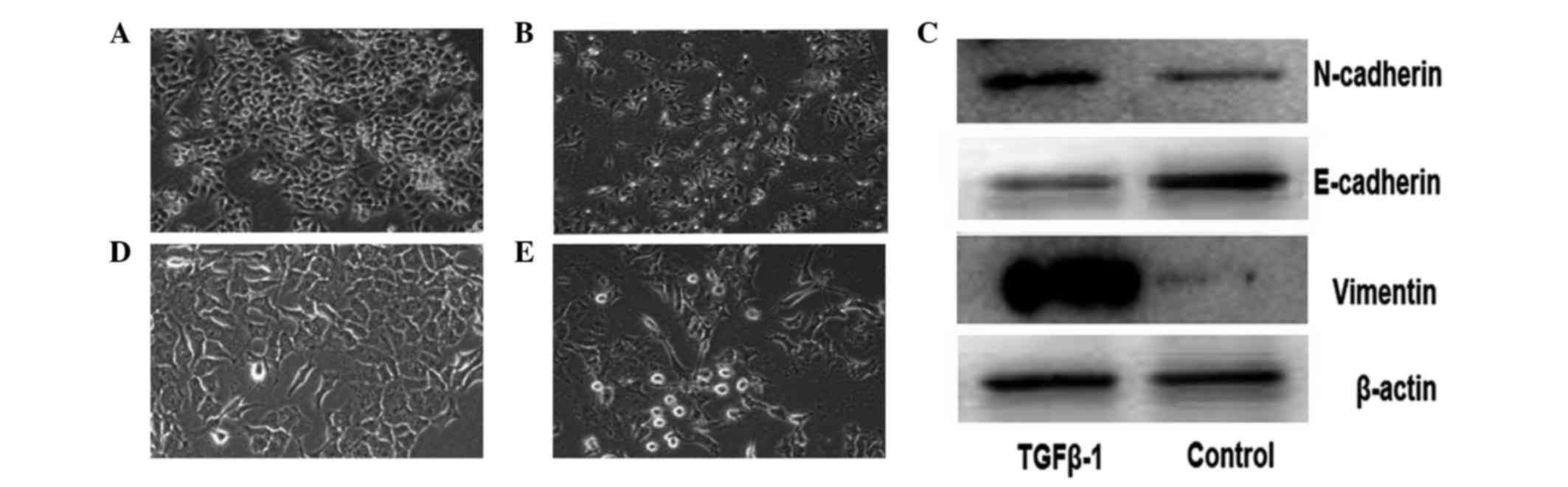
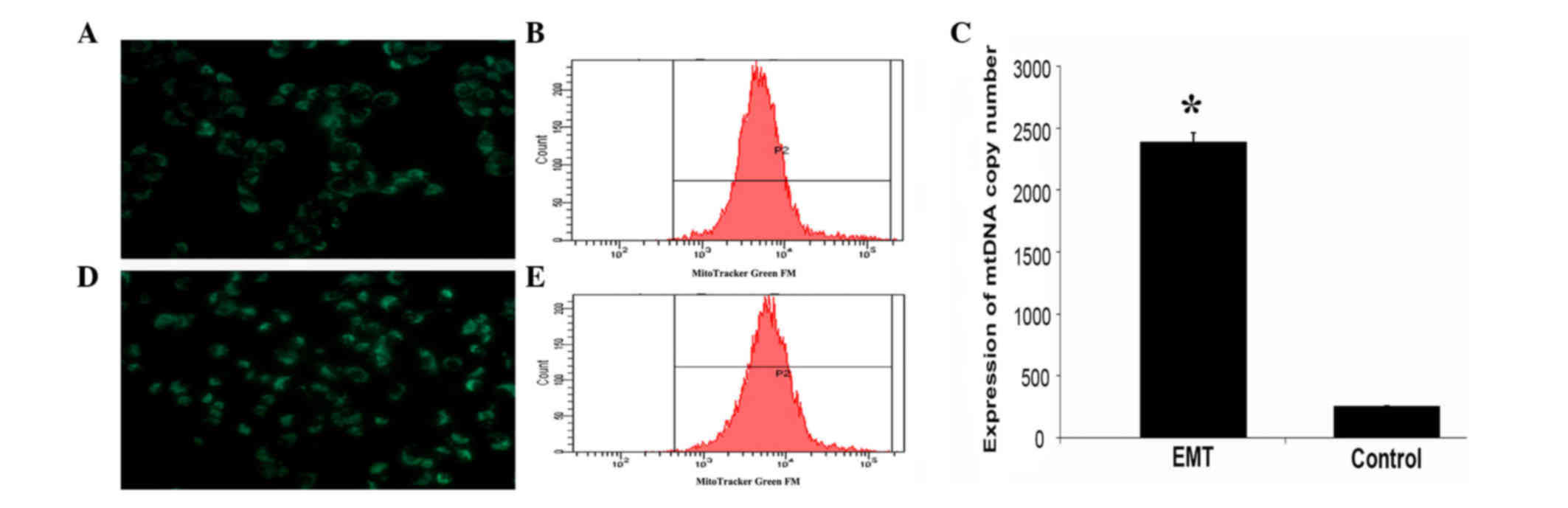
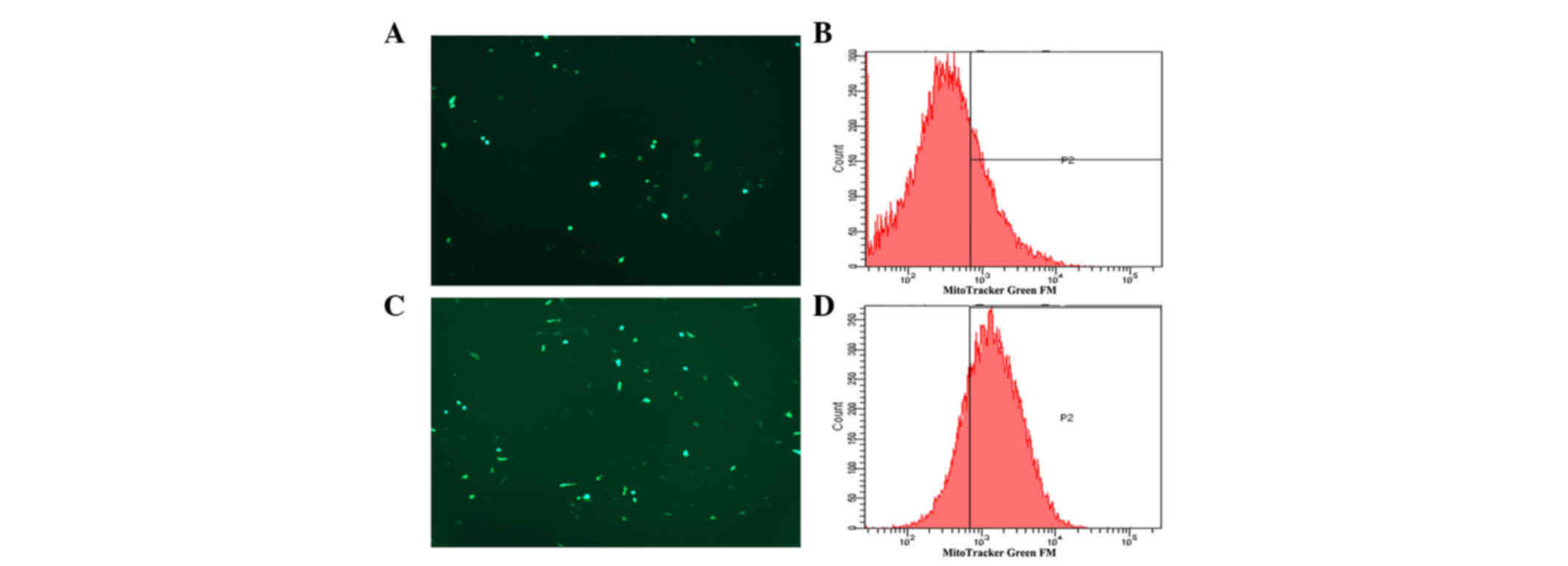
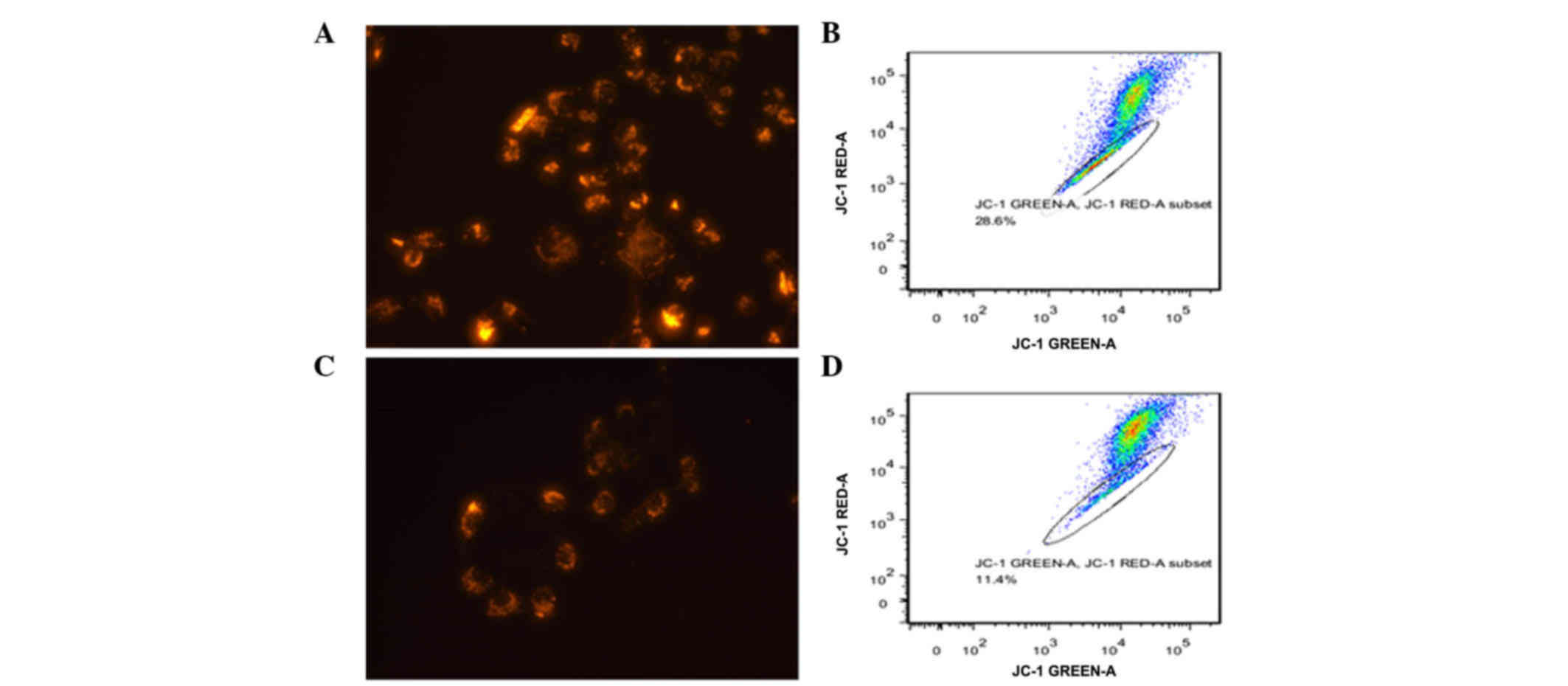
|
1
|
Kim AJ, Jee HJ, Song N, Kim M, Jeong SY
and Yun J: p21 (WAF1/C1P1)
deficiency induces mitochondrial dysfunction in HCT116 colon cancer
cells. Biochem Biophys Res Commun. 430:653–658. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Kang D and Hamasaki N: Alterations of
mitochondrial DNA in common diseases and disease states: Aging,
neurodegeneration, heart failure, diabetes, and cancer. Curr Med
Chem. 12:429–441. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Jose C and Rossignol R: Rationale for
mitochondria-targeting strategies in cancer bioenergetic therapies.
