 47:790–795. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Yoshino T, Komatsu Y, Yamada Y, Yamazaki
K, Tsuji A, Ura T, Grothey A, Van Cutsem E, Wagner A, Cihon F, et
al: Randomized phase III trial of regorafenib in metastatic
colorectal cancer: Analysis of the CORRECT Japanese and
non-Japanese subpopulations. Invest New Drugs. 33:740–750. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Rey JB, Launay-Vacher V and Tournigand C:
Regorafenib as a single-agent in the treatment of patients with
gastrointestinal tumors: An overview for pharmacists. Target Oncol.
10:199–213. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Carr BI, Cavallini A, Lippolis C,
D'Alessandro R, Messa C, Refolo MG and Tafaro A: Fluoro-Sorafenib
(Regorafenib) effects on hepatoma cells: Growth inhibition,
quiescence, and recovery. J Cell Physiol. 228:292–297. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ye L, Yang X, Guo E, Chen W, Lu L, Wang Y,
Peng X, Yan T, Zhou F and Liu Z: Sorafenib metabolism is
significantly altered in the liver tumor tissue of hepatocellular
carcinoma patient. PLoS One. 9:e966642014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Boudou-Rouquette P, Narjoz C, Golmard JL,
Thomas-Schoemann A, Mir O, Taieb F, Durand JP, Coriat R, Dauphin A,
Vidal M, et al: Early sorafenib-induced toxicity is associated with
drug exposure and UGTIA9 genetic polymorphism in patients with
solid tumors: A preliminary study. PLoS One. 7:e428752012.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lee SL, Chang ED, Na SJ, Kim JS, An HJ, Ko
YH and Won HS: Pseudocirrhosis of breast cancer metastases to the
liver treated by chemotherapy. Cancer Res Treat. 46:98–103. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Jeong WK, Choi SY and Kim J:
Pseudocirrhosis as a complication after chemotherapy for hepatic
metastasis from breast cancer. Clin Mol Hepatol. 19:190–194. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kang SP, Taddei T, McLennan B and Lacy J:
Pseudocirrhosis in a pancreatic cancer patient with liver
metastases: A case report of complete resolution of pseudocirrhosis
with an early recognition and management. World J Gastroenterol.
14:1622–1624. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kobashigawa C, Nakamoto M, Hokama A,
Hirata T, Kinjo F and Fujita J: Pseudocirrhosis in metastatic
esophageal cancer. South Med J. 103:488–489. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Harry BL, Smith ML, Burton JR Jr, Dasari
A, Eckhardt SG and Diamond JR: Medullary thyroid cancer and
pseudocirrhosis: Case report and literature review. Curr Oncol.
19:e36–e41. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wanless IR: Micronodular transformation
(nodular regenerative hyperplasia) of the liver: A report of 64
cases among 2,500 autopsies and a new classification of benign
hepatocellular nodules. Hepatology. 11:787–797. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ghabril M and Vuppalanchi R: Drug-induced
nodular regenerative hyperplasia. Semin Liver Dis. 34:240–245.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Viganò L, Rubbia-Brandt L, De Rosa G,
Majno P, Langella S, Toso C, Mentha G and Capussotti L: Nodular
Regenerative Hyperplasia in Patients Undergoing Liver Resection for
Colorectal Metastases After Chemotherapy: Risk Factors,
Preoperative Assessment and Clinical Impact. Ann Surg Oncol.
22:4149–4157. 2015. View Article : Google Scholar : PubMed/NCBI
|