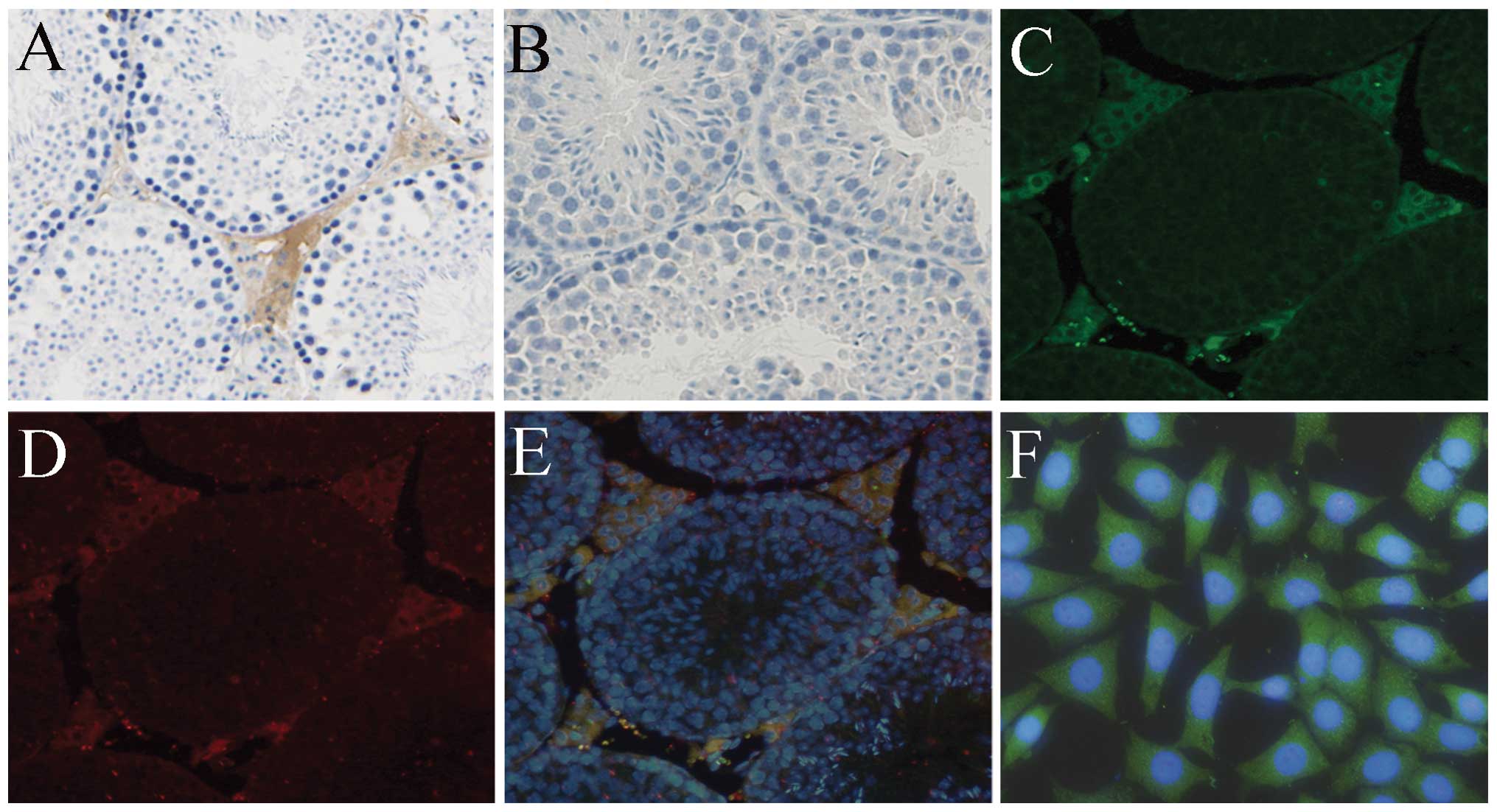
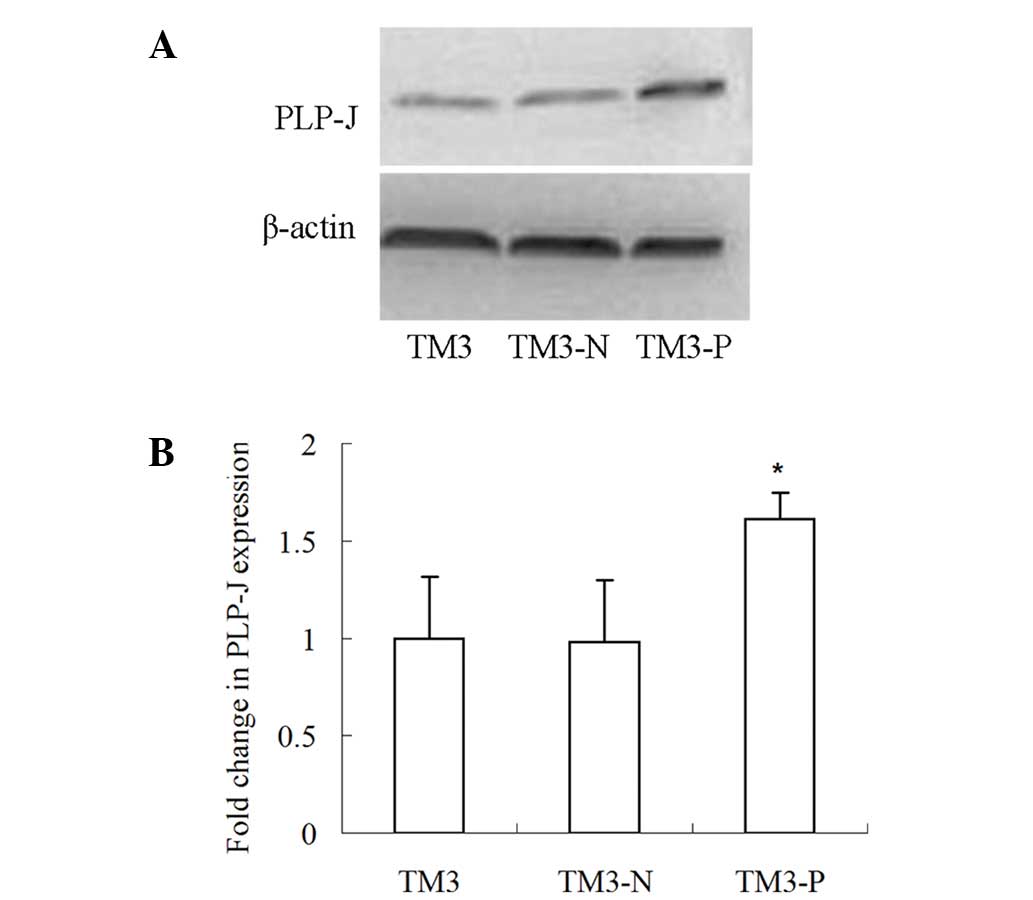
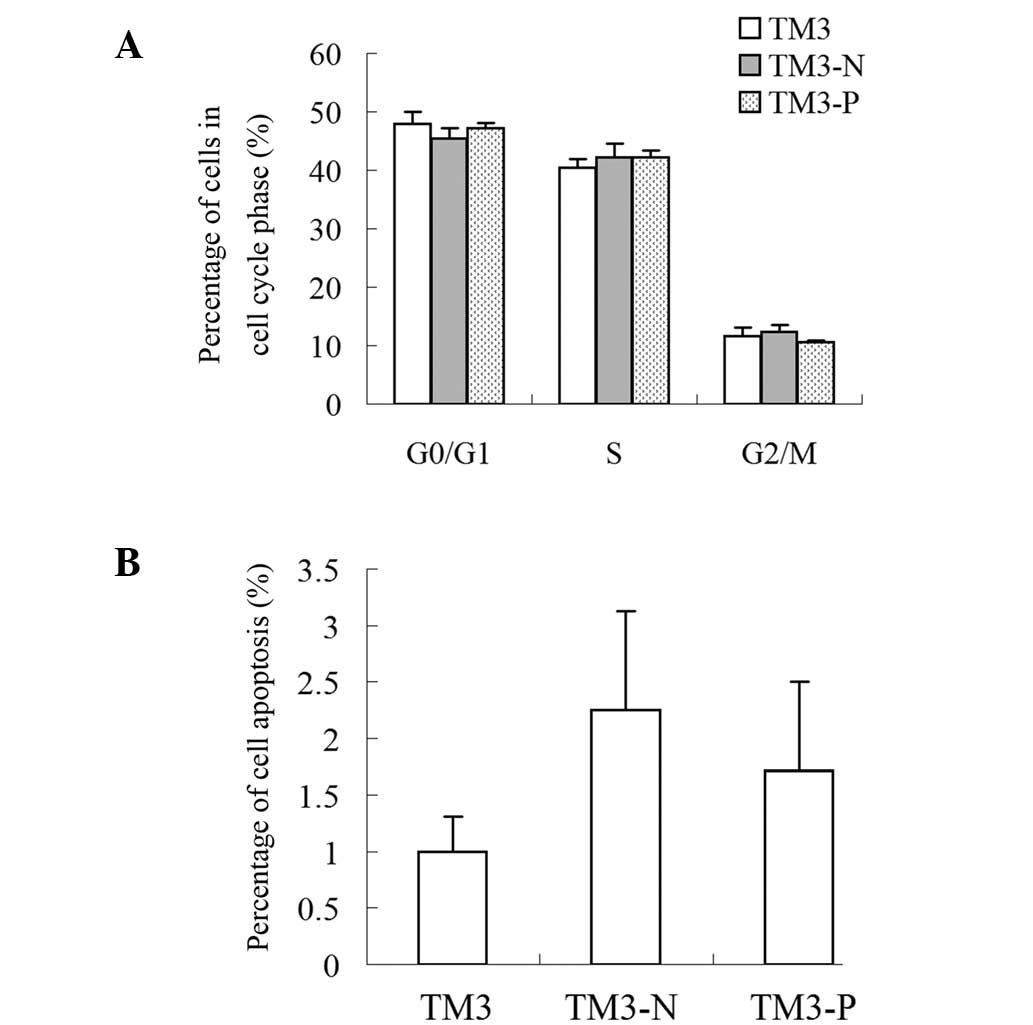
|
1
|
Mallon AM, Wilming L, Weekes J, et al:
Organization and evolution of a gene-rich region of the mouse
genome: a 12.7-Mb region deleted in the Del(13)Svea36H mouse.
Genome Res. 14:1888–1901. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Soares MJ: The prolactin and growth
hormone families: pregnancy-specific hormones/cytokines at the
maternal-fetal interface. Reprod Biol Endocrinol. 2:512004.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wiemers DO, Shao LJ, Ain R, Dai G and
Soares MJ: The mouse prolactin gene family locus. Endocrinology.