Int J Biochem Cell Biol. 45:123–129. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Gottfried E, Kreutz M and Mackensen A:
Tumor metabolism as modulator of immune response and tumor
progression. Semin Cancer Biol. 22:335–341. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Yilmaz M and Christofori G: EMT, the
cytoskeleton, and cancer cell invasion. Cancer Metastasis Rev.
28:15–33. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Pirozzi G, Tirino V, Camerlingo R, Franco
R, La Rocca A, Liguori E, Martucci N, Paino F, Normanno N and Rocco
G: Epithelial to mesenchymal transition by TGFβ-1 induction
increases stemness characteristics in primary non small cell lung
cancer cell line. PLoS One. 6:e215482011. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Moreno-Bueno G, Peinado H, Molina P,
Olmeda D, Cubillo E, Santos V, Palacios J, Portillo F and Cano A:
The morphological and molecular features of the
epithelial-to-mesenchymal transition. Nat Protoc. 4:1591–1613.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Mani SA, Guo W, Liao MJ, Eaton EN, Ayyanan
A, Zhou AY, Brooks M, Reinhard F, Zhang CC, Shipitsin M, et al: The
epithelial-mesenchymal transition generates cells with properties
of stem cells. Cell. 133:704–715. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Fetisova EK, Avetisyan AV, Izyumov DS,
Korotetskaya MV, Chernyak BV and Skulachev VP:
Mitochondria-targeted antioxidant SkQR1 selectively protects MDR
(Pgp 170)-negative cells against oxidative stress. FEBS Lett.
584:562–566. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Venegas V, Wang J, Dimmock D and Wong LJ:
Real-time quantitative PCR analysis of mitochondrial DNA content.
Curr Protoc Hum Genet Chapter. 19:Unit 19.7. 2011. View Article : Google Scholar
|
|
11
|
Ikenaga N, Ohuchida K, Mizumoto K, Akagawa
S, Fujiwara K, Eguchi D, Kozono S, Ohtsuka T, Takahata S and Tanaka
M: Pancreatic cancer cells enhance the ability of collagen
internalization during epithelial-mesenchymal transition. PLoS One.
7:e404342012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Rigoulet M, Yoboue ED and Devin A:
Mitochondrial ROS generation and its regulation: Mechanisms
involved in H(2)O(2) signaling. Antioxid Redox Signal. 14:459–468.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Figueira TR, Barros MH, Camargo AA,
Castilho RF, Ferreira JC, Kowaltowski AJ, Sluse FE, Souza-Pinto NC
and Vercesi AE: Mitochondria as a source of reactive oxygen and
nitrogen species: From molecular mechanisms to human health.
Antioxid Redox Signal. 18:2029–2074. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Brand MD and Nicholls DG: Assessing
mitochondrial dysfunction in cells. Biochem J. 435:297–312. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Chaffer CL and Weinberg RA: A perspective
on cancer cell metastasis. Science. 331:1559–1564. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Bao B, Wang Z, Ali S, Kong D, Li Y, Ahmad
A, Banerjee S, Azmi AS, Miele L and Sarkar FH: Notch-1 induces
epithelial-mesenchymal transition consistent with cancer stem cell
phenotype in pancreatic cancer cells. Cancer Lett. 307:26–36. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wang Z, Li Y, Ahmad A, Azmi AS, Kong D,
Banerjee S and Sarkar FH: Targeting miRNAs involved in cancer stem
cell and EMT regulation: An emerging concept in overcoming drug
resistance. Drug Resist Updat. 13:109–118. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Heldin CH, Vanlandewijck M and Moustakas
A: Regulation of EMT by TGFβ in cancer. FEBS Lett. 586:1959–1970.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Horiguchi K, Sakamoto K, Koinuma D, Semba
K, Inoue A, Inoue S, Fujii H, Yamaguchi A, Miyazawa K, Miyazono K
and Saitoh M: TGF-β drives epithelial-mesenchymal transition
through δEF1-mediated downregulation of ESRP. Oncogene.