144:313–325. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Simmons DG, Rawn S, Davies A, Hughes M and
Cross JC: Spatial and temporal expression of the 23 murine
Prolactin/Placental Lactogen-related genes is not associated with
their position in the locus. BMC Genomics. 9:3522008. View Article : Google Scholar
|
|
5
|
Wit JM, Drayer NM, Jansen M, et al: Total
deficiency of growth hormone and prolactin, and partial deficiency
of thyroid stimulating hormone in two Dutch families: a new variant
of hereditary pituitary deficiency. Horm Res. 32:170–177. 1989.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Featherstone K, White MR and Davis JR: The
prolactin gene: a paradigm of tissue-specific gene regulation with
complex temporal transcription dynamics. J Neuroendocrinol.
24:977–990. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ho-Chen JK, Bustamante JJ and Soares MJ:
Prolactin-like protein-f subfamily of placental hormones/cytokines:
responsiveness to maternal hypoxia. Endocrinology. 148:559–565.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ishibashi K and Imai M: Identification of
four new members of the rat prolactin/growth hormone gene family.
Biochem Biophys Res Commun. 262:575–578. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Ain R, Dai G, Dunmore JH, Godwin AR and
Soares MJ: A prolactin family paralog regulates reproductive
adaptations to a physiological stressor. Proc Natl Acad Sci USA.