31:3190–3201. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Boreddy SR and Srivastava SK: Deguelin
suppresses pancreatic tumor growth and metastasis by inhibiting
epithelial-to-mesenchymal transition in an orthotopic model.
Oncogene. 32:3980–3991. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lou C, Zhang F, Yang M, Zhao J, Zeng W,
Fang X, Zhang Y, Zhang C and Liang W: Naringenin decreases
invasiveness and metastasis by inhibiting TGF-β-induced epithelial
to mesenchymal transition in pancreatic cancer cells. PLoS One.
7:e509562012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Chen EI: Mitochondrial dysfunction and
cancer metastasis. J Bioenerg Biomembr. 44:619–622. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Malik AN and Czajka A: Is mitochondrial
DNA content a potential biomarker of mitochondrial dysfunction?
Mitochondrion. 13:481–492. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Yu M: Generation, function and diagnostic
value of mitochondrial DNA copy number alterations in human
cancers. Life Sci. 89:65–71. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Sotgia F, Whitaker-Menezes D,
Martinez-Outschoorn UE, Flomenberg N, Birbe RC, Witkiewicz AK,
Howell A, Philp NJ, Pestell RG and Lisanti MP: Mitochondrial
metabolism in cancer metastasis: Visualizing tumor cell
mitochondria and the ‘reverse Warburg effect’ in positive lymph
node tissue. Cell Cycle. 11:1445–1454. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lin CS, Lee HT, Lee SY, Shen YA, Wang LS,
Chen YJ and Wei YH: High mitochondrial DNA copy number and
bioenergetic function are associated with tumor invasion of
esophageal squamous cell carcinoma cell lines. Int J Mol Sci.
13:11228–11246. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Yuan Y, Chen Y, Zhang P, Huang S, Zhu C,
Ding G, Liu B, Yang T and Zhang A: Mitochondrial dysfunction
accounts for aldosterone-induced epithelial-to-mesenchymal
transition of renal proximal tubular epithelial cells. Free Radic
Biol Med. 53:30–43. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Hung WY, Huang KH, Wu CW, Chi CW, Kao HL,
Li AF, Yin PH and Lee HC: Mitochondrial dysfunction promotes cell
migration via reactive oxygen species-enhanced β5-integrin
expression in human gastric cancer SC-M1 cells. Biochim Biophys
Acta. 1820:1102–1110. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Ma J, Zhang Q, Chen S, Fang B, Yang Q,
Chen C, Miele L, Sarkar FH, Xia J and Wang Z: Mitochondrial
dysfunction promotes breast cancer cell migration and invasion
through HIF1α accumulation via increased production of reactive
oxygen species. PLoS One. 8:e694852013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Moreno-Sánchez R, Rodríguez-Enríquez S,
Marín-Hernández A and Saavedra E: Energy metabolism in tumor cells.
FEBS J. 274:1393–1418. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Ishikawa K, Takenaga K, Akimoto M,
Koshikawa N, Yamaguchi A, Imanishi H, Nakada K, Honma Y and Hayashi
J: ROS-generating mitochondrial DNA mutations can regulate tumor
cell metastasis. Science. 320:661–664. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Bonnard C, Durand A, Peyrol S, Chanseaume
E, Chauvin MA, Morio B, Vidal H and Rieusset J: Mitochondrial
dysfunction results from oxidative stress in the skeletal muscle of
diet-induced insulin-resistant mice. J Clin Invest. 118:789–800.
2008.PubMed/NCBI
|
|
33
|
Zhou G, Dada LA, Wu M, Kelly A, Trejo H,
Zhou Q, Varga J and Sznajder JI: Hypoxia-induced alveolar
epithelial-mesenchymal transition requires mitochondrial ROS and
hypoxia-inducible factor 1. Am J Physiol Lung Cell Mol Physiol.
297:L1120–L1130. 2009. View Article : Google Scholar : PubMed/NCBI
|