101:16543–16548. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Toft DJ and Linzer DI: Prolactin
(PRL)-like protein J, a novel member of the PRL/growth hormone
family, is exclusively expressed in maternal decidua.
Endocrinology. 140:5095–5101. 1999.PubMed/NCBI
|
|
11
|
Hiraoka Y, Ogawa M, Sakai Y, et al: PLP-I:
a novel prolactin-like gene in rodents. Biochim Biophys Acta.
1447:291–297. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Dai G, Wang D, Liu B, et al: Three novel
paralogs of the rodent prolactin gene family. J Endocrinol.
166:63–75. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Alam SM, Konno T, Sahgal N, Lu L and
Soares MJ: Decidual cells produce a heparin-binding prolactin
family cytokine with putative intrauterine regulatory actions. J
Biol Chem. 283:18957–18968. 2008. View Article : Google Scholar
|
|
14
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
|
|
15
|
Kimura F, Takakura K, Takebayashi K, et
al: Messenger ribonucleic acid for the mouse decidual prolactin is
present and induced during in vitro decidualization of
endometrial stromal cells. Gynecol Endocrinol. 15:426–432. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Knox K, Leuenberger D, Penn AA and Baker
JC: Global hormone profiling of murine placenta reveals Secretin
expression. Placenta. 32:811–816. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Johnston DS, Olivas E, DiCandeloro P and
Wright WW: Stage-specific changes in GDNF expression by rat Sertoli
cells: a possible regulator of the replication and differentiation
of stem spermatogonia. Biol Reprod. 85:763–769. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Johnston DS, Wright WW, Dicandeloro P,
Wilson E, Kopf GS and Jelinsky SA: Stage-specific gene expression
is a fundamental characteristic of rat spermatogenic cells and
Sertoli cells. Proc Natl Acad Sci USA. 105:8315–8320. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Pang AL, Johnson W, Ravindranath N, Dym M,
Rennert OM and Chan WY: Expression profiling of purified male germ
cells: stage-specific expression patterns related to meiosis and
postmeiotic development. Physiol Genomics. 24:75–85. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Midzak AS, Chen H, Papadopoulos V and
Zirkin BR: Leydig cell aging and the mechanisms of reduced
testosterone synthesis. Mol Cell Endocrinol. 299:23–31. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zirkin BR and Tenover JL: Aging and
declining testosterone: past, present, and hopes for the future. J
Androl. 33:1111–1118. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wang X and Stocco DM: The decline in
testosterone biosynthesis during male aging: a consequence of
multiple alterations. Mol Cell Endocrinol. 238:1–7. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Emmelot-Vonk MH, Verhaar HJ, Nakhai Pour
HR, et al: Effect of testosterone supplementation on functional
mobility, cognition, and other parameters in older men: a
randomized controlled trial. JAMA. 299:39–52. 2008.
|
|
24
|
Travison TG, Morley JE, Araujo AB,
O’Donnell AB and McKinlay JB: The relationship between libido and
testosterone levels in aging men. J Clin Endocrinol Metab.
91:2509–2513. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Walker WH: Molecular mechanisms of
testosterone action in spermatogenesis. Steroids. 74:602–607. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Ahn SW, Gang GT, Kim YD, et al: Insulin
directly regulates steroidogenesis via induction of the orphan
nuclear receptor DAX-1 in testicular Leydig cells. J Biol Chem.
288:15937–15946. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Matzkin ME, Yamashita S and Ascoli M: The
ERK1/2 pathway regulates testosterone synthesis by coordinately
regulating the expression of steroidogenic genes in Leydig cells.
Mol Cell Endocrinol. 370:130–137. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Mishra J, Gautam M, Dadhich R, Kowtharapu
BS and Majumdar SS: Peritubular cells may modulate Leydig
cell-mediated testosterone production through a nonclassic pathway.
Fertil Steril. 98:1308–1317. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Martin LJ and Tremblay JJ: Nuclear
receptors in Leydig cell gene expression and function. Biol Reprod.
83:3–14. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zhao L, Hao J, Hu J, et al: Expression of
proliferin-related protein in testis and the biological
significance in testosterone production. Mol Cell Endocrinol.
343:25–31. 2011. View Article : Google Scholar : PubMed/NCBI
|